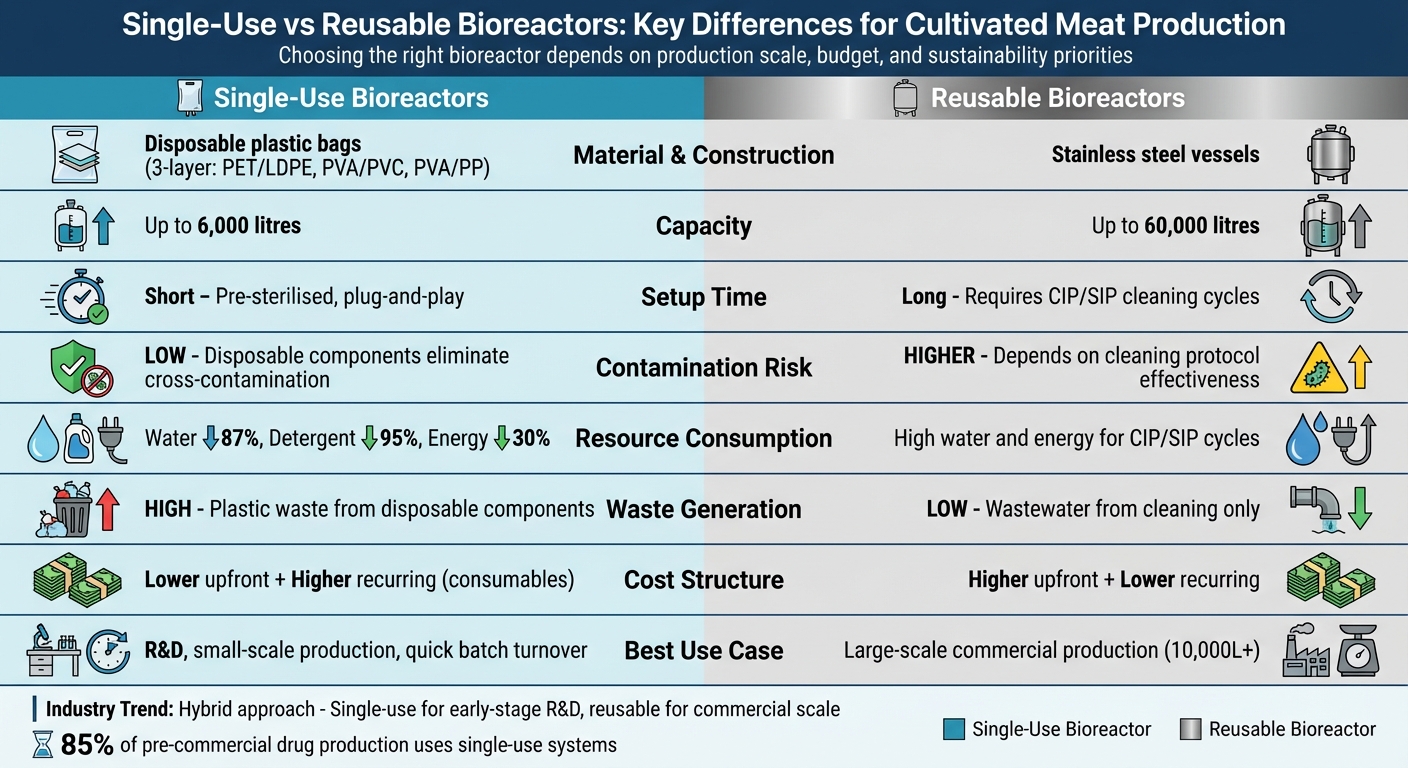
Single-use and reusable bioreactors are critical to cultivated meat production, but they serve different purposes based on scale, cost, and resource needs. Here’s the core takeaway:
- Single-use bioreactors: Use disposable plastic bags, require less setup time, and minimise contamination risks. They’re ideal for research and small-scale production but generate plastic waste and incur ongoing consumable costs.
- Reusable bioreactors: Built with stainless steel, they handle larger volumes and are cost-efficient for high-scale production. However, they require extensive cleaning, more infrastructure, and higher upfront investment.
Quick Comparison
| Feature | Single-Use Bioreactors | Reusable Bioreactors |
|---|---|---|
| Material | Disposable plastic bags | Stainless steel vessels |
| Capacity | Up to 6,000 litres | Up to 60,000 litres |
| Setup Time | Short (pre-sterilised) | Long (cleaning required) |
| Contamination Risk | Low (disposable components) | Higher (depends on cleaning) |
| Resource Use | Lower water and energy consumption | Higher water and energy use |
| Waste | High (plastic waste) | Low (wastewater from cleaning) |
| Cost | Lower upfront, higher recurring | Higher upfront, lower recurring |
Your choice depends on production scale, budget, and waste/resource priorities. Facilities often combine both systems - single-use for early-stage work and reusable for large-scale production.
Single-Use vs Reusable Bioreactors Comparison for Cultivated Meat Production
Single Use Bioreactor: Overview, Types, Advantages, Limitations and Future of Single Use Bioreactors
Single-Use Bioreactors: Design and Benefits
Single-use bioreactors are centred around disposable plastic bags, making them a perfect match for the fast-paced testing and production cycles required in cultivated meat research and development. Their design aligns seamlessly with the needs of experimental and early-stage production in cultivated meat facilities.
Materials and Construction
At the heart of these systems is a specially designed three-layer plastic bag. Each layer serves a specific purpose:
- Outer layer: Made of PET/LDPE, it provides structural strength.
- Middle layer: Composed of PVA/PVC, it acts as a barrier to gases.
- Inner layer: Crafted from PVA/PP, it ensures safe contact with the cell culture media [3].
These bags are pre-sterilised - typically through gamma irradiation - allowing for quick batch turnover. This approach shifts the responsibility for sterility validation from the production facility to the supplier [3]. Many systems also include integrated sensors for monitoring pH, oxygen, and temperature. Non-invasive technologies, like pH-sensitive dye patches read by external lasers, enable real-time tracking without compromising the sterile environment [3].
Agitation, crucial for maintaining optimal conditions, is achieved through mechanical or magnetic stirrers or a rocking motion. These methods are designed to minimise shear forces, protecting delicate animal cells. While early single-use bioreactors were limited to smaller volumes, newer models now support capacities of up to 2,000 litres [3].
These thoughtful design features contribute to fast changeovers and effective contamination control during operations.
Operational Advantages
The design of single-use bioreactors translates directly into operational benefits. By eliminating the need for Clean-in-Place (CIP) and Steam-in-Place (SIP) procedures, these systems dramatically reduce setup times and enable quicker transitions between cell lines or experiments [3]. Since the product-contacting surfaces are disposable, the risk of cross-contamination is nearly eliminated. Notably, over 85% of pre-commercial drug production relies on single-use systems, highlighting their effectiveness during R&D phases, including in cultivated meat production [3].
These systems also deliver substantial resource savings. Compared to traditional stainless steel units, they reduce water use by 87%, detergent consumption by 95%, and energy requirements by 30% [3]. While the generation of plastic waste is a drawback, the reduced demand for water, energy, and cleaning chemicals offers an alternative perspective on sustainability. Facilities must weigh these benefits against their specific production goals to determine the best path forward.
Reusable Bioreactors: Design and Benefits
Reusable bioreactors are built for durability and long-term use, featuring stainless steel vessels designed to perform consistently for decades. These systems are made from materials capable of withstanding repeated cleaning and sterilisation, making them well-suited for high-volume production where reliability and uniformity are key [2].
Compared to single-use systems, reusable bioreactors provide dependable solutions for large-scale cultivated meat production.
Materials and Construction
The foundation of reusable bioreactors lies in stainless steel vessels, which can endure rigorous cleaning and sterilisation cycles. This is crucial for ensuring food safety in cultivated meat production [2]. These systems integrate CIP (clean-in-place) and SIP (sterilise-in-place) protocols, which are essential for maintaining sterility during high-volume operations. However, they require dedicated infrastructure, such as high-capacity steam systems for sterilisation and specialised drainage setups for handling CIP chemicals [1][2].
The most common type of reusable bioreactor used in cultivated meat production is the mechanically agitated stirred tank reactor. This design ensures even distribution of nutrients and oxygen throughout the culture using precise mechanical agitation [1].
Operational Advantages
Reusable bioreactors are designed to work seamlessly with CIP/SIP infrastructure, making them ideal for large-scale, continuous production. While they require more labour, time, and effort for cleaning and sterilisation compared to single-use systems, they offer greater efficiency for long-term, high-volume batch operations [2]. Their advanced process control capabilities are particularly advantageous for cultivated meat production [1][2].
As the cultivated meat industry scales up production by 2026, many facilities are adopting hybrid approaches. Single-use systems are often used for early-stage research and process development, while reusable bioreactors are reserved for commercial-scale operations [2]. Although the initial investment in reusable systems is higher, they are more cost-effective over time due to the absence of recurring consumable expenses [2]. For large-scale facilities, especially those operating at capacities of 10,000 litres or more, stainless steel bioreactors remain the go-to choice for achieving economies of scale [1][2].
Comparing Design, Operations, and Scale
When it comes to cultivated meat production, the type of bioreactor chosen - single-use or reusable - depends heavily on factors like material composition, operational processes, and production scale. Single-use systems typically involve medical-grade plastic bags and disposable parts, while reusable bioreactors are made from durable stainless steel, designed to withstand repeated sterilisation cycles. These differences don’t just affect how quickly batches can be initiated but also have a big impact on water and energy usage in a facility. The design itself plays a key role in shaping material compatibility and performance.
Material compatibility is one of the biggest distinctions between the two systems. Single-use bioreactors, made from plastic, generally work well with most growth media and cultivated meat cell lines. However, there are ongoing concerns about potential leachables and extractables. On the other hand, stainless steel bioreactors excel in handling high pressures and temperatures without degrading, making them ideal for intensive processes like perfusion. By December 2024, companies such as Aleph Farms and Mosa Meat had secured funding to develop pilot-scale bioreactors with a 10,000-litre capacity, incorporating AI and perfusion technology. This reflects a broader industry shift towards larger-scale, more efficient production methods [4].
Setup and turnaround times also show clear differences. Single-use systems are designed for convenience, offering a plug-and-play setup that can turn around batches in just a few hours. Reusable bioreactors, however, require thorough cleaning and sterilisation between batches, which increases labour requirements and slows turnaround times. Despite this, for facilities handling high-volume production at scales of 10,000 litres or more, reusable systems often deliver better long-term efficiency, even with slower cycle times [4]. These differences in operation also tie into resource usage and environmental considerations, which are explored further below.
Energy and water consumption is another area where the systems diverge. Single-use bioreactors eliminate the need for onsite cleaning and sterilisation systems, significantly reducing water and energy use during operation. In contrast, reusable systems require dedicated steam supplies and high-purity water for cleaning, which increases their resource demands. The trade-off here involves weighing the environmental impact of plastic waste from disposable components against the resources consumed by repeated cleaning cycles.
Both approaches bring efficiencies that can lead to lower production costs. The operational and economic impacts of these systems will be examined more closely in the context of cost and environmental evaluations.
sbb-itb-ffee270
Cost and Environmental Factors
When deciding on a bioreactor for cultivated meat production, cost considerations play a major role alongside operational differences.
Financial Comparison
The financial dynamics between single-use and reusable bioreactors largely depend on the scale of production. Single-use systems are appealing for their lower upfront costs, as they eliminate the need for permanent infrastructure like steam-in-place (SIP) and clean-in-place (CIP) systems [1]. However, the recurring costs of disposable components can quickly add up, especially as production scales [1].
Reusable stainless steel bioreactors, on the other hand, require a much larger initial investment due to the need for fixed piping, sterilisation systems, and other infrastructure [1]. Yet, for large-scale operations, their lower ongoing costs can make them more economical in the long run. The decision often comes down to whether the focus is on a faster route to market with minimal setup or a long-term commitment to a scalable and efficient production system. Additionally, media costs remain a significant hurdle in cultivated meat production, making high-density cultivation methods a key strategy for economic viability [1].
While costs are a major factor, the environmental impact of these systems is another critical aspect to consider.
Environmental Footprint
Single-use bioreactors come with the challenge of managing large amounts of plastic waste from disposable components, which becomes a growing issue as production scales [1]. This raises concerns about sustainability, especially in the context of industrial-scale operations. On the flip side, reusable bioreactors require rigorous cleaning protocols, which consume substantial amounts of water and energy during each CIP and SIP cycle. These processes rely on steam and high-purity water, adding to resource demands [1].
These environmental factors influence how facilities allocate resources and manage waste.
| Environmental Factor | Single-Use Bioreactors | Reusable Bioreactors |
|---|---|---|
| Waste Generation | High (due to disposable plastics) [1] | Low (mainly wastewater from cleaning) |
| Water Consumption | Low (no rinsing required) [1] | High (needed for CIP/SIP cycles) [1] |
| Energy Consumption | Lower (no steam sterilisation) [1] | Higher (steam and hot water usage) [1] |
| Turnaround Time | Faster (no cleaning downtime) [1] | Slower (time needed for sterilisation) [1] |
The decision between single-use and reusable systems often hinges on which environmental trade-offs a facility is better equipped to handle - whether it's managing the disposal of plastic waste or addressing the resource-intensive cleaning requirements. As the cultivated meat sector evolves, finding ways to reduce environmental impacts during scale-up remains a priority.
Regulatory Requirements and Risk Management
Controlling contamination and adhering to regulations are key factors when choosing bioreactor systems for cultivated meat production. How a system ensures sterility plays a major role in determining the regulatory pathway and the type of documentation needed for approval.
Contamination Control and Sterility
Single-use bioreactors come pre-sterilised through gamma irradiation, removing the need for onsite sterilisation. Since all product-contact parts, like bags and fluid paths, are disposed of after each run, the risk of cross-contamination between batches is minimised. However, this shifts the responsibility for sterility assurance to verifying the standards of the supplier.
On the other hand, reusable bioreactors require rigorous onsite cleaning and sterilisation protocols. Each production run must follow strict Clean-in-Place (CIP) and Steam-in-Place (SIP) procedures to eliminate any residues or microbes. While this method aligns with established regulatory pathways, it demands substantial labour and meticulous documentation for each cleaning cycle. If cleaning processes are poorly executed or inconsistent, the risk of contamination increases.
| Feature | Single-Use Bioreactors | Reusable Bioreactors |
|---|---|---|
| Sterility Source | Pre-sterilised by supplier (gamma irradiation) | Onsite sterilisation (Steam-in-Place/SIP) |
| Contamination Risk | Low; components replaced after each run | Higher; depends on cleaning effectiveness |
| Validation Focus | Supplier standards and extractables | CIP and SIP protocol validation |
| Setup Time | Short; no cleaning needed | Long; cleaning and validation required |
These differences in sterility assurance directly impact how each system aligns with regulatory requirements.
Meeting Regulatory Standards
Regulatory frameworks increasingly emphasise the need for precise tracking and reproducibility. By 2026, compliance expectations for bioreactor systems have tightened, requiring facilities to adopt systems that support detailed process monitoring and consistent results. Single-use systems simplify onsite validation but come with the added need to manage extractables and leachables, ensuring suppliers meet stringent manufacturing criteria.
Reusable systems, while familiar to regulatory bodies due to their traditional compliance pathways, demand extensive documentation and readiness for audits, particularly for each sterilisation cycle. This makes them more labour-intensive but also highly reliable for large-scale production. Effective contamination control not only ensures product quality but also supports scalable, regulation-compliant production of cultivated meat.
Many facilities are now opting for a hybrid approach. Single-use systems are commonly used in research and development phases for their quick setup and reduced contamination risks. For large-scale production, facilities often transition to reusable stainless steel bioreactors, which align with well-established regulatory processes.
These rigorous compliance requirements highlight the importance of sourcing high-quality bioreactors, such as those offered by Cellbase.
Bioreactor Procurement for Cultivated Meat Production
When it comes to bioreactor procurement, strategies need to align with the specific requirements of cultivated meat production. Choosing the right bioreactor system involves balancing factors like production scale, cost considerations, and regulatory demands. Whether a facility focuses on research, scaling up processes, or large-scale commercial production exceeding 500 litres, these decisions must account for the unique challenges of working with delicate mammalian cells and the need for integrated CIP (clean-in-place) and SIP (sterilise-in-place) systems [5].
Procurement teams also face the critical task of ensuring that systems are designed to support regulatory compliance. This means prioritising robust data management and traceability, as well as verifying that materials meet food-grade standards with minimal risks of leachables and extractables - especially important for single-use components. The complexity of sourcing equipment tailored to cultivated meat production underscores the importance of working with reliable partners to simplify the process.
Sourcing Bioreactors Through Cellbase
Specialised platforms have emerged to tackle these procurement challenges, and Cellbase is one such solution. It operates as a dedicated B2B marketplace for the cultivated meat industry, offering a streamlined way to access production-grade bioreactors and related equipment. By providing verified listings of systems specifically designed for commercial-scale production, Cellbase simplifies procurement. With transparent pricing and global shipping options, including cold chain logistics, it ensures accessibility for buyers worldwide.
What sets Cellbase apart is the ability to consult with Cell Ag Experts. These experts help buyers address technical needs, such as ensuring the biocompatibility of scaffolds and hydrogels within specific bioreactor systems or identifying equipment with automated harvesting features. This guidance becomes particularly valuable in managing contamination risks, as the average batch failure rate stands at 11.2%, rising to 19.5% in larger operations [6].
Beyond bioreactors, Cellbase offers an ecosystem of solutions to meet other production needs. This includes sourcing cell lines, serum-free media supplements, scaffolds, and downstream processing equipment [5]. By providing access to compatible components from suppliers who understand the intricacies of cultivated meat production, Cellbase helps reduce technical risks and speeds up the procurement process.
Conclusion
Selecting between single-use and reusable bioreactors hinges on how well each option aligns with your production needs. Single-use systems offer the advantage of being pre-sterilised and allow for quicker turnaround times, making them particularly suitable for early-stage research and development where adaptability is key. On the other hand, reusable stainless steel bioreactors, while requiring intensive Clean-In-Place (CIP) and Steam-In-Place (SIP) protocols, can prove more cost-effective over time, especially for large-scale, stable production runs [1][2].
This choice has a direct impact on regulatory compliance and operational efficiency, both of which are critical for the consistent and controlled production of cultivated meat. For processes involving adherent cells, scaffold-compatible systems and meticulous contamination control are especially important.
Key considerations include labour, downtime, and consumable costs. While single-use systems often come with lower initial costs, recurring expenses for consumables can accumulate significantly over time. In contrast, reusable systems generally involve a higher upfront investment but provide better process control, particularly for large-batch production [2].
Specialised procurement platforms like Cellbase help simplify these decisions by offering verified listings and enabling transparent comparisons. Whether you're sourcing bioreactors compatible with scaffolds or exploring hybrid approaches that combine features of both system types, leveraging industry-specific expertise and reliable supplier networks ensures more informed choices. This streamlined procurement process supports the broader aim of optimising cultivated meat production.
Ultimately, the ideal bioreactor choice strikes a balance between immediate operational demands and long-term objectives. It must account for regulatory requirements, cost considerations, and controlled production conditions - factors that are essential for achieving efficient and scalable cultivated meat production. This balance reflects the operational insights explored throughout this analysis.
FAQs
When should I switch from single-use to reusable bioreactors?
When your cultivated meat production reaches a larger scale, it’s worth considering a switch to reusable bioreactors for better cost efficiency in the long run. While single-use bioreactors are ideal for smaller operations due to their lower initial costs, reusable stainless steel systems are a smarter choice for large-scale manufacturing.
Though these systems come with higher upfront costs, they’re designed to handle much larger volumes (20,000L or more) and are built to last. This durability helps offset the ongoing expenses associated with consumable materials in single-use systems. The transition makes sense when your production volume and efficiency needs grow to a point where the investment pays off.
How do I manage extractables and leachables in single-use bags?
Managing extractables and leachables in single-use bags for cultivated meat production requires careful attention to detail. Start by choosing high-quality, certified bags that are specifically tested to ensure low levels of these substances. For sensitive applications, it’s essential to carry out thorough leachable testing before using the bags.
Stick to the manufacturer's recommendations, store the bags under proper conditions, and implement cleaning protocols such as rinsing with appropriate solvents or water. Additionally, consult with suppliers and perform detailed risk assessments tailored to the materials and applications you’re working with. This approach helps maintain both safety and quality throughout the process.
What does CIP/SIP validation involve for reusable bioreactors?
Reusable bioreactors rely on CIP (cleaning-in-place) and SIP (sterilisation-in-place) validation to guarantee thorough cleaning and sterilisation. These procedures involve water and chemical cleaning cycles, functional tests, and rigorous assessments to ensure all contaminants are effectively removed. By adhering to these steps, the process not only meets regulatory requirements but also ensures the bioreactors remain sterile and ready for use.











