Genetic stability is critical for cultivated meat production. Without it, cell lines may mutate, leading to inconsistent quality, safety risks, and production failures. Scaling from thousands to trillions of cells amplifies these risks, making robust quality control systems essential. Regulators like the FDA and EMA require proof of stability before approving products, as even minor genetic changes can trigger allergenic or harmful outcomes.
Key challenges include genetic drift, mutation accumulation, and oncogene activation. These issues arise from extended cell passaging, selective pressures, and environmental stressors during production. Advanced testing methods, such as karyotyping, SNP arrays, and next-generation sequencing (NGS), help detect and address these risks. Preventive strategies like structured cell banking and targeted genetic engineering further safeguard against instability.
Producers must integrate quality control into every stage of production - from cell banking to large-scale bioreactors. Tools like STR profiling, contamination tests, and functional assays ensure consistency and safety. Platforms like Cellbase connect producers with resources to meet strict industry standards. Genetic stability is not just a regulatory requirement; it’s the foundation for reliable, scalable cultivated meat production.
Accelerating Cell Line Development from DNA to Master Cell Bank - AGC Biologics
Common Challenges in Maintaining Genetic Stability
Ensuring genetic stability throughout the production cycle of cultivated meat is no small feat. The sheer scale of production introduces plenty of opportunities for genetic changes to develop and spread. Recognising these challenges is key to putting effective quality control systems in place.
Genetic Drift and Mutation Accumulation
Extended cell passaging is a major source of genomic instability in cultivated meat production. Immortalised cell lines, by their nature, are prone to genomic changes, which can lead to spontaneous mutations during long-term culture [6][5]. As cells go through multiple population doublings, errors in DNA replication build up, resulting in diverse cell populations and, potentially, a loss of function. Christopher Frye and Luhong He from BioPharm International highlight this issue:
Clonally-derived CHO cell lines have often been observed to diverge, becoming a heterogeneous population over long periods of sub-culturing [6].
In industrial settings, around 20% of production cell lines show considerable transgene heterogeneity over successive generations [6]. These mutations can occur early on, during DNA replication after transfection, or due to errors when foreign genes are integrated into the host genome [5].
Selective pressures add another layer of complexity. Agents like antibiotics and metabolic markers (e.g., MTX) used to stabilise cell lines can actually increase mutation rates [6][5]. In some cases, the higher the concentration of these agents, the higher the mutation rate [6]. Environmental stressors - such as nutrient shortages, suboptimal culture conditions during scale-up, and physical stress from expansion - can further destabilise genetic integrity [6][5].
Shuai Wang, Director of Cell Line Development at WuXi Biologics, notes:
Mutation levels are likely to change over cell passaging because of Chinese hamster ovary (CHO) cells' genomic plasticity [5].
Epigenetic changes also play a role. Transgenes can be partially or completely lost or silenced during the culture process, affecting long-term stability. These accumulated mutations not only harm cell function but also increase the risk of activating oncogenes.
Oncogene Activation Risks
Oncogene activation represents a critical safety concern for cultivated meat producers, as it can compromise entire production batches. Genetic instability can lead to oncogene activation through mechanisms like hypermethylation, which can result in tumour-like profiles [3][1]. The rapid expansion required in production further increases the likelihood of accumulating these harmful mutations [5][6].
This is a well-recognised challenge. According to the International Consortium for Innovation & Quality in Pharmaceutical Development (IQ), 67% of respondents believe genetic mutations pose a greater threat than amino acid misincorporation during production [5]. A case from May 2024 illustrates the severity of this issue: WuXi Biologics discovered that 43% of clones from a cell-line development programme carried the same genetic point mutation. The root cause? A 2.1%–2.2% variant level in the plasmid DNA used during transfection, which went undetected by traditional Sanger sequencing. To address this, the company incorporated Next-Generation Sequencing (NGS) into its quality control processes to catch such variants early [5].
Early detection of genetic abnormalities is crucial, as oncogenic changes can compromise entire batches. Standard G-band karyotyping can identify abnormal subpopulations with as little as 14% mosaicism in twenty cell metaphases [1]. More advanced techniques like NGS can detect genetic mutations in clonal cells with a sensitivity of 0.5% [5].
DNA methylation analysis is another valuable tool for assessing tumourigenic potential:
Because DNA methylation levels and hypermethylation of certain genes occurs in cancer initiation and progression, the analysis of DNA methylation profiles might provide complementary information about tumourigenic potential of cells [3].
The real challenge lies in implementing robust monitoring systems capable of identifying these changes before they compromise safety. For cultivated meat producers, maintaining genetic stability during rapid cell expansion demands advanced quality control measures. Specialised platforms like Cellbase are increasingly stepping in to meet this need, as detailed in the next section.
Quality Control Testing Methods
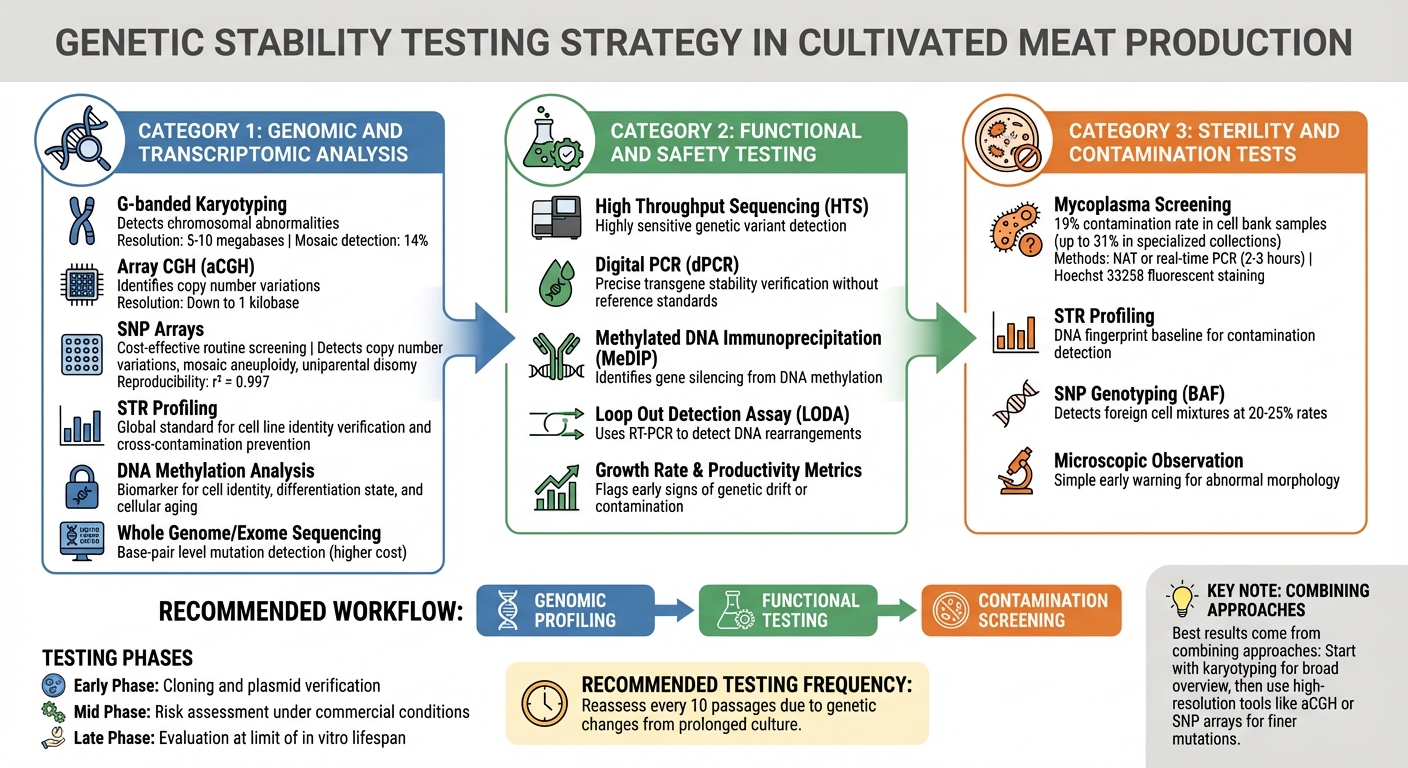
Quality Control Testing Methods for Genetic Stability in Cultivated Meat Cell Lines
Identifying genetic instability before it impacts production requires a multi-layered testing strategy. Cultivated meat producers rely on methods that detect everything from large chromosomal changes to single-base mutations. The choice of techniques depends on the risks present at each production stage. These genomic tools act as the first checkpoint, followed by detailed functional and contamination assessments.
Genomic and Transcriptomic Analysis
G-banded karyotyping is a key tool for identifying major chromosomal issues like numerical abnormalities and large structural rearrangements, such as translocations. While it can detect mosaic levels of around 14%, its resolution is limited to changes of 5–10 megabases, meaning smaller alterations might go unnoticed [1].
Array Comparative Genomic Hybridisation (aCGH) offers higher resolution, identifying copy number variations down to 1 kilobase. Similarly, Single Nucleotide Polymorphism (SNP) arrays provide a cost-effective option for routine screening, detecting copy number variations, mosaic aneuploidy, and uniparental disomy. Validation studies have shown SNP arrays to be highly reproducible, with B Allele Frequency (BAF) measurements achieving an impressive r² = 0.997 [8][1].
Rocio Aguilar-Quesada from the Andalusian Public Health System Biobank highlights the value of combining methods:
Karyotyping remains a versatile assay, especially when complemented with high-resolution testing [1].
Short Tandem Repeat (STR) profiling is the global standard for verifying cell line identity and preventing cross-contamination [1][9]. Meanwhile, DNA methylation analysis serves as a biomarker for cell identity, differentiation state, and cellular ageing [1]. For base-pair level mutation detection, whole genome or exome sequencing is an option, though it comes with higher costs compared to array-based methods [1].
The best results come from combining these approaches. Start with karyotyping for a broad overview, then use high-resolution tools like aCGH or SNP arrays to capture finer mutations. Regular reassessment - ideally every 10 passages - is critical, as prolonged culture can lead to genetic changes [10]. These genomic insights are then supplemented with functional assays to ensure consistent cell behaviour throughout production.
Functional and Safety Testing
Genomic profiling alone isn’t enough. Functional tests confirm that cells maintain their intended characteristics during expansion. Metrics like growth rates and productivity can flag early signs of genetic drift or contamination [9].
High Throughput Sequencing (HTS) is highly sensitive for detecting genetic variants, while digital PCR (dPCR) precisely verifies transgene stability without needing reference standards [11]. Christopher Frye and Luhong He from BioPharm International stress the importance of this step:
The production cell line is the foundation of any bioprocess, and thus, appropriate genetic characterisation of the production cell line is absolutely crucial to the success of process development [6].
Epigenetic stability is also critical. Methylated DNA Immunoprecipitation (MeDIP) helps identify gene silencing caused by DNA methylation, a common reason for productivity decline [7]. Tools like the Loop Out Detection Assay (LODA) use RT-PCR to detect DNA rearrangements, such as when a gene of interest is deleted while the selectable marker remains [7].
Testing should align with the production phase: early-phase tests focus on cloning and plasmid verification, mid-phase tests assess risks under commercial conditions, and late-phase tests evaluate cells at the limit of their in vitro lifespan [6]. Starting experiments with fresh, low-passage cells from a master cell bank reduces the risk of genetic drift [9].
Sterility and Contamination Tests
Contamination testing is essential to avoid factors that could destabilise genetics. Mycoplasma is particularly concerning, as it alters cell metabolism and behaviour without causing visible changes in the culture [1]. Studies reveal mycoplasma contamination in 19% of cell bank samples, with some specialised collections showing rates as high as 31% [1].
Regular mycoplasma screening using sensitive Nucleic Acid Amplification Techniques (NAT) or real-time PCR can provide semi-quantitative results within 2–3 hours [1]. Hoechst 33258 fluorescent staining is another method, revealing characteristic extracellular fluorescence patterns [9].
STR profiling establishes a DNA fingerprint for cell lines, acting as a baseline to detect cross-contamination [9]. Additionally, SNP genotyping using B Allele Frequency (BAF) distributions can identify contamination from other cell lines, detecting foreign cell mixtures at rates of 20–25% [8].
Microscopic observation is a simple yet effective early warning tool, as abnormal cell morphology often signals culture issues [9]. Implementing a Quality Management System, such as ISO 9001:2015, along with Good In Vitro Method Practices (GIVIMP), helps maintain standardised and reproducible culture conditions, reducing the risk of genomic instability [10].
For cultivated meat producers needing access to specialised testing equipment and materials, platforms like Cellbase offer a marketplace connecting industry professionals with verified suppliers of analytical instruments, reagents, and testing services.
sbb-itb-ffee270
Prevention Strategies for Genetic Instability
Detecting genetic instability is one thing; preventing it is another challenge entirely. To keep genetic stability intact, cultivated meat producers need well-thought-out systems that stop cell lines from drifting before issues arise. Two key strategies lead the way here: structured cell banking and targeted genetic engineering. Together, these approaches directly tackle the risks of genetic drift and oncogene activation.
Cell Banking and Cryopreservation
Mutation accumulation is a real concern, so a reliable cell banking system is a must. The industry standard involves a two-tier system: a Master Cell Bank (MCB) and a Working Cell Bank (WCB). This setup ensures a consistent starting point for production. Limiting the number of passages is crucial, as each passage increases the chance of mutations. By storing cells in liquid nitrogen, biological activity is effectively paused, reducing the risk of genetic changes during storage.
Instead of tracking time, cell age is measured by population doublings. For instance, a typical 5,000-litre production bioreactor involves around 30 population doublings[6]. To maintain genetic consistency, commercial manufacturing limits this number to between 45 and 60 doublings[6].
Screening methods like RT-PCR and single-cell qPCR can catch issues early, such as unusual mRNA splicing or transgene variability. Cell lines showing wide variability in copy numbers should be discarded to avoid future problems.
Quality control is non-negotiable. Alarmingly, studies have found that up to 31% of cell lines in some banks were contaminated with mycoplasma[3]. To prevent this, STR profiling is used to confirm the authenticity of cell lines throughout the banking process. As highlighted by the FSA Research and Evidence:
Because the banked cells are the starting material for the final product, high standards might be required by regulators in the future to ensure a safe meat product of high quality[2].
Genetic Engineering for Stability
Genetic engineering provides another layer of defence by directly enhancing cell line stability. Techniques like targeted integration (TI), particularly Recombinase-Mediated Cassette Exchange (RMCE), allow precise insertion of transgenes into specific genomic locations. This approach avoids the unpredictability of random integration, where position effects and copy number instability can wreak havoc. Although RMCE in CHO cells has efficiency rates below 0.1%[12], the resulting clones are more predictable and stable.
The choice of expression system matters too. For example, the Glutamine Synthetase (GS) system typically results in about five transgene copies per cell, whereas the Dihydrofolate Reductase (DHFR) system can amplify copy numbers up to 1,000[6]. While higher copy numbers might sound appealing, they increase the likelihood of DNA alterations, making GS-based systems a smarter pick for long-term stability.
To further minimise risks, site-specific mutagenesis and pre-transfection NGS screening are crucial. Since Sanger sequencing has a higher detection limit, NGS can catch plasmid mutations below 0.5%, improving clone screening success to over 90%[5].
Shuai Wang and colleagues from WuXi Biologics stress the importance of this vigilance:
Since process optimisation cannot rectify gene-level mutations, monitor mutation levels in stable clones rigorously[5].
For producers in need of specialised tools - whether for cryopreservation, genetic engineering, or cell line characterisation - Cellbase offers a marketplace connecting professionals with trusted suppliers. This ensures access to the right equipment and expertise for maintaining genetic stability in cultivated meat production.
Integrating Quality Control into Production
Incorporating quality control into every production phase is crucial. Without a structured system, even well-maintained cell lines can experience changes during expansion and scale-up. Quality control shouldn't be an afterthought - it must be a central part of production. This begins at the scale-up stage, where strict management systems and controlled environments play a key role.
Quality Control During Expansion and Scale-Up
As discussed earlier, genomic and contamination tests are vital, especially during scale-up. Moving from small volumes to thousands of litres introduces new risks, with each cell passage increasing the chance of mutations. A Quality Management System (QMS) helps manage these risks effectively. For example, between 2017 and 2022, Josep M. Canals and his team at the University of Barcelona implemented the ISO 9001:2015 QMS to standardise human pluripotent stem cell cultures. Their retrospective analysis of G-banding and aCGH data revealed a significant reduction in chromosomal aberrations compared to pre-adaptation conditions[10][13]. Canals highlighted the importance of ongoing monitoring:
The genetic instability showed by hPSCs in culture makes the frequent reassessment of genomic integrity an essential requirement when planning to use them for experiments[10].
Routine genomic screening is a must. Techniques like G-banding karyotyping and aCGH detect structural changes, while Next-Generation Sequencing (NGS) identifies mutations at levels below 0.5%[5]. Growth curve analysis can also flag issues early, such as contamination or genetic drift[9]. Environmental monitoring adds another layer of security, with practices like settle plate testing and six-monthly HEPA filter checks ensuring the production environment remains stable and stress-free for cell lines[4].
Consistency in media and reagents is equally important. Using serum-free, defined media like mTeSR1, alongside reagents with a Certificate of Analysis, helps reduce batch-to-batch variation and limits the risk of viral contamination[10][4]. Regular morphology checks - simple microscopic observations at different culture densities - can catch early signs of differentiation or stress[9]. For sourcing specialised equipment or reagents, platforms like Cellbase connect cultivated meat companies with trusted suppliers of growth media, bioreactors, and analytical tools designed for the industry's needs.
Functional Assays for Product Consistency
While genomic monitoring safeguards the process, functional assays ensure that cells perform as intended. Genetic stability alone isn't enough; cells must also maintain their ability to function properly across production batches. In cultivated meat, this means confirming that stem cells, such as muscle satellite cells, can still differentiate into mature muscle or fat tissue after expansion[2]. Differentiation assays are essential to verify this.
Metabolic assays like MTT, LDH, and Resazurin provide insight into cell health and viability[4][9]. These, combined with Short Tandem Repeat (STR) profiling, help confirm that cell lines remain authentic and free from cross-contamination throughout the production process[1][9].
Transcriptional analysis is another critical step. Xiaoyue Chen and Sam Zhang recommend:
cDNA rather than genomic-DNA sequencing is recommended for mutation detection to evaluate risks at the transcription level[5].
This method offers a more accurate picture of the final product, as it reflects how genes are expressed rather than just their genomic locations. By pairing genomic screening with functional assays, producers can ensure that every batch meets stringent standards for safety, quality, and performance at every stage of production.
Conclusion
Maintaining genetic stability is critical for producing cultivated meat safely and consistently. Francisco J. Molina-Ruiz and his colleagues from the Laboratory of Stem Cells and Regenerative Medicine highlight the risks:
Genetic changes in hPSCs could jeopardize not only the safety of hPSC-based cell products... but also lead to heterogeneous differentiation propensity of the starting material, altered gene expression profiles and inefficiency of the final cell product [10].
The stakes are significant - over 531 misidentified cell lines have been recorded by the International Cell Line Authentication Committee [1].
Tackling these issues requires a robust quality control framework. This involves combining methods like STR profiling, G-banding karyotyping, aCGH, and advanced NGS [5], alongside systems such as ISO 9001:2015 to standardise processes and minimise chromosomal abnormalities [13].
Economic factors also drive the need for these measures. Genetic drift can result in mutated cells gaining a growth advantage, potentially spoiling entire production batches [10][11]. With the growing focus on human pluripotent stem cells, the demand for stable cell lines has never been greater. As Professor David L. Kaplan from Tufts University explains:
Immortalized cell lines are generally considered a requirement for the generation of huge amounts of edible tissue from a stable, robust bioprocess [14].
For cultivated meat producers, quality control must be embedded in every step - from plasmid screening to large-scale production monitoring. By combining thorough testing with preventative strategies, producers can ensure consistent and reliable outcomes. Cellbase serves as a vital link, connecting producers with trusted suppliers of analytical tools, growth media, and bioreactors. Ultimately, achieving regulatory approval and commercial viability hinges on maintaining genetic stability in every batch.
FAQs
How often should genetic stability testing be performed during scale-up?
Genetic stability testing is a crucial step during scale-up and should be carried out on a regular basis. How often this testing occurs largely depends on the specific cell line and the process in question. To reduce the chances of mutations and maintain the cells' stemness, it's wise to establish a passage limit informed by genetic analysis.
Which tests best detect small mutations and large chromosome changes?
Tests such as SNP array analysis and genome-wide SNP genotyping are excellent for detecting small mutations, copy number changes, and aneuploidy. These methods are particularly useful for identifying large chromosomal alterations in cell lines.
What’s the simplest way to prevent genetic drift across production batches?
To minimise genetic drift, it's crucial to carry out regular genetic and functional analyses of cell lines and restrict the number of passages they undergo. Implement practices like establishing master cell banks and routinely checking genetic stability, as recommended in quality control protocols. These measures are key to maintaining consistency and ensuring dependable results across different production batches.











