Transitioning cells to serum-free media (SFM) is crucial for ethical and scalable cultivated meat production. This process eliminates animal-derived serum, reducing contamination risks and ensuring a consistent environment for cell growth. However, it comes with challenges like nutrient depletion, metabolic shifts, and maintaining cell viability. Monitoring tools such as flow cytometry, metabolomics, and transcriptomics play a key role in navigating these challenges by tracking cell health, nutrient usage, and gene expression changes.
Key Takeaways:
- Why it matters: SFM ensures consistency, reduces contamination risks, and aligns with ethical standards.
- Challenges: Cells in SFM are sensitive to stressors like pH changes, nutrient depletion, and mechanical forces.
-
Monitoring tools:
- Flow cytometry: Tracks cell viability and apoptotic markers.
- Metabolomics: Analyses nutrient consumption and metabolic shifts.
- Transcriptomics: Examines gene expression changes in key pathways.
- Applications: Data from these tools helps refine media formulations, reducing costs and improving cell growth.
Quick Insight:
Tools like HPLC, mass spectrometry, and real-time sensors provide actionable data to optimise serum-free transitions. Platforms like Cellbase simplify access to these tools and media, supporting cultivated meat research.
Serum-Free Cultures: Why and How? (February 2022)
sbb-itb-ffee270
Analytical Tools for Serum-Free Adaptation
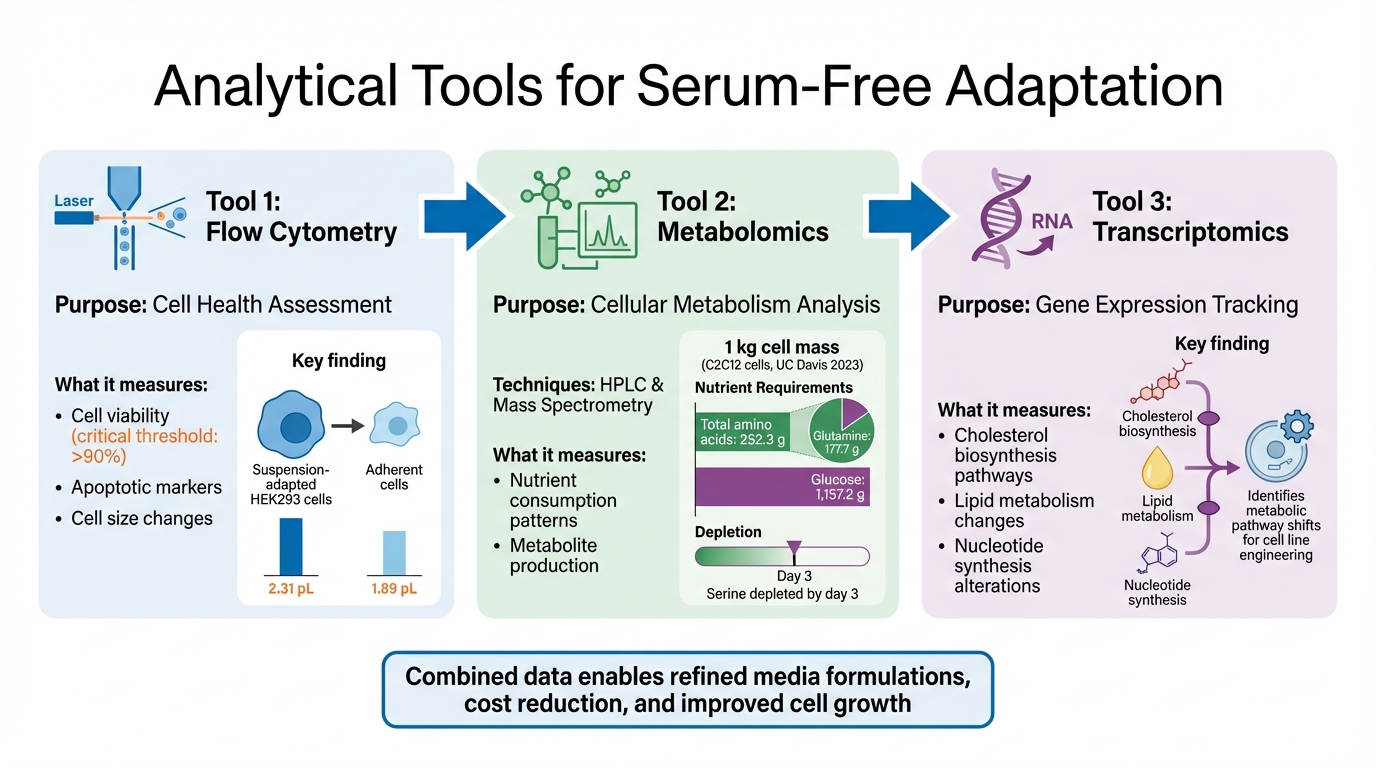
Three Key Analytical Tools for Monitoring Serum-Free Cell Adaptation
Flow Cytometry for Cell Health Assessment
Flow cytometry is a powerful tool for quickly evaluating cell viability and overall health during serum-free adaptation. Maintaining high cell viability is critical for the adaptation process to succeed[1]. This method also detects apoptotic markers and tracks changes in cell size. For instance, research shows that suspension-adapted HEK293 cells tend to have larger volumes, averaging around 2.31 pL compared to 1.89 pL in their adherent counterparts[1]. Such insights provide a clearer picture of cellular behaviour during adaptation. Beyond this, metabolomics offers a deeper dive into the metabolic shifts underlying these changes.
Metabolomics for Cellular Metabolism Analysis
Metabolomics focuses on analysing nutrient usage and metabolite production, offering a detailed view of cellular metabolism during serum-free adaptation. Techniques like HPLC and mass spectrometry are invaluable for identifying nutrient consumption patterns and metabolite outputs. These findings are essential for fine-tuning media formulations to enhance efficiency and reduce costs[2].
A noteworthy study conducted in June 2023 by researchers at the University of California, Davis, explored C2C12 muscle cells grown in Essential 8 serum-free media. They found that while growth rates matched those in serum-based media over seven days, serine levels were nearly depleted by day 3, signalling the end of exponential growth. The study calculated that producing 1 kg of wet cell mass required about 252.3 g of total amino acids (including 177.7 g of glutamine) and 1,157.2 g of glucose[2]. Such data is pivotal for refining media formulations, especially for cultivated meat production.
Additionally, intracellular metabolic profiling has revealed significant shifts in central carbon metabolism. In September 2022, researchers at the Norwegian University of Science and Technology discovered that suspension cells exhibited notably higher intracellular itaconate levels compared to adherent cells. This highlights how culture modes can profoundly impact cellular metabolism[1].
Transcriptomics for Gene Expression Tracking
To complement metabolic studies, transcriptomics sheds light on the gene regulation changes that occur during serum-free adaptation. This technique is particularly effective for identifying alterations in pathways like cholesterol biosynthesis, lipid metabolism, and nucleotide synthesis – processes frequently affected during the transition to serum-free conditions[1]. By mapping these gene expression changes, researchers can better understand how cells behave in serum-free environments and develop strategies to engineer cell lines for improved adaptability.
Mi Jang from NTNU emphasised the importance of these findings:
"The largest differences in metabolic profiles were observed between culture modes (adherent vs. suspension), followed by culture medium condition (control growth medium vs. serum-free medium)" [1].
Integrating transcriptomics with metabolic and viability data offers a more complete picture of how cells adapt, paving the way for more efficient serum-free culture systems.
Using Data to Improve Serum-Free Adaptation
Finding Problems in Cell Line Adaptation
Data plays a crucial role in identifying and addressing issues during cell line adaptation. Viability monitoring acts as an early warning system. When cell viability drops below an acceptable level, researchers revert to the previous adaptation step to avoid losing the entire culture. This approach allows adjustments to be made before moving forward[1][5].
Spent media analysis is another powerful tool for detecting nutrient limitations that hinder adaptation. For instance, mass spectrometry often reveals that cells in serum-free media deplete nutrients like glutamine and glycine much faster than anticipated, which can signal the end of exponential growth[2]. Additionally, tracking lactate accumulation is essential, as elevated lactate levels can suppress growth and myogenic activity, especially in high-density cultures[2].
Growth kinetics offer measurable indicators of performance. A noticeable increase in doubling time or a decrease in the specific growth rate suggests the media formulation may not be supporting the cell line effectively[1][4]. Deviations from established growth benchmarks highlight areas that require immediate attention.
Morphological changes also provide valuable clues. For example, severe clumping or rounding combined with slower doubling times indicates adaptation failure[5]. However, if doubling times remain stable despite morphological shifts, it suggests that adaptation is progressing successfully. These observations enable researchers to refine serum-free media based on data-driven insights.
Developing Custom Serum-Free Media
Using these diagnostic metrics, researchers can fine-tune serum-free formulations tailored to specific cell lines. Gene expression profiling sheds light on metabolic pathways that struggle during adaptation, such as cholesterol biosynthesis, nucleotide synthesis, and lipid metabolism[1]. This information guides the selection of serum-free media supplements to enhance formulations.
One example comes from Menarini Biotech srl, where a team led by Leonardo Sibilio successfully adapted a CHO-DG44 cell line in September 2019. They screened 20 chemically defined formulations using a Media Assessment Panel and determined that Medium #27 was the best fit. This medium supported consistent growth and viability above 90% over more than 10 passages[7].
Similarly, transcriptomic and spent media data can lead to actionable insights. In July 2025, Merck Healthcare KGaA researchers used comparative proteomics to study HepG2 cells transitioning to serum-free conditions. They found significant overexpression of antioxidative enzymes like glutathione peroxidase. By linking these findings to enzyme activity assays, they discovered that "selenium supranutrition" was necessary to protect cells from oxidative stress. Adjusting selenium levels in their custom media directly addressed this issue[6].
Metabolic profiling also helps optimise costs while maintaining performance. For example, research on C2C12 cells revealed that producing 1 kg of wet cell mass requires about 250–275 g of amino acids and 1,100–1,500 g of glucose[2]. These precise metrics allow researchers to remove unnecessary nutrients, reducing costs without compromising cell health. Spent media analysis further identifies unused expensive components, enabling cost-effective reformulation of media.
How Cellbase Supports Serum-Free Adaptation
Sourcing Analytical Tools Through Cellbase
Cellbase connects cultivated meat researchers with trusted suppliers offering specialised tools for monitoring serum-free adaptation processes. Through the platform, researchers can find metabolite analysis systems like High-Performance Liquid Chromatography (HPLC) for tracking glucose and lactate levels. It also features advanced instruments such as the REBEL analyser from 908 Devices, which employs microfluidic capillary electrophoresis to provide real-time measurements of amino acids and vitamins[2].
For continuous monitoring, technologies such as Raman and NIR spectroscopic sensors, soft-sensors, and PAT tools are available, enabling real-time adjustments during cultivation[8]. Cell health assessment is supported by high-content screening systems like the ImageXpress Pico microscope, which automates processes like cell counting and nuclear stain image segmentation[2]. These tools align with Quality by Design (QbD) principles and adhere to current Good Manufacturing Practices (cGMP), shifting the focus from post-batch testing to real-time quality control[8].
In addition to analytical tools, Cellbase offers access to essential serum-free media formulations, including Essential 8 (E8), Hybridoma-SFM, CHO-S-SFM II, and Pro293a. It also provides key media components like recombinant human FGF2, insulin, transferrin, and TGF-β[2][4]. Researchers can source bioprocess hardware such as ultrafiltration modules, membranes, and centrifuges for cell retention systems[8]. This integrated sourcing approach simplifies the process of acquiring the tools needed for serum-free adaptation.
Accessing Technology for Serum-Free Adaptation
Beyond equipment, Cellbase makes it easier to find specialised technology tailored for serum-free adaptation. By addressing the challenges of fragmented supplier networks, the platform offers curated listings that are tagged with specific attributes like scaffold compatibility, serum-free suitability, or GMP compliance. This targeted approach helps researchers quickly locate products that meet their unique adaptation requirements.
This focus is especially important given the financial pressures facing the cultivated meat industry. As David E. Block from the University of California, Davis, points out:
"CM media will need to be substantially cheaper than all existing commercial animal cell culture media to permit price parity of CM products with conventional meat"[2].
Conclusion
Accurate monitoring plays a key role in achieving successful serum-free adaptation. Tools like HPLC, mass spectrometry, and flow cytometry are indispensable for tracking metabolic changes, pinpointing bottlenecks, and maintaining cell viability above the crucial 90% threshold[3][9]. Without these technologies, it becomes difficult to determine whether cells are genuinely adapting or simply surviving under stress. These findings highlight the need to address the logistical and technical barriers associated with acquiring the necessary equipment.
Obtaining specialised equipment remains a significant hurdle. Serum-free adaptation is a complex process, often taking over a month to complete[9]. This complexity requires access to advanced tools, which are not always easy to source. Platforms that simplify the procurement of such equipment are therefore vital. As Sebastian Juan Reyes from Polytechnique Montreal points out:
"A historically conservative industry is encouraged to innovate and improve its production platforms by adopting new technologies as they emerge to enhance process monitoring without additional regulatory burden"[8].
The introduction of real-time quality control systems has made it possible to adjust processes immediately, using continuous data to guide decisions[8].
Building on these analytical methods, Cellbase offers a solution by streamlining the procurement of equipment and media for cultivated meat researchers. By harnessing insights from flow cytometry, metabolomics, and transcriptomics, Cellbase connects researchers to verified analytical tools and chemically defined media, ensuring they have the resources needed to carry out serum-free adaptation more efficiently.
FAQs
Which metrics best prove cells are truly adapted to serum-free media?
Key indicators of a successful shift to serum-free media include growth kinetics, intracellular metabolite profiles, nutrient usage rates (such as glucose, glutamine, glycine, and cystine), and cell viability. These metrics provide a clear picture of how well the cells are adapting while preserving their normal functions.
How can I detect nutrient bottlenecks early during serum-free adaptation?
To spot nutrient limitations early on, it's essential to keep an eye on metabolic activity and nutrient usage. Techniques like spent media analysis can pinpoint variations in the consumption of crucial nutrients such as glucose, glutamine, glycine, and cystine. Additionally, metabolic profiling can uncover changes in glycolytic and oxidative pathways. By routinely tracking metabolite levels and nutrient uptake rates, you can make timely adjustments to your media or feeding methods, boosting the chances of a smooth serum-free adaptation.
When should I use flow cytometry, metabolomics, or transcriptomics?
Flow cytometry, metabolomics, and transcriptomics each offer unique insights when studying how cell lines adjust to serum-free media. Flow cytometry is ideal for evaluating cell traits such as viability and surface markers. Metabolomics dives into the analysis of cellular metabolites, shedding light on nutrient consumption and metabolic changes. Meanwhile, transcriptomics focuses on gene expression, helping to identify molecular responses and regulatory pathways. The choice between these methods depends on whether you're targeting cell phenotype, metabolism, or gene regulation.











