Cultivated meat production is limited by slow cell growth and early senescence in primary vs immortalised cell lines. CRISPR gene editing offers targeted solutions to overcome these challenges.
Here are the top five CRISPR targets and their roles in improving cell proliferation, differentiation, and scalability for cultivated meat:
- Myostatin (MSTN): Boosts muscle cell growth by removing natural growth limits.
- P53 (TP53): Extends cell lifespan and increases proliferation rates, though it may reduce differentiation.
- HIF1A: Helps cells survive in low-oxygen environments, essential for dense bioreactor cultures.
- Myogenic Regulatory Factors (MYOD1, MYOG): Drives muscle cell formation and alignment.
- CDKN2A: Bypasses senescence, enabling long-term cell proliferation.
These targets address key issues like replicative senescence, low yields, and serum dependency. However, balancing proliferation with differentiation and ensuring safety are critical for success.
Quick Comparison:
| CRISPR Target | Key Benefit | Challenges |
|---|---|---|
| Myostatin (MSTN) | Promotes muscle growth | Risk of off-target effects; viability issues |
| P53 (TP53) | Extends lifespan, boosts proliferation | Reduced differentiation; safety concerns |
| HIF1A | Supports low-oxygen survival | Requires precise editing to avoid disruptions |
| MYOD1, MYOG | Enhances muscle formation | Balancing proliferation and differentiation |
| CDKN2A | Enables long-term proliferation | Off-target risks; requires serum-free media |
CRISPR technology is reshaping how cultivated meat is produced, aiming for higher yields and lower production costs while addressing ethical concerns.
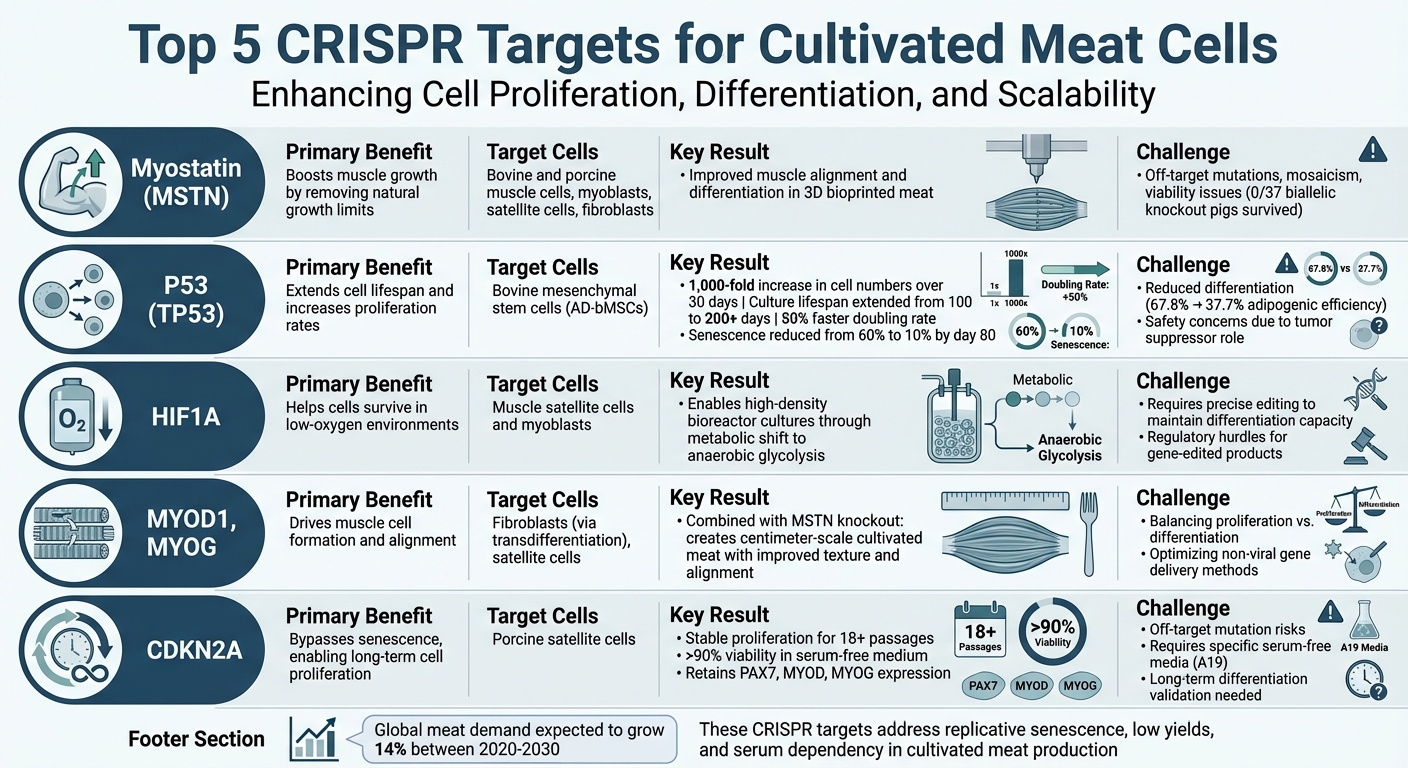
Top 5 CRISPR Targets for Cultivated Meat: Benefits and Challenges Comparison
1. Myostatin Gene (MSTN)
Removing the natural brake on muscle growth is possible by knocking out the MSTN gene. This process promotes increased muscle cell proliferation and differentiation through both hyperplasia and hypertrophy [5][6].
Primary Benefit
In March 2025, researchers at Seoul National University made significant strides by combining MSTN knockout bovine cells with digital light processing 3D bioprinting. This approach improved muscle alignment and differentiation, resulting in cultivated meat with characteristics similar to traditional steak [5].
Earlier, in May 2022, scientists at Northwest A&F University in China used an optimised CRISPR/Cas9 delivery system (100 ng/μL Cas9 mRNA and 200 ng/μL sgRNAs) to create homozygous MSTN knockout sheep. Out of 16 lambs born, four were confirmed as homozygous knockouts. These lambs showed significantly higher body weights at 30, 60, and 90 days compared to their unedited counterparts, all while maintaining meat quality parameters like pH, intramuscular fat, and crude protein levels [6].
Cell Type Applicability
Editing the MSTN gene enhances the myogenic potential of various cell types, including primary myoblasts, satellite cells, fibroblasts (through MYOD1-driven trans-differentiation), and mesenchymal stem cells. This is achieved by overcoming the natural limits on cell proliferation [5][1].
Potential Challenges
Despite its benefits, MSTN knockout is not without complications. It has been linked to viability issues in live animals and technical hurdles such as off-target mutations and mosaicism. For instance, a study in June 2022 reported that while MSTN-edited pigs displayed increased muscle growth, none of the 37 biallelic knockout pigs survived [7][8][6].
"MSTN knockout enhances the production of MYOD1-mediated steak-type cultivated meat." [5]
Next, we’ll explore the P53 tumour suppressor gene and its importance in ensuring sustained cell proliferation.
sbb-itb-ffee270
2. P53 Tumour Suppressor Gene
Disabling the TP53 gene removes critical cell cycle checkpoints, which significantly speeds up cell proliferation. P53 plays a central role as a tumour suppressor, initiating cell cycle arrest and senescence in response to cellular stress. Without this checkpoint, cells can accumulate biomass much faster and sustain longer culture periods [1].
Primary Benefit
In early 2025, Communications Biology published a study highlighting the transformative effects of TP53 editing on bovine mesenchymal stem cells. The findings were striking: a 1,000-fold increase in cell numbers over 30 days and an extended culture lifespan from 100 to over 200 days. Edited cells showed a 50% faster cell doubling rate and, by day 80, senescence levels dropped significantly - from approximately 60% in unedited cells to just 10% in the modified ones. Furthermore, these cells retained a "younger" gene expression profile, marked by enhanced DNA replication and sustained protein synthesis, mirroring early-passage cells [1].
Cell Type Applicability
Bovine adipose-derived mesenchymal stem cells (AD‑bMSCs) are particularly well-suited for TP53 modifications. These cells naturally encounter replicative senescence, which limits their expansion potential. Given that mesenchymal stem cells make up about 25% of the cell sources used in cultivated meat production, TP53 editing offers a practical solution, balancing their ability to remain multipotent with industrial scalability [1].
Potential Challenges
However, this approach is not without its challenges. One significant drawback is reduced differentiation capacity. The Communications Biology study reported a decline in adipogenic differentiation efficiency, from 67.8% in non-edited cells to 37.7% in TP53 knockout clones. Transcriptomic analysis revealed an increase in cell-cycle gene activity but a decrease in genes related to muscle differentiation and adhesion. Additionally, since TP53 is a crucial tumour suppressor and its inactivation is a hallmark of cancer, this strategy raises safety and regulatory concerns. While these cells are intended for consumption rather than medical use, such issues warrant careful consideration [1].
"Among all candidates, TP53 knockout produced the most pronounced effect, with an over 1,000-fold increase in abundance by day 30."
- Communications Biology [1]
Next, let’s explore another important CRISPR target.
3. Hypoxia-Inducible Factor 1-Alpha (HIF1A)
HIF1A plays a critical role in helping cultivated meat cells adapt to low-oxygen environments often encountered in bioreactors with integrated sensors. This regulator becomes especially important when oxygen penetration is limited. By using CRISPR to stabilise HIF1A, cells can sustain energy production and remain viable, even under reduced oxygen levels.
Primary Benefit
Editing HIF1A reprogrammes cell metabolism, shifting it from oxygen-reliant respiration to anaerobic glycolysis. This shift ensures cells continue producing energy in hypoxic conditions. The result? The ability to culture cells at higher densities without the risk of oxygen deprivation. This is a game-changer for scaling up cultivated meat production, especially when creating thicker tissue constructs.
Cell Type Applicability
Muscle satellite cells and myoblasts benefit the most from HIF1A edits. These are the key players in muscle fibre development, and their survival in tightly packed bioreactors is crucial for achieving high yields. Stabilised HIF1A allows these cells to switch metabolic pathways effectively, ensuring they stay viable even during long culture periods.
Potential Challenges
One major challenge is ensuring that edited cells maintain their ability to differentiate into functional muscle fibres after multiple passages. This requires technical fine-tuning to avoid any loss of differentiation capacity. Beyond the lab, regulatory hurdles and public perception add complexity. Gene-edited meat products must pass extensive safety assessments for human consumption and environmental impact before they can hit the market. Meanwhile, consumer acceptance of such products varies widely across different regions [3]. These challenges highlight the need to perfect gene editing techniques before expanding to new targets. Next, we’ll explore genes that further enhance myogenic differentiation.
4. Myogenic Regulatory Factors (MRFs: MYOD1, MYOG)
MYOD1 plays a critical role in committing cells to the myogenic lineage, while MYOG facilitates the fusion of myoblasts into mature myotubes. Interestingly, overexpressing MYOD1 can reprogramme fibroblasts into myogenic cells, effectively bypassing the natural senescence limits seen in primary satellite cells [5].
Primary Benefit
When MYOD1 overexpression is combined with MSTN knockout in bovine fibroblasts, and integrated with DLP 3D bioprinting on 100‑µm groove‑patterned hydrogels, the results are impressive. This approach enhances muscle alignment and differentiation, enabling the creation of centimetre‑scale cultivated meat structures. A study published in March 2025 in the Journal of Animal Science and Biotechnology showcased this method, using non‑viral delivery of MYOD1 alongside CRISPR-mediated MSTN knockout to engineer bovine fibroblasts [5]. By eliminating inhibitory signals on muscle differentiation, this strategy directs cells towards a stronger myogenic identity, resulting in cultivated meat with better texture. This dual approach highlights the importance of precisely balancing proliferation and differentiation pathways.
Cell Type Applicability
Fibroblasts are an excellent starting point for MYOD1 targeting. Easily obtained through simple skin biopsies (similar to routine ear tagging), these cells, derived from the mesoderm, respond well to MYOD1-induced transdifferentiation [5]. On the other hand, satellite cells, while capable of contributing up to 30% of muscle nuclei in neonates, diminish significantly with age. This makes fibroblasts a more practical and scalable option for industrial-scale cultivated meat production.
Potential Challenges
One of the main hurdles is finding the right balance between cell proliferation and differentiation. For instance, genetic modifications aimed at boosting cell expansion - like TP53 knockout - can inadvertently suppress key muscle-differentiation factors, potentially hindering the cells' ability to mature into functional muscle tissue [1]. Additionally, while non-viral methods such as the Piggybac transposon system are preferred for food safety reasons, they require careful optimisation to ensure efficient gene delivery. External factors, like 3D-printed microgrooves, remain vital for achieving proper muscle fibre alignment [5].
5. Cell Cycle Regulators (e.g., CDKN2A)
CDKN2A plays a key role in triggering senescence, effectively halting cell division. By using CRISPR/Cas9 to knock out CDKN2A, researchers can bypass the Hayflick limit. This allows muscle stem cells to continue dividing far beyond their usual lifespan while still maintaining their ability to differentiate into functional muscle tissue. This breakthrough tackles one of the biggest challenges in cultivated meat production: producing the massive quantities of viable, functional cells needed for industrial-scale manufacturing.
Primary Benefit
Targeting CDKN2A directly addresses the problem of limited cell proliferation in cultivated meat production.
Editing CDKN2A enhances scalability and reduces costs. For example, in June 2025, a research team from Nanjing Agricultural University, led by Shijie Ding, Chunbao Li, and Guanghong Zhou, published their findings in Food Materials Research. They successfully developed CRISPR-edited porcine satellite cell lines with a CDKN2A knockout. These cells demonstrated stable proliferation for over 18 passages in A19 serum-free medium, with viability rates exceeding 90%. Importantly, the cells retained the expression of key myogenic regulators (PAX7, MYOD, and MYOG) and differentiated into mature, MyHC-positive myotubes. When seeded onto plant-based 3D scaffolds, these edited cells formed meat-like constructs with improved chewiness and gumminess [2].
"The CRISPR-based CDKN2A knockout cells provide a renewable source of muscle progenitors, reducing dependence on repeated animal biopsies." – Food Materials Research [2]
Cell Type Applicability
Porcine satellite cells, which are crucial for muscle regeneration, respond particularly well to CDKN2A editing. This approach also holds potential for other livestock species. A key advantage of CDKN2A-edited cells is their compatibility with serum-free media formulations. This eliminates the need for costly and ethically contentious foetal bovine serum, reducing variability between batches and minimising contamination risks [2].
Potential Challenges
While the Nanjing study highlighted significant benefits, there are challenges to broader applications of CRISPR in cultivated meat. Off-target mutations remain a concern and must be carefully monitored. Additionally, regulatory safety standards for genetically modified food products must be rigorously followed. Researchers also need to ensure long-term differentiation to guarantee that the final product closely resembles natural muscle tissue. This makes protocol refinement and thorough validation of 3D scaffolds essential [2].
These findings, alongside other CRISPR targets, are summarised in the following comparison table.
Comparison Table
Table: The following summarises the five CRISPR targets that improve cell proliferation, differentiation, and metabolic adaptation for scalable cultivated meat production.
| CRISPR Target | Primary Benefit | Target Cell Types | Challenges |
|---|---|---|---|
| Myostatin (MSTN) | Boosts muscle growth | Bovine and porcine muscle cells | Requires detailed genomic understanding; risk of unintended phenotypic changes if not carefully managed [4] |
| P53 (TP53) | Dramatically increases proliferation; delays replicative ageing (over 1,000-fold rise in cell abundance by day 30) [1] | Bovine mesenchymal stem cells (bMSCs) | Reduced differentiation capacity; adipogenic differentiation drops from 67.8% to 37.7%; downregulation of muscle-related genes [1] |
| HIF1A | Improves metabolic adaptation | Bovine and porcine cells | Requires careful editing to avoid metabolic disruptions [4] |
| MRFs (MYOD1, MYOG) | Key for muscle fibre formation and regeneration | Porcine satellite cells (muscle stem cells) [2] | Challenging to sustain high expression levels during rapid expansion for industrial scaling [2] |
| CDKN2A | Supports stable proliferation over 18+ passages with >90% viability; bypasses ageing [2] | Porcine satellite cells (muscle stem cells) [2] | Needs specific serum-free media (e.g., A19) to preserve stemness and differentiation over long-term culture [2] |
Selecting the right targets involves balancing cell proliferation with the ability to differentiate effectively. This highlights the importance of fine-tuning these processes in cultivated meat cell engineering.
Conclusion
CRISPR technology holds immense promise for addressing critical challenges in cultivated meat production, including limited cell proliferation, senescence, and high production costs. For instance, TP53 knockout has been shown to increase cell abundance by over 1,000 times within just 30 days [1]. Similarly, CDKN2A edits allow cells to proliferate stably over 15–18 passages with more than 90% viability in serum-free conditions [2]. This reduces the reliance on expensive animal serum and minimises the need for repeated animal biopsies.
However, achieving the right balance between rapid cell proliferation and the ability to differentiate into muscle tissue remains a key challenge. While TP53 knockout significantly boosts cell numbers, it can hinder differentiation. Therefore, maintaining the role of regulators like MYOD1 and MYOG is crucial for generating mature muscle tissue suitable for cultivated meat.
For research teams aiming to apply these genetic strategies, Cellbase provides an invaluable resource. This platform offers access to specialised CRISPR tools, validated cell lines, and bioreactors tailored for genetic engineering in cultivated meat. By connecting cultivated meat companies with trusted suppliers, Cellbase helps bridge the gap between lab research and industrial-scale production, ensuring researchers have the tools needed to scale these advancements effectively.
With global meat demand expected to grow by 14% between 2020 and 2030 [1], these CRISPR targets pave the way for scalable and cost-efficient solutions in cultivated meat production.
FAQs
Which CRISPR target boosts growth most without affecting differentiation?
The best CRISPR target for enhancing growth while maintaining differentiation is the serum-free, genetically engineered satellite cell system. This method supports consistent cell growth and effective differentiation, making it a strong choice for large-scale cultivated meat production.
How can TP53 or CDKN2A edits be made safe for cultivated meat?
To make sure that TP53 or CDKN2A edits are safe for cultivated meat, several important steps are taken. These include thorough genetic stability testing, establishing structured cell banking systems, and using advanced tools like next-generation sequencing to spot any mutations. On top of this, following strict regulatory compliance guidelines ensures both safety and consistency throughout the production process.
What edits help cells thrive in low-oxygen, high-density bioreactors?
Developing serum-free media tailored with the right mix of nutrients, growth factors, lipids, non-essential amino acids, and antioxidants plays a key role in boosting cell proliferation and differentiation. These adjustments not only support better cell viability but also enhance functionality, especially in challenging conditions like low-oxygen and high-density environments.











