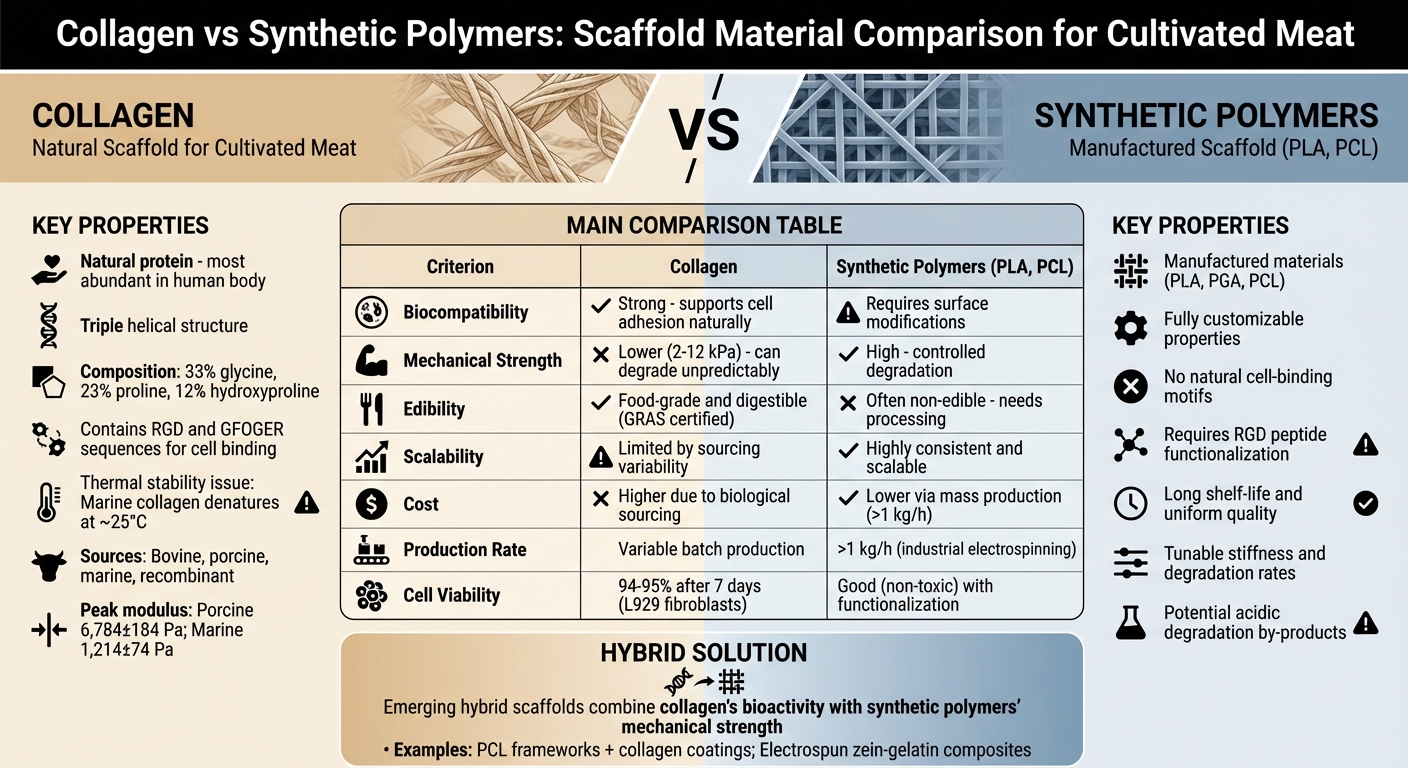
When producing cultivated meat, scaffolds are essential for creating structured products like steaks or chicken breasts. Two main materials dominate this space: collagen and synthetic polymers. Here’s a quick breakdown:
- Collagen: A natural protein offering strong bioactivity that supports cell growth and attachment. It mimics the extracellular matrix but struggles with stability, strength, and cost.
- Synthetic Polymers: Manufactured materials like PLA and PCL provide consistent strength and scalability. However, they lack natural cell-binding properties and often aren't food-grade.
The decision between these materials depends on priorities like biocompatibility, mechanical performance, food safety, and production costs. Hybrid scaffolds, combining both, are emerging as a solution to balance bioactivity and mechanical strength.
Quick Comparison
| Criterion | Collagen | Synthetic Polymers |
|---|---|---|
| Biocompatibility | Strong, supports cell adhesion | Requires surface modifications |
| Strength | Lower, can degrade unpredictably | High, with controlled degradation |
| Edibility | Food-grade and digestible | Often non-edible, needs processing |
| Scalability | Limited by sourcing variability | Highly consistent and scalable |
| Cost | Higher due to biological sourcing | Lower via mass production |
Hybrid scaffolds aim to combine the benefits of both materials, offering a way forward for cultivated meat production.
Collagen vs Synthetic Polymers Scaffold Comparison for Cultivated Meat
Dr. Amy Rowat: Marbling cultivated meat with hydrogel scaffolds
Collagen Scaffolds: Properties and Characteristics
Collagen stands out as the most abundant protein in the human body [4], making it an ideal choice for replicating the extracellular matrix in cultivated meat production. Its triple helical structure - composed of three α-chains with repeating glycine-X-Y sequences - provides the tensile strength necessary for cell attachment and tissue organisation. These collagen molecules naturally assemble into tropocollagen fibrils and fibres, closely imitating the architecture of muscle tissue, which is essential for myoblast maturation.
What makes collagen particularly effective is its natural bioactivity, which sets it apart from other scaffold materials. Specific amino acid sequences, such as RGD (arginyl-glycyl-aspartic acid) and GFOGER, act as ligands for cell surface integrins, triggering pathways that promote cell growth and differentiation. As noted by PatSnap:
Collagen is inherently recognised by the body's cells, which facilitates cell attachment and proliferation [1].
This natural recognition makes collagen scaffolds highly effective in supporting muscle cell alignment and fusion - key factors in achieving the texture required for structured cultivated meat products.
Collagen's composition - approximately 33% glycine, 23% proline, and 12% hydroxyproline [4] - is central to its structural properties. However, it does have nutritional drawbacks, as it lacks the essential amino acid tryptophan [3]. Its edibility and GRAS (Generally Recognised as Safe) certification make it suitable for direct use in cultivated meat. These structural and bioactive properties contribute to several key benefits.
Advantages of Collagen Scaffolds
One of collagen's standout benefits is its excellent biocompatibility. As a natural component of the extracellular matrix, cells easily recognise and interact with collagen scaffolds without the need for additional surface modifications. For example, in bioprinting experiments, L929 fibroblasts encapsulated in collagen hydrogels maintained a viability rate of 94% to 95% after seven days of culture [5], showing its ability to support cell survival and growth effectively.
Collagen's fibrillar structure supports muscle cell alignment and the fusion of cells into multinucleated myotubes, which are essential for creating structured meat. This hierarchical organisation, from molecules to fibres, helps replicate the complex three-dimensional environment required for authentic meat texture. Additionally, collagen's mechanical properties can be fine-tuned using enzymatic or chemical cross-linking techniques, allowing researchers to match the stiffness of native muscle tissue, which typically ranges from 2–12 kPa [3].
Another advantage is its versatility in sourcing. Collagen can be derived from bovine, porcine, marine, or recombinant origins, offering flexibility for different applications and catering to varying consumer preferences.
Limitations of Collagen Scaffolds
Despite its benefits, collagen also has notable limitations that affect its practical use in cultivated meat.
One major challenge is its stability. Collagen loses its triple helical structure and bioactivity when it transforms into gelatin above its melting point. This issue is particularly evident with marine-derived collagen. For instance, research on Blue Grenadier (Macruronus novaezelandiae) reveals that marine collagen denatures at around 25°C - 12°C lower than porcine-derived collagen [5]. As highlighted by Nature:
Fish muscle collagen has low thermal stability, resulting in structure loss during cooking. This phenomenon is responsible for the scaly texture of cooked fish due to collagen fusion [3].
Another limitation is collagen's mechanical weakness. Compared to synthetic polymers, collagen scaffolds generally lack the mechanical strength needed for load-bearing applications or maintaining structural integrity in thick, multilayered constructs [1][2]. For instance, methacrylated porcine collagen has demonstrated peak moduli of up to 6,784 ± 184 Pa, while marine-derived collagen reached only 1,214 ± 74 Pa under the same conditions [5].
Sourcing variability also poses challenges. Animal-derived collagen carries risks such as disease transmission (e.g., BSE or FMD) and potential immunogenic reactions. Additionally, its degradation rate can be inconsistent and unpredictable [1]. Recombinant collagen, produced through fermentation, can address these concerns but adds complexity and cost. Hydroxyproline content further varies significantly between sources: while warm-blooded animals like pigs typically have around 10% hydroxyproline, ensuring stability at 37°C, Antarctic Ice-fish collagen contains only about 4.5%, with a melting temperature as low as 6°C [5].
Synthetic Polymer Scaffolds: Properties and Characteristics
Synthetic polymers like polylactic acid (PLA), polyglycolic acid (PGA), and polycaprolactone (PCL) stand out due to their fully customisable physical and chemical properties. Unlike collagen, which is derived from biological sources, these materials are manufactured, allowing for precise control over their characteristics. However, synthetic polymers lack natural cell-binding motifs, which means they need functionalisation - such as adding RGD peptides - to support cell adhesion effectively [3][6]. Despite this, their tunable mechanical properties and consistent production make them an appealing choice for large-scale applications. For instance, industrial electrospinning systems can produce polymer scaffolds at rates exceeding 1 kg/h [3].
One of the key strengths of synthetic polymers is their mechanical robustness, which far surpasses collagen. Their properties can be tailored to meet the specific requirements of various tissues. Additionally, their degradation rates can be precisely controlled, ensuring the scaffold supports tissue formation without leaving unwanted residues. These features make synthetic polymers a compelling option for cultivated meat production.
Advantages of Synthetic Polymers
Synthetic polymers offer a level of reproducibility and scalability that natural materials struggle to match. As noted in Nature:
Synthetic polymers also have a major advantage over other materials because they can be produced in large, uniform quantities and have a long shelf-life [3].
This consistency eliminates the batch-to-batch variability common with animal-derived materials and addresses concerns about disease transmission or ethical issues tied to biological sourcing. For companies aiming for commercial-scale production of cultivated meat, this reliability is crucial for meeting regulatory standards and maintaining consistent quality.
Another major benefit is their customisability. Cell Guidance Systems highlights this:
Synthetic biomaterials allow an extra layer of fine control over the properties of the material. Stiffness and charge can be easily tuned for the particular cell type or tissue [6].
This flexibility enables the creation of scaffolds with varying mechanical properties within a single structure. For example, researchers can design scaffolds that support both muscle and fat tissue development by combining regions of different stiffness. Synthetic polymers can also be engineered to achieve high porosity with small pore sizes, promoting efficient nutrient diffusion and waste removal in dense cell cultures. Their mechanical durability makes them particularly suitable for structured meat products that require load-bearing capacity, where collagen may fall short.
Limitations of Synthetic Polymers
Despite their advantages, synthetic polymers come with challenges. The most notable issue is their lack of inherent bioactivity. Unlike collagen, which cells naturally recognise, synthetic polymers require surface modifications or functionalisation to support cell adhesion and growth. This often involves adding bioactive molecules like RGD peptides or applying protein coatings, which increases both the complexity and cost of production [2][3].
Another challenge is related to their degradation by-products. While their degradation rates can be controlled, materials like PLA and PGA break down into acids that may cause inflammation if not carefully managed [1]. This requires precise engineering to ensure the degradation process aligns with tissue formation without inducing cellular stress.
A particularly critical issue for cultivated meat applications is edibility. Many synthetic polymers commonly used in medical tissue engineering are not classified as GRAS (Generally Recognised as Safe) for food consumption [2][3]. As a result, these materials often need to be removed from the final product, adding extra processing steps and increasing costs. While progress is being made towards developing food-safe synthetic polymers, current options frequently require dissociating cells from the scaffold before the meat reaches consumers. This creates a significant hurdle for commercial-scale production and highlights the trade-offs involved in choosing scaffold materials for cultivated meat.
Collagen vs Synthetic Polymers: Side-by-Side Comparison
This section breaks down the key trade-offs between collagen and synthetic polymer scaffolds, focusing on factors like biocompatibility, mechanical properties, edibility, cost, and scalability.
When it comes to biocompatibility, collagen stands out. Its natural bioactivity, including RGD motifs that promote cell adhesion, gives it an edge over synthetic polymers. These polymers are inert by nature and require surface modifications to enable cell interactions.
Mechanical properties are another area of contrast. Native muscle tissue typically has an elastic modulus between 10 and 100 kPa [2]. Collagen's lower strength can result in scaffold failure during processing [1]. On the other hand, synthetic polymers offer adjustable strength and predictable degradation, making them better suited for specific tissue requirements. While collagen degrades into harmless amino acids, synthetic polymers can release acidic by-products, potentially causing inflammation [1].
The edibility of these materials is a practical concern. Collagen and its derivative, gelatin, are inherently food-grade and digestible, making them easy to integrate into final products. Many synthetic polymers, however, are not classified as GRAS (Generally Recognised as Safe) for food use. This often necessitates additional removal steps, increasing both complexity and cost [2].
Here’s a quick comparison of these materials:
| Criterion | Collagen Scaffolds | Synthetic Polymer Scaffolds (e.g., PLA, PCL) |
|---|---|---|
| Biocompatibility | Excellent; inherent RGD motifs support cell adhesion | Good (non-toxic) but lacks inherent bioactivity |
| Mechanical Properties | Low strength; unpredictable degradation | High strength; tunable and predictable degradation |
| Cost | High; dependent on biological sourcing | Lower; mass-produced via chemical synthesis |
| Scalability | Limited by animal sources and batch variability | High; consistent and reproducible manufacturing |
| Edibility | Fully edible and food-grade | Generally non-edible; requires processing or regulatory clearance |
| Risk Factors | Potential for immunogenicity or pathogens | Potential for inflammatory degradation by-products |
When considering scalability and cost, synthetic polymers often have the upper hand. They can be manufactured in large, uniform batches with consistent properties. Collagen, however, varies depending on its biological source, leading to inconsistencies and contamination risks [1]. Recombinant, animal-free collagen offers a potential solution, but its current production costs remain a barrier [3]. For companies navigating these challenges, platforms like Cellbase help connect researchers with trusted suppliers for scaffold materials.
sbb-itb-ffee270
Hybrid Scaffolds: Combining Collagen and Synthetic Polymers
Hybrid scaffolds bring together the biological advantages of collagen with the strength and durability of synthetic polymers, addressing the shortcomings of using each material on its own. This combination creates a balance between bioactivity and mechanical stability.
Synthetic polymers like polycaprolactone (PCL) act as a sturdy backbone, maintaining the scaffold's structural integrity. Meanwhile, collagen coatings provide the necessary cues for cell adhesion. For example, researchers have successfully used PCL frameworks coated with fibrillated collagen to improve myoblast alignment. Similarly, electrospun zein-gelatin composites have been shown to not only support aligned myotube formation but also replicate the texture of cooked meat, offering exciting possibilities for cultured meat applications [2].
"Scaffolds serve not merely as passive supports but as bioactive architectures that actively regulate cellular behavior." - Sun Mi Zo et al., School of Chemical Engineering, Yeungnam University [2]
Hybrid scaffolds also address the challenge of synchronising scaffold degradation with tissue growth. If a scaffold degrades too quickly, it can leave developing tissue vulnerable and unsupported [1]. By carefully adjusting the degradation rate of synthetic polymers, hybrid systems ensure the scaffold remains intact long enough for tissue to form, all while preserving collagen's biological activity. For researchers and companies looking to source these materials, platforms like Cellbase provide access to verified suppliers offering both collagen derivatives and synthetic polymers tailored for hybrid scaffold fabrication.
Applications and Future Developments
Cultivated meat companies are employing a range of scaffold strategies to refine their products. For example, Aleph Farms has adopted a "bottom-up" approach using 3D bioprinting to create beef steaks. Their method relies on a bioink containing pea protein scaffolds to support muscle and fat cells [8]. Wildtype, on the other hand, uses plant-based scaffolds to produce sushi-grade cultivated salmon [8]. Interestingly, companies like UPSIDE Foods and 3DBT have taken a different route by developing scaffold-free methods. UPSIDE's FDA-approved cultivated chicken and 3DBT's cultivated pork fillet are labelled as "100% meat", avoiding plant-based supports entirely [8]. These varied approaches highlight the ongoing balance between maintaining natural bioactivity and achieving engineered strength.
The use of food-grade materials is becoming more widespread. Production capacities for hydrogel precursors such as agarose, gellan, and xanthan are already sufficient to support the manufacture of 1–3 million tonnes of cell-free scaffolds annually [7]. Additionally, companies are increasingly turning to specialised B2B providers like Matrix Food Technologies and Gelatex for high-quality, food-grade scaffolds [8].
"Scaffolds intended for food applications must not only satisfy the functional requirements of tissue engineering but must also be edible, non-toxic, and compatible with food regulatory standards." - Sun Mi Zo et al., Yeungnam University [2]
Advances in functionalisation techniques are improving scaffold performance even further. Methods like TEMPO-mediated oxidation for cellulose, enzymatic crosslinking with transglutaminase, and the integration of RGD motifs are being utilised to enhance cell-material interactions [2][3]. Recent research has showcased practical progress. For example, in August 2025, Eom et al. developed multichannel-grooved scaffolds using GelMA hydrogel bioinks, which significantly enhanced the myogenic differentiation of MSTN knock-out cells [2]. Similarly, Melzener et al. created edible scaffolds by weaving alginate fibres coated with zein, which successfully guided C2C12 myoblasts into aligned myotubes [2].
As these technologies advance, sourcing high-quality, GRAS-approved materials becomes increasingly important. Procurement teams can now rely on platforms like Cellbase to find verified suppliers of collagen derivatives and synthetic polymers. With global meat production exceeding 300 million tonnes in 2020 [7], scaling from laboratory prototypes to commercial production will depend on securing materials that meet regulatory standards and align with consumer demands for clean-label products.
Conclusion
Deciding between collagen and synthetic polymers comes down to production priorities. Collagen brings natural bioactivity to the table but falls short in strength, while synthetic polymers offer adaptable mechanical properties at the cost of lacking inherent bioactivity [1][2][3].
Hybrid scaffolds, which blend natural biopolymers with synthetic reinforcements, aim to strike a balance. They address the long-standing "stiffness-degradability" trade-off by combining bioactivity with structural stability [2].
Material selection needs to align with biological requirements, such as achieving an elastic modulus of 10–100 kPa [2], while also considering production constraints. The ideal scaffold should mimic the mechanical characteristics of the target tissue and comply with food safety standards like GRAS approval [2][3].
One of the biggest challenges for scaling cultivated meat production is securing high-quality, food-grade scaffold materials. Platforms like Cellbase are stepping in to help procurement teams find verified suppliers for collagen derivatives, synthetic polymers, and hybrid materials tailored to cultivated meat. This shift allows producers to move away from expensive biomedical-grade materials and opt for food-safe, cost-effective alternatives.
FAQs
When should cultivated meat producers choose collagen over synthetic polymers?
Collagen works exceptionally well when aiming to mimic the structure of natural muscle tissue and improve tenderness. Being a natural protein, it aids in tissue development, is biodegradable, compatible with biological systems, and safe to consume. While synthetic polymers can be tailored and scaled up, they often need additional strengthening and can face regulatory hurdles. Collagen stands out for uses where texture, compatibility with biological systems, and food safety are key priorities.
How can synthetic polymer scaffolds be made food-safe and edible?
Synthetic polymer scaffolds can become food-safe and edible by opting for non-chemical crosslinking methods. Techniques like physical or enzymatic crosslinking eliminate the risk of harmful chemical residues. Using food-grade polymers, such as gelatin, alginate, or plant-based proteins, adds another layer of safety. These approaches ensure the scaffolds not only support cell growth but also align with regulatory requirements and consumer expectations for cultivated meat production.
What are hybrid scaffolds, and how do they improve on single-material scaffolds?
Hybrid scaffolds are composite materials made by combining substances like collagen with nanocellulose. These materials are designed to improve the performance of scaffolds used in cultivated meat production. Single-material scaffolds often struggle with issues like weak mechanical strength and poor stability. Hybrid scaffolds solve these problems by offering greater strength, adjustable porosity, and enhanced biochemical functionality. These features create an environment that supports cell growth and tissue development, making hybrid scaffolds a better option for producing structured, meat-like tissues.











