Immortalised cells are solving a key challenge in cultivated meat production: the limited proliferation of primary cells. Unlike primary cells, which stop dividing after a set number of cycles, immortalised cells can divide indefinitely, making them ideal for large-scale production. These cells are created through genetic modifications (e.g., TERT and CDK4 expression) or spontaneous mutations, enabling high-density growth in bioreactors.
Key Points:
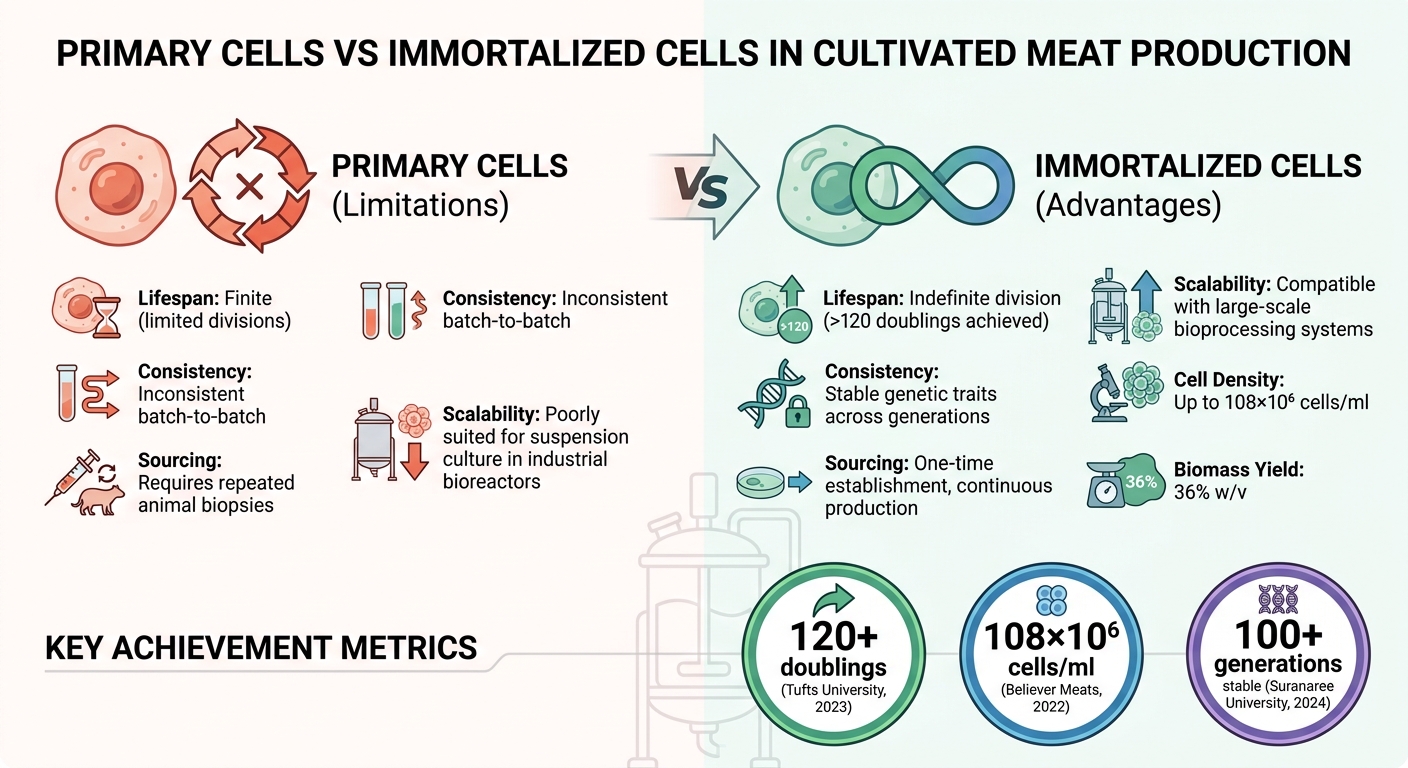
- Primary Cell Limitations: Primary cells have finite lifespans and are inconsistent, requiring repeated animal biopsies. They are also poorly suited for suspension culture in industrial bioreactors.
- Immortalised Cell Advantages: Continuous division, stable genetic traits, and compatibility with scalable bioprocessing systems.
-
Case Studies:
- Tufts University (2023): Developed immortalised bovine satellite cells using TERT and CDK4, achieving over 120 doublings.
- Believer Meats (2022): Created spontaneously immortalised chicken fibroblasts with high cell densities (108×10⁶ cells/ml).
- Suranaree University (2024): Produced hTERT-immortalised porcine muscle stem cells capable of indefinite proliferation.
Immortalised cells also enable the production of complex cultivated meat products by differentiating into muscle, fat, and other tissues. However, challenges remain, such as ensuring genetic stability, transitioning to serum-free media, and meeting regulatory requirements. Despite these hurdles, immortalised cells are becoming a cornerstone of scalable cultivated meat production.
Primary Cells vs Immortalized Cells in Cultivated Meat Production
Case Studies: How Companies Are Using Immortalised Cells
Tufts University's Immortalised Bovine Satellite Cells
In May 2023, researchers at the Tufts University Centre for Cellular Agriculture (TUCCA) shared a breakthrough in ACS Synthetic Biology. They successfully developed immortalised bovine satellite cells (iBSCs) by introducing TERT and CDK4 expression. This allowed the cells to exceed the Hayflick limit, achieving more than 120 doublings while still maintaining their ability to differentiate into muscle fibres [2][5].
"Using these new persistent bovine cell lines, studies can be more relevant, literally getting right to the meat of the matter." - Andrew Stout, Lead Researcher, Tufts University Centre for Cellular Agriculture [5]
These cell lines were made available through the TUCCA Open Cell Bank and distributed by commercial providers like Kerafast. In 2024, TUCCA collaborated with the Good Food Institute to further expand the bank, incorporating immortalised bovine fibroblast lines (e.g., TU-GFI-SCL1). These fibroblast lines were originally developed by SCiFi Foods using CRISPR/Cas9 technology [4]. By adopting this open-access approach, the initiative could save the cultivated meat industry between £16 million and £80 million for every 10 start-ups, as developing a single commercial cell line can cost between £1.6 million and £8 million [6].
Meanwhile, Upside Foods has taken a different route, focusing on chicken cells.
Upside Foods' Chicken Cell Line Approach
Upside Foods has implemented a proprietary strategy that combines TERT overexpression with CRISPR-based modifications. While both Tufts and Upside Foods leverage TERT to prevent telomere shortening, Upside Foods opts for CRISPR modifications instead of CDK4 expression to achieve immortalisation at a commercial scale [3].
This method has helped the company secure key regulatory achievements, such as preliminary FDA approval for its cultivated chicken [5]. However, Upside Foods continues to face challenges, particularly in scaling production while maintaining the differentiation capacity needed to produce authentic muscle tissue.
These examples highlight how immortalised cell lines are helping to address production challenges and scale cultivated meat manufacturing.
Mesenchymal Stem Cells for Immortalisation
Benefits of MSCs in Cultivated Meat
Immortalised mesenchymal stem cells (MSCs) offer the potential for unlimited proliferation and the ability to differentiate into multiple cell types, such as muscle, fat, and bone, making them ideal for producing complex cultivated meat products [7].
By overexpressing hTERT (human telomerase reverse transcriptase), researchers can restore telomerase activity in MSCs. This allows the cells to divide indefinitely without losing their stem cell properties [7]. For example, in December 2024, a team at Suranaree University of Technology, led by Parinya Noisa, successfully developed hTERT-immortalised porcine muscle stem cells. These cells demonstrated indefinite proliferation and retained their ability to differentiate into myofibres in vitro. Impressively, the study showed that these cells could be cultured for over 100 generations without losing their differentiation potential [7].
"hTERT can immortalise primary porcine MSCs and preserve their stem cell characteristics. For research and cultivated meat technologies, immortality may be valuable."
- Parinya Noisa, Corresponding Author, Suranaree University of Technology [7]
Immortalised MSCs also exhibit accelerated growth and biomass accumulation, which is advantageous for scaling up production [1]. Some immortalised lines are further optimised for growth in single-cell suspensions and serum-free media, enabling them to achieve the high cell densities required for large-scale bioreactors [1]. However, findings from the Suranaree study highlighted a potential limitation: while low-passage cells remained stable, tumour formation was observed in cells cultured beyond 100 generations [7].
The next section delves into MSC sourcing from various species and their specific roles in cultivated meat production.
MSC Sources Across Species
MSCs can be derived from a variety of species, each contributing unique benefits to cultivated meat production. For instance:
- Bovine MSCs: These are often obtained from bone marrow or muscle-derived progenitor cells and are critical for developing beef muscle fibres [2][7].
- Porcine MSCs: Sourced from muscle satellite cells and bone marrow stromal cells, these are used in producing cultured pork muscle and fat [7].
- Chicken embryonic fibroblasts: While not traditional MSCs, these cells share similar traits. They can be transdifferentiated into adipocyte-like cells, which play a role in enhancing flavour and aroma [1].
The effectiveness of MSC sources depends significantly on their proliferative capacity and ability to adapt to suspension culture. Primary cells from these sources typically have limited lifespans and lose their differentiation potential over time, making immortalisation a critical step for commercial applications [7]. Suspension-adapted MSCs are particularly valuable for achieving high-density growth in bioreactors, which is essential to meeting the production demands of cultivated meat at scale [1].
Regulatory and Production Requirements
Food Safety and Genetic Stability
As immortalised cell lines become a cornerstone of cultivated meat production, addressing regulatory and scalability challenges is essential. In the United States, the Food and Drug Administration (FDA) oversees the initial stages, including cell collection and banking, ensuring the safety of the production process and the establishment of cell lines [8]. Once harvesting begins, the United States Department of Agriculture's Food Safety and Inspection Service (USDA-FSIS) takes over, focusing on processing and labelling for livestock and poultry products [9,10].
A major regulatory focus lies in ensuring genetic stability and the safety of modifications used for immortalisation. Companies must demonstrate that cell lines remain stable over multiple generations without oncogenic transformations [9,4]. Notably, in December 2022, Believer Meats (formerly Future Meat Technologies) published findings in Nature Food showcasing the spontaneous immortalisation of chicken fibroblasts. Led by Chief Scientific Officer Yaakov Nahmias, the study revealed that these cells maintained genetic stability and achieved densities of 108 × 10⁶ cells per millilitre in continuous cultures, all without relying on genetic modification [1]. This approach allows companies to sidestep challenges linked to genetically modified organisms, especially in regions with stringent GM food regulations. As of March 2025, the FDA had completed pre-market consultations for cultured chicken, seafood, and pork fat cells, marking a critical milestone for the industry's regulatory pathway [8].
Production facilities are required to adhere to Current Good Manufacturing Practices (CGMP) while implementing Hazard Analysis and Critical Control Points (HACCP) systems. USDA-FSIS inspections occur at least once per shift during harvest and processing, ensuring compliance and consistency [9,10]. These rigorous standards are vital for maintaining batch uniformity and achieving high production yields.
Consistency and Scalability
Beyond genetic stability, producers must ensure that cell lines can transition smoothly into scalable production systems. Achieving consistent, reproducible performance at an industrial scale requires constant monitoring of cell line integrity. To this end, producers conduct CNV (copy number variation) and SNV (single nucleotide variation) analyses while adapting immortalised cells to suspension growth in serum-free media. This step is pivotal for enabling high-density expansion in large-scale bioreactors [1]. Such genomic monitoring ensures that cell lines retain their desired traits across multiple generations.
Immortalised cell lines capable of reaching densities of 108 × 10⁶ cells per millilitre and achieving 36% w/v biomass yields exemplify the level of consistency demanded by regulatory authorities [1].
"While some may question whether it is safe to ingest immortalised cells, in fact, by the time the cells have been harvested, stored, cooked, and digested, there is no viable path to continued growth."
- David Kaplan, Stern Family Professor of Biomedical Engineering, Tufts University [5]
Before commercialisation, the final biomass undergoes rigorous screening for pathogens such as Salmonella and Listeria, alongside thorough pesticide testing [1]. Species verification processes are also applied throughout production to ensure consistency. For producers navigating these stringent regulatory and production requirements, platforms like Cellbase offer access to a network of verified suppliers specialising in cultivated meat production. These measures are key to driving the industry's progress toward large-scale commercial viability.
sbb-itb-ffee270
Barriers and Opportunities
Current Development Challenges
Immortalised cell lines face several technical and regulatory hurdles. One significant issue is genetic modification restrictions, which limit the use of advanced tools like CRISPR or viral oncogenes in food production [1]. As a result, researchers are turning to spontaneous immortalisation, a process that requires extensive time and resources to identify and characterise viable cell lines.
Another key issue is genetic stability. Maintaining chromosomal integrity is critical, as regular monitoring for copy number variations (CNVs) and single nucleotide variations (SNVs) is essential. For instance, a December 2024 study by Suranaree University found that hTERT-immortalised porcine muscle stem cells remained stable through many cycles. However, passaging beyond 100 cycles increased tumourigenic risks, highlighting a safety threshold that must not be overlooked [7].
Technical challenges also include suspension adaptation and the transition to serum-free media. Converting anchorage-dependent primary cells into single-cell suspensions suitable for high-density bioreactor expansion remains complex. Similarly, designing serum-free media that supports rapid cell growth while preserving differentiation potential continues to be a major obstacle. Overcoming these challenges is crucial for advancing cultivated meat production.
Future Opportunities in Research and Commercialisation
Despite these challenges, research is uncovering promising strategies to address these barriers. For example, spontaneous immortalisation and transdifferentiation techniques are emerging as viable solutions for scalable production.
Spontaneous immortalisation offers a non-GMO alternative. In December 2022, Believer Meats demonstrated that spontaneously immortalised chicken fibroblasts could achieve cell densities of 10⁸ cells per millilitre in continuous culture, with biomass yields reaching 36% w/v [1]. Sensory trials of the resulting cultured chicken product were highly successful, scoring 4.5 out of 5.0. Among 150 participants, 85% indicated they were "extremely likely" to replace traditional meat with this product [1].
Transdifferentiation techniques present another innovative path. By using biochemical triggers like lecithin-activated PPARγ, researchers can convert immortalised fibroblasts into fat-storing adipocytes without additional genetic modifications [1]. This method addresses regulatory concerns while expanding production options. To support these advancements, platforms like Cellbase offer access to verified suppliers of specialised tools, including bioreactors, serum-free media formulations, and analytical equipment. These resources are accelerating the shift from laboratory research to commercial-scale production, paving the way for broader adoption of cultivated meat technologies.
Lab meat: a love story | Dr. Natalie Rubio | TEDxTufts
Conclusion
Immortalised cell lines are reshaping the cultivated meat industry. By overcoming cellular senescence, these cell lines eliminate the need for repeated animal biopsies, offering a dependable and consistent biomass source [1]. This reliability addresses a critical issue for the sector: batch-to-batch variability, which can compromise both product quality and regulatory compliance.
Evidence from Tufts University and Believer Meats highlights the viability of both genetic and spontaneous immortalisation for achieving commercial benchmarks. For instance, Tufts' bovine satellite cells demonstrated over 120 doublings while retaining their ability to differentiate into muscle cells [2]. Similarly, Believer Meats achieved biomass yields of 36% w/v and reported positive consumer feedback [1]. These milestones pave the way for tackling remaining technical and regulatory hurdles.
Future progress will depend on several key factors: precise genetic monitoring, the use of tailored serum-free media, and optimised suspension culture systems. Spontaneous immortalisation offers a non-GMO pathway, potentially easing regulatory challenges, while transdifferentiation techniques could enable a single cell line to produce both muscle and fat components [1]. As Professor Yaakov Nahmias and his team have observed:
"immortalisation without genetic modification and high-yield manufacturing are critical for the market realisation of cultured meat" [1]
For teams navigating these complexities, platforms like Cellbase provide access to verified suppliers of essential tools such as bioreactors, analytical equipment, and serum-free media formulations. The alignment of improved cell line technologies, clearer regulatory frameworks, and robust industrial infrastructure is positioning immortalised cells as a foundational element of scalable cultivated meat production. Resources like Cellbase, a specialised B2B marketplace for cultivated meat, will play a pivotal role as the industry integrates these advancements.
FAQs
Are immortalised cells safe to eat in cultivated meat?
Immortalised cells, when used in cultivated meat, are typically regarded as safe for consumption after they have been harvested, stored, and cooked. This is because they undergo processing methods comparable to those applied to other food ingredients. However, discussions continue around potential safety concerns, largely stemming from their unique ability to proliferate indefinitely.
How do producers prove an immortalised cell line stays genetically stable?
Producers maintain the genetic stability of immortalised cell lines through detailed testing across numerous cell passages. This process involves genomic analyses, such as karyotyping and whole-genome sequencing, to identify any mutations. Additionally, functional assays are performed to evaluate growth and differentiation capabilities. By routinely monitoring cell behaviour and genetic markers, producers ensure these cell lines remain stable and meet the stringent safety and quality requirements essential for cultivated meat production.
What makes a cell line suitable for serum-free, suspension bioreactor growth?
For scalable cultivated meat production, a suitable cell line must exhibit several key traits. It should be immortalised to enable indefinite proliferation, maintain genetic stability over time, and demonstrate rapid growth in a serum-free, suspension bioreactor environment. These characteristics are essential for efficient and large-scale production processes.











