Cultivated meat is transforming how we think about food production, offering the taste and texture of conventional meat without the same health concerns. A significant focus is improving fat composition to make it healthier.
Here’s what you need to know:
- Healthier fats like monounsaturated and omega-3 fatty acids are being prioritised over saturated fats, which are linked to cardiovascular risks.
- Pathway engineering uses metabolic and genetic techniques to influence fat production at a cellular level.
- Methods include:
- CRISPR-Cas9 gene editing to reduce saturated fat production.
- Enzyme overexpression (e.g., stearoyl-CoA desaturase) to increase monounsaturated fats.
- Growth media supplementation to boost omega-3 content without genetic modification.
- Challenges include scaling production and maintaining flavour while improving nutritional value.
This approach is helping cultivated meat producers create products that are healthier and better suited to modern dietary needs.
Engineering Cell Lines for Cultured Meat and Sustainable Cellular Agriculture #culturedmeat
sbb-itb-ffee270
How Fatty Acid Synthesis Works in Cultivated Meat
Fatty acid synthesis plays a key role in shaping the fat content of cultivated meat, particularly when aiming to reduce saturated fat levels. By managing fat composition at the cellular level, scientists can influence whether the resulting meat contains saturated, monounsaturated, or polyunsaturated fats. This is achieved through three interconnected metabolic pathways, each contributing to the fat profile. Let’s break them down.
The Fatty Acid Synthase Pathway
The process starts with the fatty acid synthase (FAS) pathway, which is responsible for producing saturated fats. At the heart of this pathway is the enzyme acetyl-CoA carboxylase (ACC), which catalyses the first step of fatty acid synthesis in the cytoplasm. This enzyme also serves as a marker for mature adipocytes - cells that are crucial in cultivated meat production [5].
Interestingly, the way cells produce fatty acids can vary by species. For instance, bovine cells tend to use acetate, while human cells rely more on glucose for fatty acid synthesis [1]. These differences highlight the importance of tailoring the pathway to suit specific needs.
Desaturase Enzymes and Monounsaturated Fats
Once saturated fats are synthesised, desaturase enzymes step in to convert them into monounsaturated fatty acids (MUFAs), which are considered healthier. For example, these enzymes can transform saturated fats like palmitic acid or stearic acid into oleic acid (C18:1), a fat commonly associated with the health benefits of olive oil [5].
Cultured fat derived from fibro-adipogenic progenitor cells tends to have higher levels of oleic acid and lower levels of palmitic acid compared to conventional beef fat [5]. This shift in composition can be further influenced by culture conditions. For instance, using serum-free medium formulations has been shown to increase triglyceride accumulation in bovine adipose stem cells by 66% compared to traditional serum-containing media [1].
Beyond MUFAs, further adjustments target polyunsaturated fats to improve the nutritional profile.
Polyunsaturated Fatty Acid Pathways
Polyunsaturated fatty acids (PUFAs), such as omega-3 and omega-6 fatty acids, offer a way to enhance the nutritional value of cultivated meat. These essential fats, including linoleic acid, are not produced by the human body and must be obtained from food.
However, cultivated meat often has lower PUFA levels than conventional meat [5]. To address this, researchers focus on the expression of genes involved in triglyceride synthesis, such as PPARγ, Gpd1, and FABP4 [6][1]. By targeting these pathways, PUFA content can be increased, making the meat more nutritious.
Additionally, the composition of PUFAs can be fine-tuned through media supplementation. By adding specific lipids to the growth medium, scientists can either replicate the fat profile of natural animal tissue or create a product with enhanced nutritional benefits, all without genetic modification [3].
| Pathway/Enzyme | Primary Function | Impact on Fat Composition |
|---|---|---|
| Fatty Acid Synthase (FAS) | Produces long-chain saturated fatty acids | Increases saturated fat content (e.g., palmitic acid) |
| Acetyl-CoA Carboxylase (ACC) | Rate-limiting step in fatty acid synthesis | Essential for overall lipid accumulation levels |
| Desaturase Enzymes | Converts saturated bonds to double bonds | Increases monounsaturated fats (MUFAs) like oleic acid |
| PPARγ Signalling | Regulates adipogenic gene expression | Controls the maturation and volume of lipid storage |
Genetic and Metabolic Engineering Methods for Improved Fat Profiles
Understanding how fatty acids are synthesised has opened up opportunities to refine fat composition in cultivated meat using genetic and metabolic engineering. These approaches aim to lower saturated fat levels while boosting healthier fatty acids, tailoring the nutritional profile to modern dietary preferences.
CRISPR-Cas9 for Targeted Gene Editing
CRISPR-Cas9 technology allows scientists to adjust fat composition by precisely editing DNA. This method can target and disable genes responsible for producing saturated fats, without introducing foreign DNA from other species [7].
"CRISPR is a gene editing tool that we can think of as a pair of molecular scissors, and we can take those scissors and guide them to a specific location in the genome and make a precise cut in the DNA." - Dawn Cayabyab, Ph.D. student, UC Davis [7]
In June 2025, researchers at Nanjing Agricultural University, including Shijie Ding, Chunbao Li, and Guanghong Zhou, demonstrated the potential of CRISPR/Cas9 in cultivated meat production. By knocking out the CDKN2A gene in porcine satellite cells, they addressed cell ageing, creating a renewable source of muscle progenitors. These modified cells maintained stable growth for over 18 passages with more than 90% viability. Using 3D edible scaffolds, the team successfully developed meat-like constructs, showcasing the scalability and genetic optimisation possible with CRISPR [8].
This precise editing tool enables researchers to directly select cells with lower saturated fat levels. Additionally, modifying enzyme expression offers another pathway to refine fat profiles.
Overexpression of Stearoyl-CoA Desaturase (SCD)
Another method to improve fat composition involves boosting the activity of stearoyl-CoA desaturase (SCD). This enzyme converts saturated fatty acids, like stearic acid, into monounsaturated fatty acids, such as oleic acid [2]. By increasing SCD expression, the lipid profile can be shifted towards monounsaturated fats, which are widely regarded as healthier.
This approach works particularly well when paired with serum-free media systems. Studies have shown that these systems can increase triglyceride accumulation by 66% compared to traditional serum-containing media [9]. The result is cultivated meat with a healthier fat composition, aligning with nutritional recommendations.
Growth Media Supplementation for Omega-3 Enrichment
Beyond genetic modifications, adjusting the growth medium can further enhance fatty acid profiles. For instance, supplementing the culture medium with unsaturated fatty acids like linolenic acid boosts intracellular lipid levels without harming cell viability [4].
A carefully designed mix of fatty acids can replicate the fat profile of natural beef. This method supports total lipid concentrations up to 400 µM in the medium - well above the toxic threshold for saturated fats such as palmitic acid. Unsaturated fatty acids, like linolenic acid, are better tolerated by cells, with non-toxic levels reaching up to 200 µM, compared to palmitic acid's toxicity at around 40 µM [4].
"The incorporation of health-promoting fatty acids, such as n-3 polyunsaturated fatty acids (PUFAs), represents a potential strategy to enhance the nutritional value of these products." - Waris Mehmood et al., Aarhus University [4]
When combined with biomaterials for 3D culture systems, such as spheroids, media supplementation becomes even more impactful. This combination has been shown to increase triglyceride accumulation by up to 34% compared to 2D monolayer cultures [9]. However, omega-3 levels must be carefully managed to avoid creating "fish-like" flavours in the final product [4].
Comparing Different Pathway Engineering Approaches
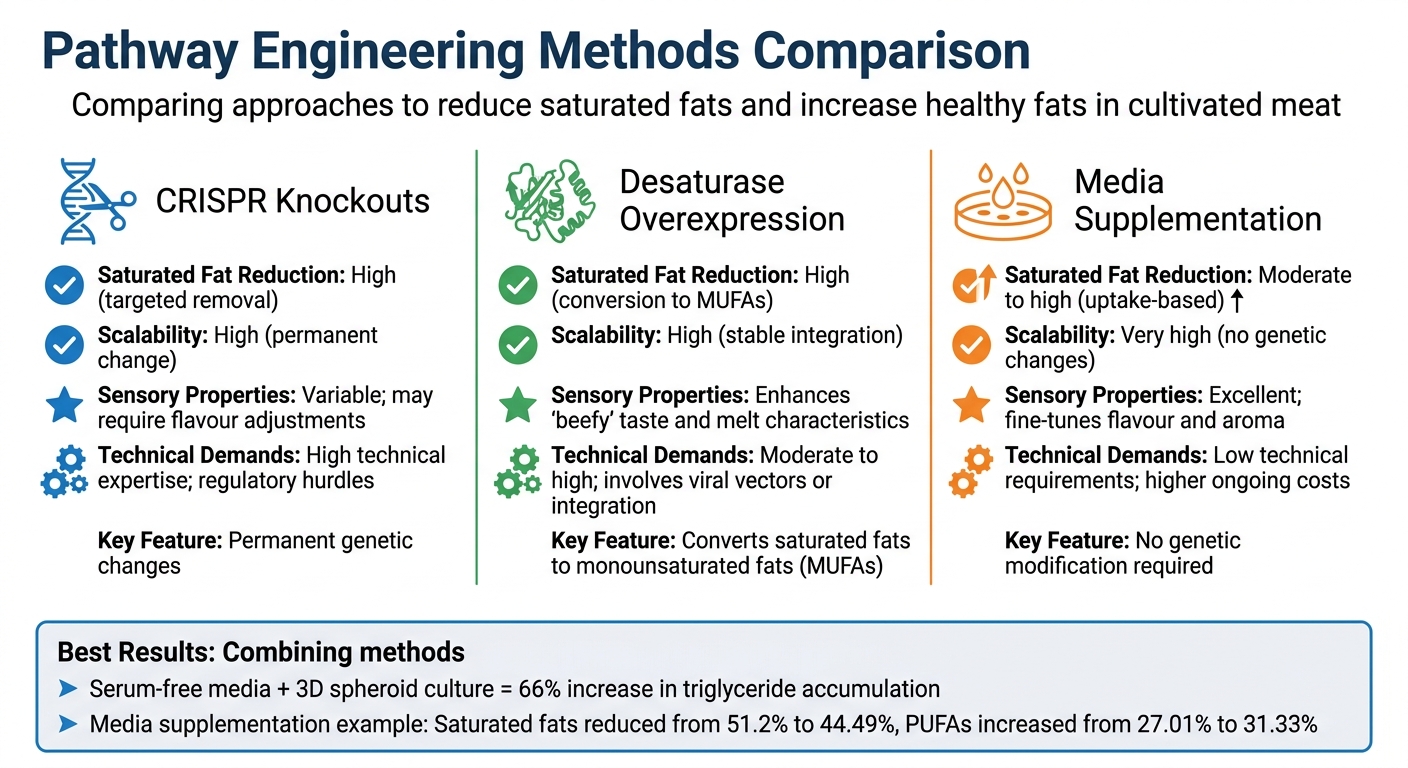
Pathway Engineering Methods for Healthier Fat Profiles in Cultivated Meat
This section dives into the strengths and trade-offs of various pathway engineering methods, building on the techniques discussed earlier. Each approach offers unique benefits for improving fat profiles in cultivated meat, and the choice largely hinges on production goals, technical resources, and nutritional targets.
Let’s start with CRISPR-based gene knockouts. These create permanent genetic changes, making them highly scalable once implemented. However, they come with challenges, including strict regulatory requirements and the need for advanced technical expertise. On the other hand, desaturase overexpression, particularly involving the SCD enzyme, strikes a balance. This method establishes stable cell lines that continuously convert saturated fats into healthier monounsaturated fats (MUFAs), eliminating the need for ongoing external inputs.
Then there’s media supplementation, which shines for its simplicity and quick application. A 2026 study demonstrated its effectiveness: using olive oil and soybean lecithin as lipogenic inducers reduced saturated fatty acids in cultivated pork from 51.2% to 44.49%, while increasing polyunsaturated fatty acids from 27.01% to 31.33% [10]. Although straightforward and effective, media supplementation comes with recurring costs, requiring careful financial planning. When combined with advanced 3D spheroid systems, this method can further boost triglyceride accumulation.
Method Comparison Table
| Method | Saturated Fat Reduction | Scalability | Sensory Properties | Technical Demands |
|---|---|---|---|---|
| CRISPR Knockouts | High (targeted removal) | High (permanent change) | Variable; may require flavour adjustments | High technical expertise; regulatory hurdles |
| Desaturase Overexpression | High (conversion to MUFAs) | High (stable integration) | Enhances "beefy" taste and melt characteristics | Moderate to high; involves viral vectors or integration |
| Media Supplementation | Moderate to high (uptake-based) | Very high (no genetic changes) | Excellent; fine-tunes flavour and aroma | Low technical requirements; higher ongoing costs |
From this comparison, it’s clear that the best results often come from combining methods. For instance, pairing serum-free media with 3D spheroid culture has been shown to increase triglyceride accumulation by 66% and 34%, respectively, compared to traditional techniques [9]. This layered approach allows researchers to refine both genetic and environmental factors, creating cultivated meat with optimised fat profiles that appeal to consumers and meet health standards.
Equipment and Materials for Pathway Engineering
Creating healthier fat profiles in cultivated meat requires specialised tools and biological materials that aren't typically available from general suppliers. This field has seen significant growth, with over 140 companies projected to invest more than £2.7 billion by 2025 [12].
Key resources for this work include:
- Cell lines: Examples include porcine adipose stem cells, bovine myosatellite cells, and water buffalo adipose cells [11].
- Serum-free media formulations: Essential for scalable production [4].
- Fatty acids: Such as oleic, linoleic, linolenic, stearic, and palmitic acids to fine-tune fat profiles [4].
- Bioreactors: Options include stirred-tank, airlift, packed-bed, or perfusion systems [12].
- 3D spheroid culture systems: Used for enhanced cell maturation [12].
- Analytical tools: Including RT-qPCR, flow cytometry, and high-resolution imaging systems like the Agilent BioTek Cytation 5 [4].
Finding Equipment and Materials on Cellbase
For researchers in cultivated meat, sourcing these specialised materials can be streamlined through Cellbase. This platform connects scientists with verified suppliers and categorises products specifically for pathway engineering needs - covering cell lines, growth media, scaffolds, bioreactors, metabolite sensors, and downstream processing equipment [12].
Cellbase simplifies procurement with industry-specific filters, such as GMO status, biosafety levels, regulatory compliance, scale compatibility, and species [11]. For instance, when selecting cell lines for lipid accumulation studies, users can apply filters like "Adipose Stem Cell" and "GMO Status" to narrow down suitable options quickly [11].
Sensitive biological materials, such as primary cell lines and growth factors, are handled with cold chain logistics to maintain viability during shipping. Additionally, researchers can consult "Cell Ag Experts" on Cellbase for technical advice, particularly when scaling up from lab-based equipment to manufacturing-scale bioreactors. The platform’s "Ask us anything" feature offers valuable support during this transition [12]. These tools and services make sourcing faster and reduce the risks associated with navigating multiple general suppliers.
Setting Up a Pathway Engineering Workflow
Establishing an efficient pathway engineering workflow requires careful attention to material compatibility and process control. For example, scaffolds need to endure 37°C culture conditions, sterilisation, and cooking processes [12]. Real-time sensors for glucose, lactate, and ammonium levels are critical for maintaining precise metabolic control [12].
Cellbase also enhances the procurement experience with transparent pricing, fast checkout, and global shipping, ensuring researchers worldwide can access vital materials [12]. For teams transitioning to serum-free media - a pivotal step in scaling sustainable cultivated meat production - the platform offers verified materials to support this shift [12]. By using a specialised marketplace like Cellbase, researchers can reduce technical risks and make sourcing decisions more efficiently compared to relying on multiple general suppliers.
Conclusion and Future Directions
Pathway engineering has opened up exciting possibilities for fine-tuning fat profiles in cultivated meat. By leveraging techniques like serum-free media optimisation and advanced 3D culture systems, researchers can now achieve a level of nutritional precision that traditional livestock farming simply cannot replicate.
Some of the most promising breakthroughs come from combining multiple strategies. For example, the FaTTy pig cell line showcases how improved MUFA profiles can be achieved without the need for gene editing [2]. Similarly, Martin Krøyer Rasmussen from Aarhus University demonstrated in December 2025 that exposing differentiated bovine satellite cells to a carefully balanced fatty acid mix at 400 µM resulted in the highest lipid droplet accumulation while maintaining cell viability [4].
However, challenges remain, particularly when it comes to scaling up production. In 3D cultures, mass transport limitations - like oxygen and nutrient gradients - can lead to cell death in dense tissue cores [1]. A practical solution lies in two-step bioprocessing, which uses high-density bioreactors for cell expansion, followed by specialised 3D differentiation phases [1]. Additionally, while enriching products with omega-3 fatty acids shows promise, careful calibration is essential to avoid the risk of fish-like off-flavours at higher concentrations [4].
The shift towards serum-free media is another critical area of progress. Beyond the ethical and environmental advantages, serum-free formulations are proving effective in enhancing both cell proliferation and lipid accumulation [1]. These advances are transforming the way cultivated meat is produced.
Ultimately, success in this field depends on selecting the right combination of cell types, culture systems, and media formulations to achieve specific product goals. Whether the aim is to lower saturated fat levels, boost omega-3 content, or create realistic marbling, the pathway engineering strategies outlined here provide a strong foundation for crafting the next generation of nutritionally optimised cultivated meat. These developments signal a healthier, more commercially viable future for the cultivated meat industry.
FAQs
Which pathway engineering method best reduces saturated fat in cultivated meat?
One effective way to lower saturated fat in cultivated meat is by using serum-free media. This technique fine-tunes lipid accumulation in muscle satellite cells, allowing for greater control over fatty acid profiles. As a result, it helps decrease the saturated fat content in the final product. These advancements play a key role in creating healthier fat profiles for cultivated meat.
How can omega-3 levels be increased without changing the cells’ DNA?
Adding microalgae-derived omega-3 fatty acids to the culture media can increase omega-3 levels in cultivated meat. This method improves its nutritional profile without altering the DNA of the cells.
Will healthier fat profiles affect the flavour, aroma, or mouthfeel of cultivated meat?
Healthier fat profiles are expected to influence the flavour, aroma, and texture of cultivated meat. Fat is a key player in shaping these sensory qualities. The good news? Cultivated fat has already shown it can closely mimic traditional fat in both its chemical makeup and sensory attributes. This means it strikes a balance between offering health benefits and maintaining the taste people love.











