Switching from fetal bovine serum (FBS) to serum-free media (SFM) is critical for scaling cultivated meat production. FBS reliance creates challenges like high costs, limited supply, and inconsistent quality. SFM offers a safer, more controlled alternative, but it comes with hurdles:
- Cell Attachment Issues: Myoblasts struggle to adhere without serum, often requiring expensive coatings like laminin or Matrigel. Conditioned media or specific supplements can improve adhesion.
- Slower Growth Rates: Serum-free systems lack key nutrients, leading to reduced proliferation and ammonia buildup. Adding growth factors and replacing glutamine with alternatives can help.
- Inconsistent Media Performance: Many commercial SFMs, optimised for human cells, fail to support livestock myoblast growth effectively. Testing across species and over longer periods with a media optimisation discovery kit is crucial.
Solutions include tailored formulations, partial medium replacement, and co-culture systems to mimic serum-like conditions. While SFM can approach the performance of FBS systems, scaling to 3D bioreactors introduces complexities like adhesion and waste management. Careful monitoring of cell quality ensures success in large-scale production.
Switching to SFM isn't just about better science - it's becoming a necessity as FBS prices continue to rise. Researchers and producers must focus on optimising media and sourcing reliable materials to make cultivated meat production viable and cost-effective.
Plant-based scaffolds that induce serum free cell adhesion for cultured meat - Indi Geurs - ISCCM9
sbb-itb-ffee270
Common Problems in Serum-Free Media for Myoblasts
Switching from serum-based to serum-free formulations can present several technical challenges that disrupt workflows and push up costs. These issues often show up in specific ways, starting with cell attachment.
Reduced Cell Attachment and Survival
One of the biggest hurdles is that myoblasts don’t attach well in serum-free media. Serum naturally provides a mix of proteins, growth factors, and lipids that help cells stick to surfaces. Without these components, myoblasts struggle to adhere, which often leads to early cell death.
To address this, many serum-free systems rely on pricey coating agents like laminin 511 or Matrigel. But even with these coatings, attachment levels often fall short of what’s seen in serum-based cultures. For example, a 2024 study found that standard serum-free media supported only 2,210 ± 319 cells/cm² on uncoated dishes. In contrast, a conditioned serum-free medium - supplemented with secreted factors from other cell lines - nearly tripled that figure to 5,985 ± 1,558 cells/cm² [2].
Another issue is increased sensitivity to antibiotics. In serum-free setups, antibiotics like Penicillin, Streptomycin, and Amphotericin B can cut proliferation by as much as 62%, compared to a 20–26% reduction in serum-based systems [1]. Without serum’s protective elements, cells are more vulnerable to stress, which further hampers their survival and growth.
Slower Cell Growth
Even if cells manage to attach, growth rates often lag behind. Serum provides essential nutrients like growth factors, cytokines, cholesterol, and fatty acids - many of which are missing or inadequate in most commercial serum-free formulations. This nutritional gap results in lower cell yields and longer production times.
Another complication is ammonia buildup from glutamine metabolism. Ammonia inhibits growth and, in serum-free conditions, where cells are already under metabolic strain, this toxicity can severely hinder expansion. Many commercial media were originally designed for human cells, so they may not meet the specific nutritional needs of bovine or porcine myoblasts [1][3].
Partial medium replacement, such as swapping out 75% of the medium during feeding, can help retain some endogenous growth factors. While this modestly improves growth rates, it doesn’t fully close the gap between serum-free and serum-based systems [1].
Variable Performance Across Commercial Products
Not all commercial serum-free media perform equally. In a study comparing seven formulations, only three - FBM™, Essential 8™, and TeSR™-E8™ - supported consistent bovine myoblast growth over six days. Others, like StemPro™ and mTeSR1™, supported growth for just four days before stalling, while STEMmacs™ failed entirely to sustain proliferation [1].
The problem lies in the fact that most commercial media are optimised for human stem cells or fibroblasts, not livestock myoblasts. What works well in biomedical research often falls short for cultivated meat production. This inconsistency highlights the need for formulations tailored specifically to livestock myoblasts. Manufacturer data for human cells can’t reliably predict how well a medium will perform with bovine or porcine cells.
To find the right serum-free medium, it’s crucial to conduct extended testing - ideally over six to ten days - to ensure it supports sustained cell expansion rather than just short-term growth.
Comparing Commercial Serum-Free Media Options
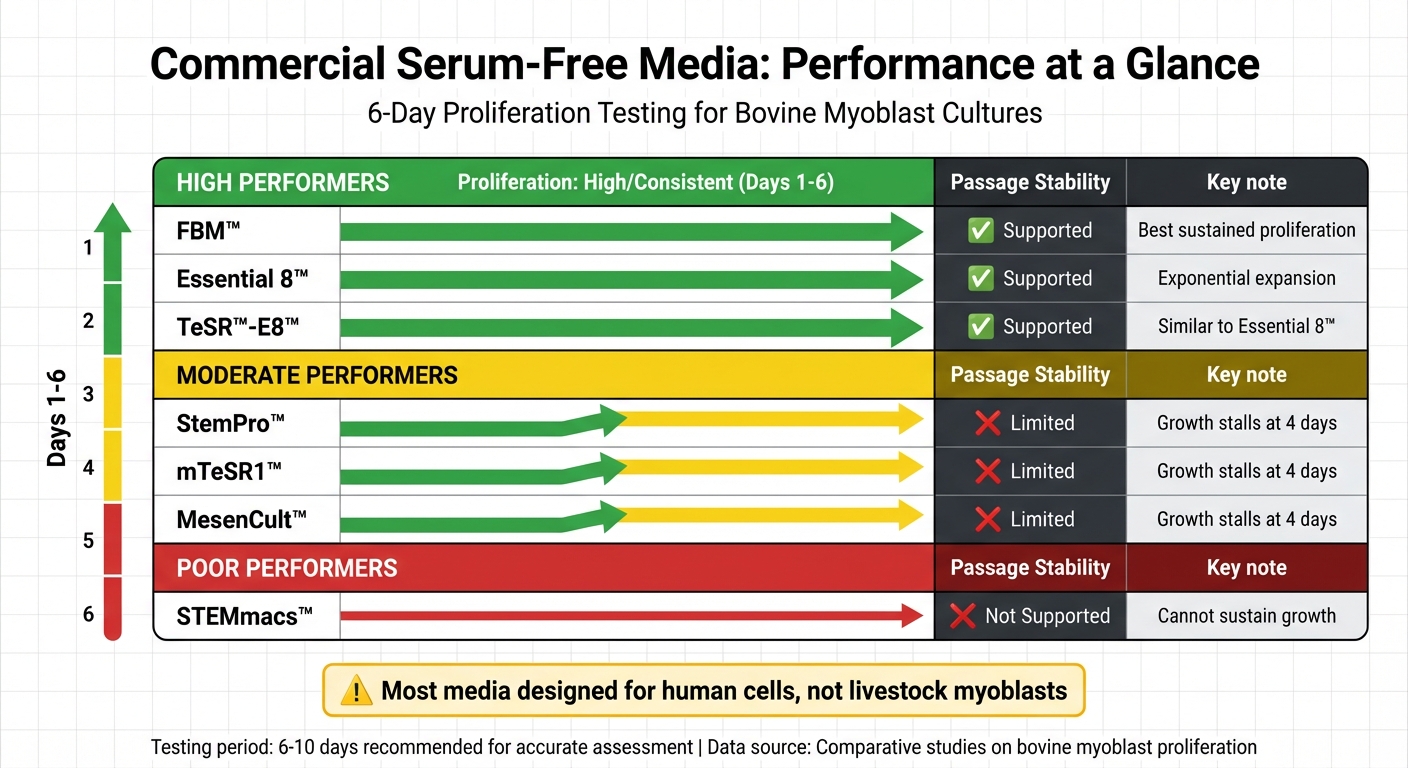
Performance Comparison of Commercial Serum-Free Media for Bovine Myoblast Cultures
Performance Data for Common Media
When it comes to serum-free media for myoblast cultures, performance can vary widely. Some products, like FBM™, Essential 8™, and TeSR™-E8™, consistently support bovine myoblast proliferation over six days. In contrast, others, such as StemPro™, mTeSR1™, and MesenCult™, tend to stall after just four days. Meanwhile, STEMmacs™ fails to sustain growth altogether [1].
Here’s a quick comparison of performance metrics for these media:
| Medium | Proliferation (Days 1–6) | Passage Stability | Key Observations |
|---|---|---|---|
| FBM™ | High/Consistent | Supported | Offers the best potential for sustained proliferation [1] |
| Essential 8™ | High/Consistent | Supported | Supports exponential expansion, though less than serum-based [1] |
| TeSR™-E8™ | High/Consistent | Supported | Similar to Essential 8™ for bovine myoblasts [1] |
| StemPro™ | Moderate | Limited | Growth stalls after four days [1] |
| mTeSR1™ | Moderate | Limited | Growth stalls after four days [1] |
| MesenCult™ | Moderate | Limited | Growth stalls after four days [1] |
| STEMmacs™ | Low/None | Not Supported | Unable to sustain bovine myoblast growth [1] |
Interestingly, most media - except FBM™ - show significantly lower cell counts within 24 hours of seeding when compared to serum-based controls. This highlights the importance of evaluating these metrics when choosing a medium, especially considering regulatory trends in growth media for food safety.
How to Choose the Right Serum-Free Medium
Selecting the best serum-free medium isn't just about growth rates; it requires a balance of several factors like proliferation, attachment, and cost-effectiveness. Testing media over a six-day period is crucial, as shorter assays might provide misleading results [1].
Species specificity is another vital consideration. Many serum-free options are designed with human cells in mind, which means they may not meet the nutritional demands of livestock myoblasts like bovine or porcine cells. The nutrient needs of different species and cell states can vary significantly, so testing is essential [3].
Coating requirements also play a big role. Some media need pricey coatings like laminin or Matrigel to ensure cell adhesion. If your process involves uncoated surfaces or food-grade materials, it’s worth testing whether the medium can support attachment without these additives. Conditioned media or formulations tailored for uncoated dishes can be a cost-effective alternative [2].
Another critical factor is the use of antibiotics. Standard antibiotic cocktails, such as Penicillin/Streptomycin, can reduce myoblast proliferation by 20–26% in serum-containing media and by as much as 62% in serum-free systems. Eliminating antibiotics can lead to significantly higher cell yields [1].
Lastly, don’t overlook metabolic waste management. Ammonia buildup can be toxic to cultures, so it’s a good idea to supplement media with non-ammoniagenic compounds like α-ketoglutarate or pyruvate. These additives help reduce ammonia toxicity and extend the longevity of cultures [3].
Methods to Improve Serum-Free Myoblast Cultures
Addressing the challenges of serum-free myoblast cultures requires targeted strategies. Here are some practical methods to enhance their performance.
Adding Key Supplements
Incorporating specific supplements can significantly improve myoblast growth. A blend of FGF-2 (10 ng/ml), EGF (5 ng/ml), IGF (5 ng/ml), and insulin (10 μg/ml) has been shown to enhance cell expansion in basal media like FBM [1]. These growth factors work together to promote cell proliferation while maintaining the undifferentiated state necessary for production.
Amino acids and vitamins are also crucial. Compounds such as pyridoxamine (Vitamin B6), asparagine, and glutamic acid play a key role in encouraging cell adhesion and proliferation, especially on uncoated surfaces [2]. These supplements help replace the metabolic support typically provided by serum, addressing adhesion-related challenges.
"Component analysis and validation experiments suggested that pyridoxamine, asparagine, and glutamic acid contributed to the acquisition of the culture function of the developed medium." - npj Science of Food [2]
However, caution is needed with lipid-based supplements like LipoGro. While they can stimulate growth, they may also induce adipogenic differentiation, causing myoblasts to develop fat vacuoles and lose their muscle cell identity [1].
Tailoring Media Formulations
Customising media formulations can optimise serum-free cultures using a growth factor discovery kit. One effective approach involves using conditioned media. Media conditioned by co-culturing HepG2 (human hepatoma) and NIH/3T3 (mouse fibroblast) cells replicates the metabolic profile of the foetal liver. This method achieves a cell density of 5,985 ± 1,558 cells/cm² on uncoated dishes, comparable to the 6,722 ± 1,500 cells/cm² achieved with serum-containing media [2]. The interaction between these cell types promotes the secretion of serum-like components, enhancing growth.
Another cost-effective strategy is partial medium replacement. By replacing only 75% of the medium instead of a full change, endogenous growth factors produced by the cells are preserved, improving growth rates without the need for additional supplements [1].
Preventing Early Differentiation with Inhibitors
Maintaining a proliferative state requires careful control of differentiation signals. For instance, conditioned media from HepG2 cells can suppress the expression of the myogenic differentiation marker Desmin, keeping cells undifferentiated and ready for expansion [2].
Additionally, tracking markers like CD29 (integrin beta-1) and Ki67 helps ensure the formulation is effective in maintaining cell proliferation, reducing the risk of premature differentiation. These markers provide a reliable way to monitor and adjust culture conditions for optimal results.
Scaling Serum-Free Myoblast Cultures for Production
Moving to 3D Culture Systems
Shifting serum-free myoblast cultures from flat 2D dishes to 3D bioreactor systems comes with its own set of challenges, especially when it comes to cell adhesion. Coating bioreactor components with expensive agents like laminin isn't practical for large-scale production. However, using conditioned media from HepG2 and NIH/3T3 co-cultures or enriching basal media with compounds like pyridoxamine, asparagine, and glutamic acid has proven effective. These methods allow myoblasts to adhere to uncoated 3D scaffolds and microcarriers, tackling the adhesion issues without resorting to costly coatings [2].
Another critical factor in scaling is managing metabolic waste. Dense bioreactor cultures can experience toxic ammonia build-up, which can be avoided by replacing glutamine with non-ammoniagenic alternatives such as α-ketoglutarate, glutamate, or pyruvate [3]. These adjustments are essential when moving beyond small-scale systems and require careful quality control and sensor monitoring to maintain the integrity of myoblasts during production.
Confirming Cell Quality in Adapted Cultures
As cultures are adapted for larger-scale production, ensuring the quality of the cells is crucial. Techniques like transcriptomic, metabolomic, and functional assays are used to verify that cells maintain high levels of CD29 and Ki67 while suppressing Desmin expression. These markers indicate that the cells remain in a proliferative, undifferentiated state during the scaling process [2]. Monitoring these indicators is particularly important when cost-saving measures, such as switching to food-grade components or using partial media changes, are introduced. This step ensures that the transition from research-grade to production-grade systems doesn’t compromise cell quality. Fine-tuning these parameters is a critical step towards making cultivated meat production scalable and cost-efficient.
Serum-Free vs Serum-Based Culture Performance
When optimised, serum-free systems can achieve results that are close to traditional serum-based cultures. The table below highlights key metrics from bovine myoblast cultures grown on uncoated surfaces:
| Metric | Serum-Based (20% FBS + 10% HS) | Conditioned Serum-Free |
|---|---|---|
| Cell Adhesion (24h) | ~6,722 cells/cm² | ~5,985 cells/cm² |
| Cell Proliferation (72h) | ~10,050 cells/cm² | ~8,998 cells/cm² |
| CD29 Expression | High | High |
| Ki67 Expression | High | High |
| Desmin Expression | Suppressed | Suppressed |
Data sourced from npj Science of Food [2]
While serum-based systems still have a slight edge in cell density, serum-free media delivers comparable results in adhesion marker expression and keeps cells undifferentiated - key factors for production. The gap narrows further when specific supplements are added to optimise formulations, making serum-free systems an increasingly practical option for large-scale cultivated meat production.
Conclusion
Switching myoblast cultures to serum-free media comes with its fair share of hurdles: early attachment issues, slower cell growth, and inconsistent results from commercial products. However, simple changes - like removing antibiotics and opting for partial medium replacements - can significantly improve proliferation rates [1]. By carefully choosing media and adding specific growth factors, researchers can close the gap between serum-free and serum-based systems in terms of performance. These advancements pave the way for scaling up production.
Scaling serum-free cultures, however, introduces new layers of complexity. Transitioning cells to 3D bioreactor systems while ensuring they maintain their phenotype demands rigorous quality control. Still, evidence shows that well-optimised serum-free systems can achieve cell densities comparable to those grown in serum-based media. This makes serum-free methods increasingly practical for commercial cultivated meat production.
The economic argument for serum-free media is hard to ignore. With FBS prices continuing to climb, serum-based methods are becoming financially unworkable [1]. This shift isn’t just about technical improvements - it’s about economic survival for the cultivated meat industry.
For researchers and production teams making this transition, sourcing the right materials is essential. From chemically defined media to recombinant growth factors, having access to reliable supplies is key. This is where Cellbase comes in - a dedicated B2B marketplace tailored for the cultivated meat sector. By connecting professionals with verified suppliers of growth media, supplements, and bioreactor systems, Cellbase plays a vital role in helping teams achieve optimised, production-ready serum-free cultures.
FAQs
How can I improve myoblast attachment in serum-free media without laminin or Matrigel?
To improve myoblast attachment in serum-free media without using laminin or Matrigel, consider using conditioned serum-free medium. This approach can promote adhesion and proliferation even on uncoated dishes. Another option is to optimise the medium by adding components such as FGF2, fetuin, and BSA. These adjustments can make a noticeable difference in enhancing cell attachment and growth, eliminating the need for extracellular matrix coatings.
What’s the quickest way to reduce ammonia build-up in serum-free myoblast cultures?
To reduce ammonia build-up in serum-free myoblast cultures, focus on improving the media formulation. One approach is to use conditioned media that promotes both cell adhesion and proliferation while keeping ammonia levels low. Additionally, refining culture conditions can help minimise ammonia production. This might involve adjusting factors like pH, temperature, or nutrient concentrations to better align with the cells' metabolic needs.
How do I validate that myoblasts stay undifferentiated after switching to serum-free media?
To ensure myoblasts remain in their undifferentiated state when cultured in serum-free media, it's crucial to track specific markers. Pax7 is a reliable indicator of undifferentiated myoblasts, while the absence of differentiation markers like myosin heavy chain (MHC) confirms they haven't started differentiating.
You can use techniques such as:
- Immunocytochemistry: To visualise protein expression in cells.
- Flow cytometry: For analysing marker expression in a large cell population.
- qPCR: To measure mRNA levels of key markers.
Additionally, observing the cells under a microscope is essential. Myoblasts should maintain their characteristic appearance, avoiding the formation of multinucleated myotubes, which are a clear sign of differentiation. By combining these methods and monitoring regularly, you can ensure the cells stay undifferentiated.











