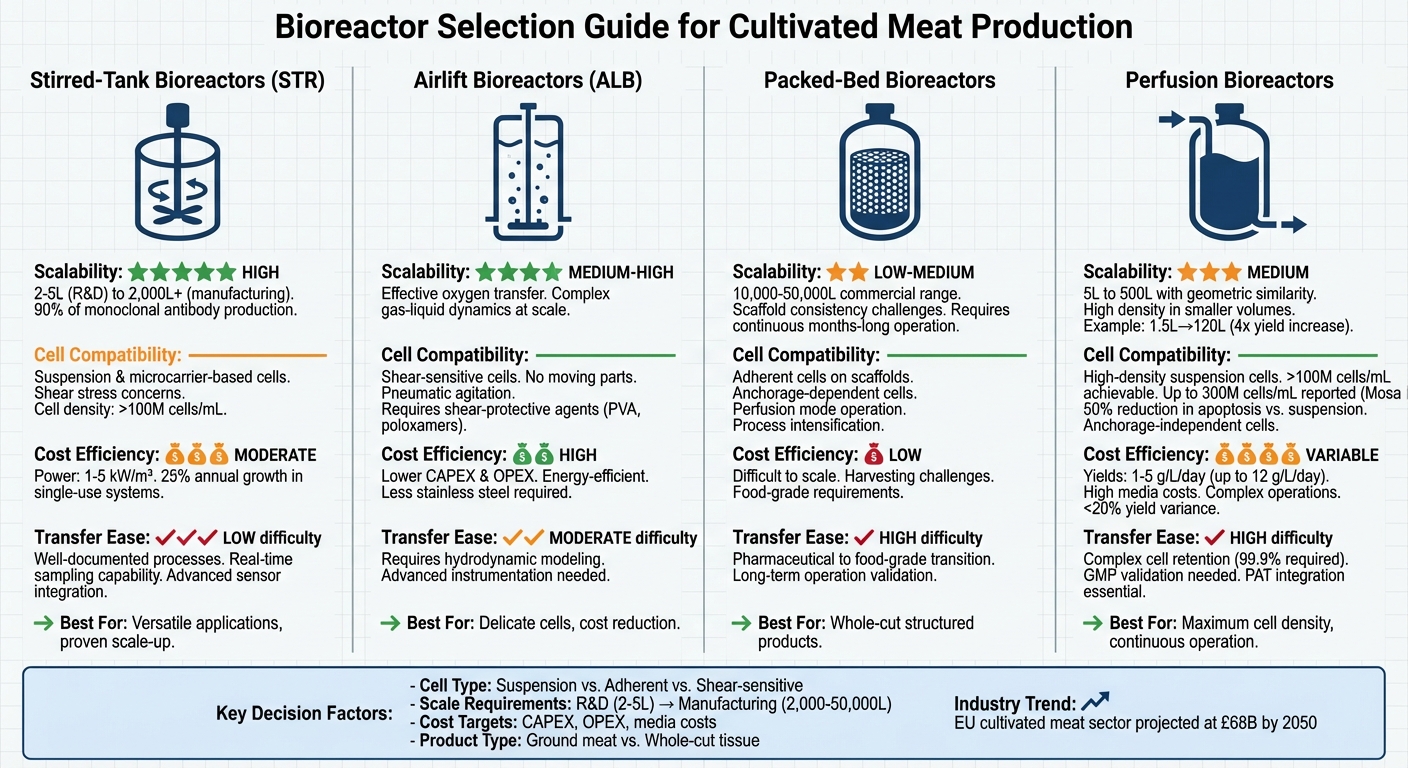
Scaling cultivated meat production requires choosing the right bioreactor to balance cell viability, cost efficiency, and process control. Each bioreactor type - stirred-tank, airlift, packed-bed, and perfusion - offers distinct advantages and challenges depending on the cell type and production goals.
- Stirred-Tank Bioreactors (STRs): Reliable for suspension and microcarrier-based cells, with proven scalability and control systems. However, they may cause shear stress on sensitive cells.
- Airlift Bioreactors (ALBs): Gentle on shear-sensitive cells and cost-efficient but require precise hydrodynamic modelling for scaling.
- Packed-Bed Bioreactors: Ideal for adherent cells using scaffolds but face challenges in scaling and harvesting.
- Perfusion Bioreactors: Achieve high cell densities with continuous media exchange but involve complex systems and higher operational costs.
Key takeaway: Selecting the right bioreactor depends on your specific cell type, scale-up needs, and cost targets. STRs are versatile and widely used, while ALBs and perfusion systems excel in protecting delicate cells and supporting high-density cultures. Packed-bed systems are more suited for structured products like whole cuts.
Quick Comparison:
| Bioreactor Type | Scalability | Cell Compatibility | Cost Efficiency | Challenges |
|---|---|---|---|---|
| Stirred-Tank (STR) | High | Suspension, microcarrier | Moderate | Shear stress on cells |
| Airlift (ALB) | Medium to High | Shear-sensitive cells | High | Complex scaling |
| Packed-Bed | Low to Medium | Adherent cells on scaffolds | Low | Scaling and harvesting |
| Perfusion | Medium | High-density suspension cells | Variable | Complex operation |
Choosing the right option ensures a smoother transition from research to manufacturing while meeting production and economic goals.
Bioreactor Types Comparison for Cultivated Meat Production
Dr. Marianne Ellis: Designing large-scale bioreactors and bioprocesses for cultivated meat
1. Stirred-Tank Bioreactors
Stirred-tank bioreactors (STRs) have been the backbone of industrial bioprocessing for more than half a century, supporting around 90% of monoclonal antibody production. This long-standing reliability makes them a natural choice for cultivated meat companies looking to scale up production. These cylindrical vessels, equipped with impellers, ensure uniform mixing of the medium, which helps distribute nutrients and oxygen evenly throughout the vessel [2].
Scalability
One of the standout features of STRs is their ability to scale seamlessly, from small R&D volumes of 2–5 litres to manufacturing capacities exceeding 2,000 litres [2][3]. Research shows that maintaining consistent oxygen transfer rates and power inputs per volume (typically 1–5 kW/m³) is key to ensuring high cell viability and productivity during scale-up [2]. However, achieving this requires careful attention to mixing conditions, especially to protect sensitive cells, as discussed below.
Cell Compatibility
Cultivated meat cells are particularly vulnerable to damage from the shear forces caused by impellers [5]. To address this, low-shear impeller designs, such as marine or hydrofoil impellers, are often used. These designs strike a balance between protecting delicate cells and maintaining effective mixing. More advanced options, like segmented pitched-blade impellers, can enhance mass transfer by up to 40% while minimising shear, creating an environment conducive to stem cell proliferation. STRs operating in perfusion modes can achieve cell densities exceeding 100 million cells/mL - comparable to specialised perfusion systems but with simpler sterilisation-in-place (SIP) and clean-in-place (CIP) processes. Validating cell compatibility at each stage, starting with 1–5 litre glass vessels before scaling up to stainless steel systems, is a common practice to ensure success.
Ease of R&D to Manufacturing Transfer
STRs also excel in bridging the gap between R&D and manufacturing. Their proven methodologies and extensive data make the transition more predictable [3]. Unlike alternative systems like airlift or packed-bed bioreactors, STRs allow real-time sampling and the integration of advanced sensors, which are essential for process analytical technology (PAT) and R&D optimisation. Modern STR setups typically include sensors to monitor dissolved oxygen, pH, temperature, nutrient levels, and cell density [2]. Computational fluid dynamics (CFD) modelling further simplifies the process by predicting shear and mixing dynamics at larger scales, potentially cutting experimental iterations in half.
The adoption of single-use STRs has surged in recent years, growing by 25% annually since 2020. These systems reduce contamination risks and simplify transitions between development and production, making them an increasingly popular choice. For cultivated meat companies, this combination of predictability, flexibility, and ease of integration underscores why STRs remain a cornerstone for scaling from R&D to full-scale manufacturing.
For those in search of dependable STR systems, the B2B marketplace Cellbase offers a curated selection of verified suppliers, transparent pricing, and tailored industry insights to support an efficient transition from research to production.
2. Airlift Bioreactors
Airlift bioreactors (ALBs) stand out as a gentler alternative to traditional stirred-tank bioreactors, making them an excellent option for cultivated meat production. Instead of relying on mechanical impellers for mixing, ALBs use pneumatic agitation to circulate the medium. This approach creates a more uniform environment while significantly reducing shear stress, which is crucial for protecting the delicate cells used in cultivated meat production [1]. With no moving parts like seals or motors, ALBs simplify mechanical design and provide a safer environment for sensitive cells [8].
Scalability
One of the strengths of ALBs is their ability to scale up efficiently, thanks to their effective oxygen transfer and mixing capabilities, which are essential for high-density cell cultures. This makes them well-suited as cultivated meat production transitions from lab-scale research to industrial manufacturing [1]. However, scaling up is not without its challenges. Oxygen delivery and carbon dioxide removal must align precisely with the metabolic demands of the cells at larger volumes [7]. Matt McNulty, GFI Research Fellow, highlights the potential of airlift reactors, stating:
Initial computational evaluations of alternative bioreactor geometries for cultivated meat suggest there may be value in further investigation (e.g., airlift reactor) [9].
At larger scales, changes in gas–liquid mass transfer and the emergence of localised gradients can complicate the process. This means that simply replicating hardware design does not guarantee biological outcomes will remain consistent [7]. ALBs, however, offer a promising framework for creating a cell-friendly environment at scale.
Cell Compatibility
The gas-driven circulation in ALBs results in a gentler environment, making them particularly suitable for cell types that are highly sensitive to shear stress [8]. For cultivated meat production, maintaining shear stress below harmful levels is critical, often requiring the addition of shear-protective agents like polyvinyl alcohol (PVA) or poloxamers [7]. When scaling up, it becomes essential to assess oxygen transfer capabilities in relation to the culture's peak Oxygen Uptake Rate (OUR), rather than relying solely on the volumetric oxygen mass-transfer coefficient (kLa) [7]. Equally important is monitoring the efficiency of carbon dioxide removal, as excessive CO₂ accumulation can hinder cell growth at larger scales [7].
Cost Considerations
Upstream bioprocessing is a major cost driver in cultivated meat production, with traditional designs often leading to inefficiencies [9]. ALBs offer a potential solution by lowering both capital (CAPEX) and operating costs (OPEX). This is achieved by reducing material needs, such as using less stainless steel and fewer sensors per unit [9]. The growing adoption of single-use airlift systems further streamlines operations by simplifying cleaning and sterilisation processes, though concerns about plastic waste remain [1]. These cost benefits make ALBs an attractive choice for scaling up production.
Ease of R&D to Manufacturing Transfer
ALBs are equipped with advanced instrumentation and control systems, which help address the technological challenges of large-scale bioprocessing. This makes it easier to transition from research and development to full-scale manufacturing [1]. For anchorage-dependent cells used in cultivated meat, the inclusion of microcarriers or scaffolds facilitates cell adhesion and growth [1]. By late 2024, airlift and bubble column bioreactors have joined stirred-tank reactors as some of the most commonly utilised systems in cultivated meat production [1].
For those navigating the shift from R&D to industrial manufacturing, platforms like Cellbase offer curated listings of bioreactor systems specifically designed to meet the unique needs of cultivated meat production.
3. Packed-Bed Bioreactors
Packed-bed bioreactors are specifically designed to support the production of cultivated meat, particularly for structured products like whole-cut tissue, as opposed to unstructured options like ground meat. Their design revolves around scaffolds that facilitate cell attachment, growth, and differentiation into tissue ready for consumption [12][13]. This focus on scaffolds plays a critical role in determining both the scalability and compatibility of these reactors in large-scale production.
Scalability
Scaling packed-bed bioreactors from small R&D setups to full-scale commercial production is no small feat. The industry is now working with bioreactors that can hold up to 50,000 litres, with most commercial facilities operating in the 10,000 to 50,000-litre range [11][12]. At these scales, specialised 3D scaffolds need to perform consistently and effectively, even at massive volumes [11]. Unlike the short-term operations typical in R&D, commercial production demands that these systems run uninterrupted for months. David Bell, Founder of Cultigen Group, highlights this challenge:
Suppliers who understand that your bioreactor needs to run continuously for months, not days [11].
Cell Compatibility
One of the strengths of packed-bed bioreactors is their ability to support anchorage-dependent cells. These reactors operate in perfusion mode, ensuring a constant supply of nutrients while removing waste. This setup promotes both high cell density and effective differentiation, aligning with the concept of "process intensification" [9][10]. Essentially, the reactor doubles as a platform for both cultivation and differentiation, optimising the entire process [9].
Ease of R&D to Manufacturing Transfer
Shifting from R&D to large-scale manufacturing introduces a new set of requirements for packed-bed bioreactors. They must transition from pharmaceutical-grade standards to food-grade systems to meet the specific needs of cultivated meat production [11]. Unlike drug development, the production of cultivated meat involves different regulatory and operational demands. The European Union, for instance, is expected to contribute £68 billion to the cultivated meat sector by 2050, underscoring the need for systems capable of long-term, continuous operation [11]. Platforms like Cellbase are helping bridge this gap by connecting companies with suppliers of validated food-grade scaffolds and bioreactors.
sbb-itb-ffee270
4. Perfusion Bioreactors
Perfusion bioreactors differ from traditional batch systems by continuously introducing fresh medium while simultaneously removing spent medium. This method allows for the cultivation of high cell densities over extended periods. Such continuous operation is particularly crucial for cultivated meat production, where reaching cell densities of over 100 million cells per millilitre is necessary for economic viability [2][3].
Scalability
Perfusion systems offer a clear advantage when moving from research to manufacturing scales. By maintaining geometric similarity, scaling up from 5 litres to 500 litres is achievable, with yields ranging from 1 to 5 grammes per litre per day and less than 20% yield variance in muscle cell cultures [2][3][5]. For example, Upside Foods successfully scaled their perfusion process from 1.5 litres in R&D to 120 litres using alternating tangential flow (ATF) perfusion. This adjustment increased yields fourfold to 12 grammes per litre per day for chicken cells [3][6]. Similarly, Mosa Meat reported achieving cell densities of 300 million cells per millilitre in their 500-litre pilot systems [3][6]. This reliable scalability ensures a controlled environment, which is critical for maintaining cell compatibility.
Cell Compatibility
Once scalability is established, maintaining cell viability becomes a priority. Perfusion bioreactors are particularly effective for anchorage-independent cells - commonly used in cultivated meat production - such as immortalised bovine and myoblast cell lines. These systems can support cell densities exceeding 100 million cells per millilitre by using microcarriers [4][14]. The continuous supply of nutrients and removal of waste reduces cellular stress. For instance, ATF perfusion has been shown to cut apoptosis rates by 50% compared to suspension cultures [4][14]. However, shear-sensitive cells, like primary myocytes, require careful handling, including the use of low-shear impeller designs, to preserve their viability.
Ease of R&D to Manufacturing Transfer
Perfusion bioreactors not only support high cell densities but also simplify the transition from R&D to manufacturing. Scaling up is straightforward, as process parameters such as flow rates and cell retention devices scale predictably using dimensionless numbers like the perfusion rate index [2][5]. A key challenge lies in validating cell retention devices - for example, achieving 99.9% retention in hollow-fibre modules - and ensuring compliance with regulatory standards for GMP [2][5]. Experts from the Good Food Institute recommend incorporating process analytical technology (PAT), such as online biomass sensors, to maintain over 95% parameter consistency during scale-up [5][15]. Platforms like Cellbase further streamline the process by connecting procurement teams with suppliers of PAT-compatible perfusion systems, simplifying validation and manufacturing transfer.
Advantages and Disadvantages
When scaling cultivated meat production from research to manufacturing, each bioreactor type comes with its own set of strengths and challenges. Stirred-tank bioreactors are widely regarded as the industry standard for rapid scaling, thanks to their reliable control systems. However, their mechanical agitation results in higher energy usage as volumes increase [1]. The following table provides a clear comparison of key bioreactor types.
Airlift bioreactors, on the other hand, offer cost-saving benefits due to pneumatic agitation, which eliminates moving parts and reduces energy consumption. They are particularly suitable for shear-sensitive cultivated meat cells. The trade-off? Scaling these systems requires precise hydrodynamic modelling, adding a layer of complexity [1].
Packed-bed bioreactors are particularly effective for adherent cells that grow on scaffolds. However, they face significant hurdles when it comes to scaling up production [1].
Here’s a breakdown of how these systems perform across key parameters:
| Bioreactor Type | Scalability | Cell Compatibility | Cost-Effectiveness | Transfer Difficulty |
|---|---|---|---|---|
| Stirred-Tank (STR) | High; widely used for large-scale production | Suitable for suspension and microcarrier-based adherent cells | Moderate; energy demands increase with scale | Low: Well-documented and easy to control |
| Airlift | Medium to High | Best for shear-sensitive cells due to pneumatic agitation | High; energy-efficient with no moving parts | Moderate: Requires advanced hydrodynamic modelling |
| Packed-Bed | Low to Medium | Ideal for adherent cells on scaffolds | Low; difficult to scale and harvest | High: Challenges in scaling and harvesting processes |
| Perfusion | Medium (high density achievable in smaller volumes) | Supports high-density suspension cultures | Variable; yields are high, but media and operational costs can be significant | High: Complex cell retention systems required |
Another noteworthy trend is the adoption of single-use technology, which simplifies manufacturing processes. These systems minimise the need for extensive validation and reduce capital costs associated with cleaning infrastructure [1].
Conclusion
Stirred-tank bioreactors are a solid choice for suspension cells or microcarrier systems, thanks to their well-established scale-up capabilities and reliable control systems [1].
For adherent cells, modified stirred-tank systems equipped with microcarriers or packed-bed reactors provide the right environment for effective attachment and growth [1].
When working with shear-sensitive cells, airlift bioreactors stand out. They use pneumatic agitation to reduce mechanical stress while ensuring efficient oxygen transfer, making them a better fit for these delicate cell types [1]. This range of reactor designs highlights the diverse requirements of different cell types and production goals.
Perfusion bioreactors are designed to achieve high cell densities in smaller volumes through continuous media exchange. That said, they come with added complexity, requiring advanced cell retention systems and meticulous operation [1].
Single-use bioreactors, on the other hand, eliminate the need for laborious cleaning and sterilisation, speeding up processes and simplifying workflows [1]. Each type of bioreactor plays a vital role in creating a smooth transition from research to manufacturing.
FAQs
How do I choose a bioreactor for my specific cultivated meat cell type?
When selecting a bioreactor for your cultivated meat production, it's essential to align its design with the specific needs of your cell type. For instance, stirred-tank bioreactors work well for bovine muscle cells because they offer controlled shear forces and are suitable for scaling up production.
To ensure cell viability, it's crucial to understand how sensitive your cells are to shear stress. Tools like computational fluid dynamics (CFD) can be invaluable in this process, helping you predict and manage the effects of scaling up. Focus on matching the bioreactor's design features - such as its mixing method, shear protection mechanisms, and ability to maintain optimal environmental conditions - to the requirements of your production goals.
What should I measure during scale-up to maintain cell viability and productivity?
To maintain optimal cell viability and productivity during scale-up, it's essential to keep a close eye on several key parameters. These include sterility, as any contamination can derail the entire process, and environmental conditions like temperature, pH, and oxygen levels, which directly affect cell growth.
Additionally, managing shear stress is crucial to prevent cell damage, while ensuring effective nutrient delivery and waste removal keeps the cells healthy and thriving. Lastly, mixing efficiency plays a significant role in maintaining uniform conditions throughout the system. Together, these factors are central to achieving consistent results in cultivated meat production.
When is single-use better than stainless steel for manufacturing transfer?
Single-use bioreactors work well for smaller-scale operations, early development stages, or situations where flexibility and fast turnaround matter most. They come with benefits like lower initial costs, faster setup times, and no need for extensive cleaning, making them a practical choice for pilot projects or limited production runs.
On the other hand, stainless steel systems shine in large-scale manufacturing. With capacities exceeding 20,000 litres, they provide greater durability and lower costs over time. However, they do require a higher upfront investment and can be more complex to maintain.











