To ensure consistency in cultivated meat production, precise control of bioreactor parameters is critical. Factors like temperature, pH, dissolved oxygen (DO), and nutrient levels must remain within specific ranges to optimise cell growth and quality. Even slight deviations can disrupt production, causing cell death or reduced yields.
Key takeaways:
- Temperature: 37–39°C supports growth; deviations slow metabolism or induce stress.
- pH: 7.2–7.4 is ideal; shifts affect enzyme activity and cell viability.
- DO Levels: 30–60% saturation avoids hypoxia or oxidative stress.
- Nutrient Levels: Glucose (5–20 mM) and glutamine (2–4 mM) must remain stable to sustain growth.
Advanced monitoring tools, like Raman spectroscopy and inline sensors, enable real-time adjustments, reducing variability and improving yields. Bioreactor design - stirred-tank, perfusion, or packed-bed - also plays a role, with each suited to specific production goals. Consistent quality relies on automated control systems, regular parameter validation, and managing transitions from cell proliferation to differentiation. These practices minimise batch failures and ensure reliability as production scales up.
Trends in cultivated meat scale-up and bioprocessing
Critical Bioreactor Parameters and Their Impact on Consistency
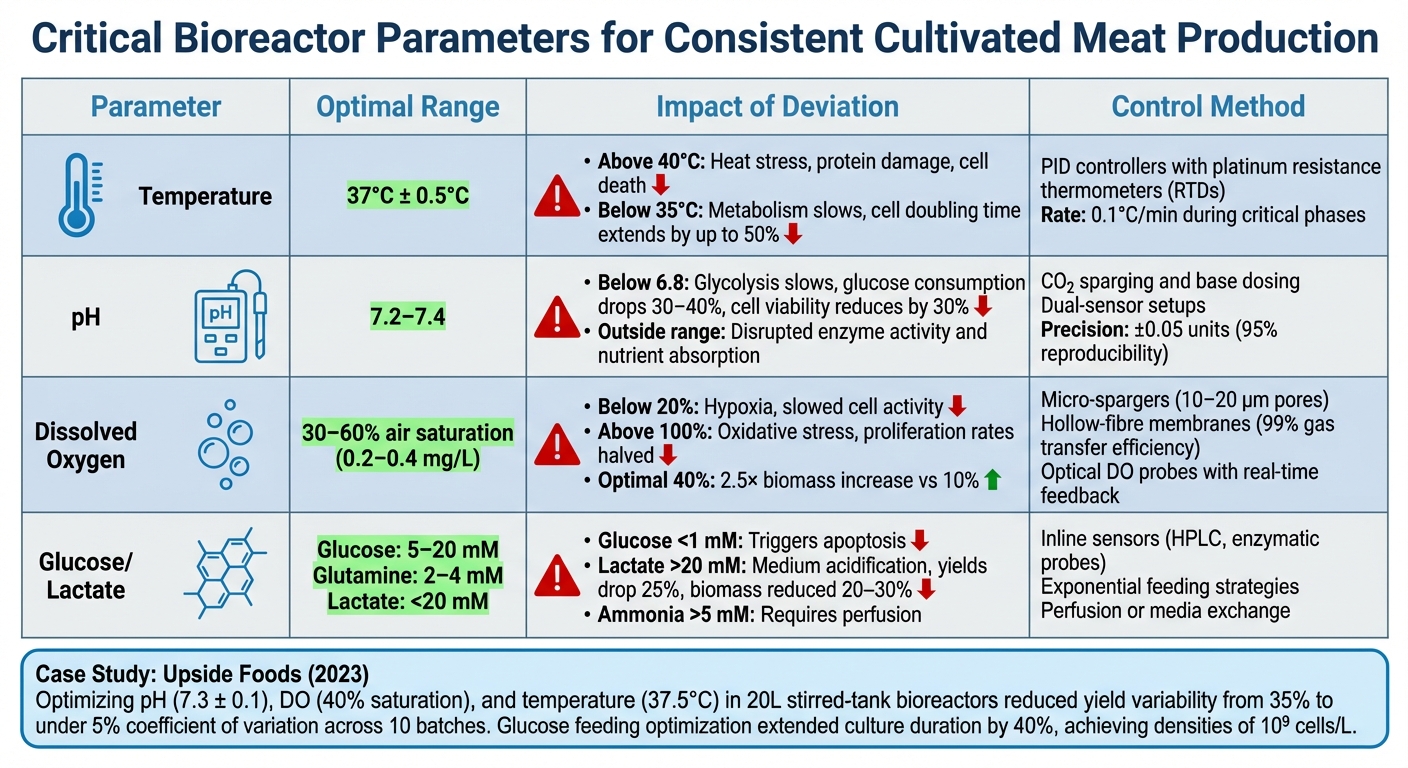
Critical Bioreactor Parameters for Cultivated Meat Production
Producing cultivated meat consistently hinges on maintaining tight control over key bioreactor parameters like temperature, pH, dissolved oxygen (DO), and nutrient levels. These factors directly affect cell metabolism, growth, and the quality of the final product. Even minor deviations can lead to significant variability between batches. By carefully managing these parameters, producers can lay a solid foundation for further process improvements.
Temperature Control
Cultivated meat cells thrive at temperatures between 37–39°C, mimicking the conditions within the body [3]. If the temperature climbs above 40°C, heat stress can occur, leading to protein damage and cell death. On the other hand, temperatures below 35°C slow metabolism, extending cell doubling times by as much as 50% [3]. High-precision tools like platinum resistance thermometers (RTDs) are paired with PID controllers to regulate temperature changes gradually - typically at a rate of 0.1°C per minute during critical phases like inoculation and expansion [3][4]. To ensure uniform conditions, redundant sensors are strategically placed across different zones of the bioreactor, helping to eliminate temperature gradients that could disrupt cell growth.
pH Regulation
For optimal cell performance, the pH of the culture environment should remain between 7.2 and 7.4 [4]. Straying outside this range can disrupt enzyme activity and nutrient absorption. For example, when pH drops below 6.8 - often due to lactate build-up - glycolysis slows, cutting glucose consumption by 30–40% and reducing cell viability by up to 30% [4]. Automated systems, like CO₂ sparging and base dosing, help maintain pH stability. Dual-sensor setups offer redundancy, while peristaltic pumps assist with precise acid or base adjustments. Predictive control algorithms, which account for metabolite production, can maintain pH levels within ±0.05 units, achieving up to 95% reproducibility in pilot-scale trials [5].
Dissolved Oxygen and Gas Exchange
DO levels between 30–60% air saturation (approximately 0.2–0.4 mg/L) are ideal for consistent cell growth [5]. Levels below 20% can lead to hypoxia, slowing cell activity, while levels above 100% may cause oxidative stress, reducing proliferation rates by half [5]. Maintaining a DO level of 40% saturation has been shown to boost biomass production by 2.5× compared to cultures at 10%. Efficient oxygen delivery systems, such as micro-spargers with 10–20 μm pores, ensure proper gas exchange while preventing foam formation. Hollow-fibre membranes, with up to 99% gas transfer efficiency, support uniform DO distribution. Real-time feedback from optical DO probes allows dynamic adjustments to gas flow rates, ensuring optimal conditions [6].
Nutrient Concentration and Metabolite Accumulation
Keeping nutrient levels stable is essential for batch consistency. Glucose concentrations should remain between 5–20 mM to sustain glycolysis without causing osmotic stress. Similarly, glutamine levels should stay within 2–4 mM to avoid nitrogen shortages [6]. A drop in glucose below 1 mM can trigger apoptosis, while lactate levels above 20 mM can acidify the medium, reducing yields by around 25%. Excess lactate also inhibits pyruvate dehydrogenase, forcing cells into less efficient metabolic pathways and cutting biomass by 20–30%. Ammonia accumulation above 5 mM may require perfusion or media exchange [3][4]. Inline sensors, such as HPLC or enzymatic probes, enable real-time monitoring and feeding strategies like exponential feeding. A 2023 study by Upside Foods demonstrated how optimising pH (7.3 ± 0.1), DO (40% saturation), and temperature (37.5°C) in 20 L stirred-tank bioreactors reduced yield variability from 35% to under 5% coefficient of variation across 10 batches. Additionally, fine-tuning glucose feeding extended culture duration by 40%, achieving densities of 10⁹ cells/L [5].
| Parameter | Optimal Range | Impact of Deviation | Control Method |
|---|---|---|---|
| Temperature | 37°C ± 0.5°C | Up to 50% slower growth; stress induction | PID, RTD |
| pH | 7.2–7.4 | Up to 30% viability loss; metabolic shifts | CO₂/base, dual probes |
| Dissolved Oxygen | 30–60% saturation | Hypoxia or oxidative stress; yield ↓ (~25%) | Sparging, membranes |
| Glucose/Lactate | 5–20 mM / <20 mM | Growth inhibition; yield ↓ (15–40%) | Perfusion, inline sensors |
Careful management of these parameters not only ensures batch consistency but also sets the stage for more advanced bioreactor systems and control techniques.
Bioreactor Design and Parameter Control
Building on the importance of managing critical parameters, the design of a bioreactor plays a major role in ensuring process consistency. Choosing the right bioreactor design is essential for maintaining stable conditions - like temperature, pH, dissolved oxygen (DO), and nutrient levels - throughout cultivated meat production. However, each design comes with its own set of benefits and challenges.
Stirred-Tank Bioreactors
Stirred-tank bioreactors are widely used in the biopharma industry and can scale up to 20,000 L for animal cell production [1]. They rely on mechanical impellers to evenly mix heat, oxygen, and nutrients, ensuring precise control over parameters like temperature, pH, and DO. However, the turbulence caused by impellers and bubble rupturing can create hydrodynamic shear stress, which may harm fragile cultivated meat cells. To address this, newer impeller designs that promote laminar flow or the use of poloxamers can help minimise cell damage [1]. These adjustments are key to maintaining stable conditions and optimising the production process.
Perfusion Systems
Perfusion systems work by continuously exchanging media, providing fresh nutrients while removing waste products like lactic acid and ammonia. This constant exchange helps maintain stable levels of nutrients and metabolites, reducing the variability often seen in batch processes. For instance, hollow fibre perfusion reactors support cell densities of 10⁸ to 10⁹ cells/mL, outperforming the 10⁷ to 10⁸ cells/mL typically achieved in stirred-tank reactors [1]. Economic studies suggest that integrated continuous processing with perfusion systems can lead to a 55% reduction in capital and operating expenses over a decade compared to batch processing [1]. However, the trade-off lies in their complexity - managing microfluidics and flow rates demands advanced control systems and precise monitoring.
Packed-Bed Bioreactors
Packed-bed bioreactors are particularly effective for scaling adherent cells, thanks to their high surface-to-volume ratio. These systems often use microcarriers, which allow cells to migrate between surfaces without requiring harsh detachment enzymes during expansion. In one experiment using a 3 L stirred-tank bioreactor, bovine satellite cells achieved a density of 60,000 cells/cm² by employing an intermittent stirring regime (30 minutes off, 5 minutes on) to facilitate bead-to-bead transfer [2]. This approach reduces the need for manual intervention, lowering contamination risks and labour costs. However, packed-bed designs can face challenges with nutrient and oxygen gradients, especially in larger volumes, which can affect consistency across the culture.
The table below highlights the main features of these bioreactor designs:
| Feature | Stirred-Tank Bioreactor | Perfusion System | Packed-Bed Bioreactor |
|---|---|---|---|
| Mixing Mechanism | Mechanical impeller/stirring | Continuous media flow/recycling | Flow through a fixed bed/substrate |
| Cell Density | 10⁷–10⁸ cells/mL [1] | 10⁸–10⁹ cells/mL [1] | High (via microcarriers/scaffolds) |
| Consistency Focus | Uniform control of temperature, pH, and DO | Stable nutrient and metabolite levels | Stable cell adherence and surface area |
| Primary Challenge | Hydrodynamic shear stress | Complex microfluidics and flow rates | Risk of nutrient/oxygen gradients |
High-throughput miniature bioreactors offer a practical and cost-effective way to fine-tune parameters before scaling up production [1]. Platforms like Cellbase provide access to these miniature bioreactors, along with verified suppliers for stirred-tank, perfusion, and packed-bed systems tailored to cultivated meat production. This enables early-stage optimisation and helps procurement teams select equipment that aligns with their specific needs and production goals. Combined with parameter controls, thoughtful bioreactor design is a crucial step towards reducing batch variability.
Real-Time Monitoring and Process Control
To get the best results from bioreactors, it's essential to keep a close eye on key factors like pH, dissolved oxygen (DO), and metabolite levels. Real-time monitoring tools make it possible to track these variables continuously, allowing production teams to make quick adjustments when needed. This kind of proactive approach helps minimise inconsistencies between batches in cultivated meat production. Let’s dive into the tools and systems that make this level of precision possible.
Process Analytical Technology (PAT) Tools
Process Analytical Technology (PAT) is all about keeping manufacturing processes on track by measuring critical quality attributes in real time. In the world of cultivated meat bioreactors, PAT tools can monitor multiple variables at once. For example:
- Raman spectroscopy can measure glucose, lactate, glutamine, pH, and biomass in less than a minute without extracting samples.
- Near-infrared spectroscopy is great for tracking biomass and metabolites.
- Capacitance biosensors provide direct information about viable cell density.
These tools don’t just measure - they help prevent problems. For instance, multi-wavelength fluorescence and near-infrared spectroscopy can detect early signs of issues, like lactate levels exceeding 20 mM, which could harm cell viability. Raman spectroscopy has even been shown to spot glutamine depletion 2–4 hours faster than traditional methods like HPLC analysis, helping avoid yield losses.
A practical example? In June 2022, Upside Foods used Raman spectroscopy combined with model predictive control in a 50 L bioreactor for bovine myoblast cultures. This reduced batch failure rates from 18% to just 2% over 12 runs and boosted cell densities to 5×10⁷ cells/mL - 25% above their target.
Other tools like optical dissolved oxygen probes and pH electrodes provide continuous, precise measurements, ensuring parameters stay within strict limits. Companies like Cellbase make it easier for teams to source specialised PAT tools, including Raman spectrometers and biosensors, specifically designed for cultivated meat production.
Integration of Monitoring Data for Automated Control
Real-time measurements are just the beginning. Automated control systems take this data and turn it into immediate actions to keep processes on track. For example, if pH starts to drift, the system might automatically adjust the base addition. A drop in dissolved oxygen? The system can tweak gas sparging rates to compensate.
Basic adjustments, like controlling agitator speeds (usually between 50 and 150 rpm for shear-sensitive cells), are handled by PID controllers. Meanwhile, machine learning models can predict metabolite trends, enabling preemptive tweaks - like adjusting nutrient feeds before lactate builds up.
Recent examples highlight the power of these systems:
- In September 2023, Mosa Meat used near-infrared PAT and soft sensors in perfusion bioreactors to maintain pH between 6.8 and 7.2 and dissolved oxygen above 30% for 21 days. This resulted in a 45% yield improvement, reaching 1.8×10⁸ cells/g tissue.
- In March 2024, CellX integrated multi-parameter biosensors with AI in 200 L stirred-tank systems. By detecting pH drifts three hours early and automatically adjusting CO₂ levels, they stabilised cell proliferation rates at 0.35 per day across eight batches, achieving a 2.2-fold increase in biomass compared to their baseline.
These automated systems don’t just improve consistency - they also reduce batch failures by 40–60%, cut down on labour costs by limiting manual sampling, and increase yields by 20–30%. In one study, monitored bioreactors reached cell densities 1.5 times higher than manually controlled ones, hitting 10⁸ cells/mL.
Of course, challenges remain. Sensor fouling in high-protein media can be addressed with self-cleaning probes. Data overload can be tackled with AI analytics, and calibration drift over time (7–14 days) can be resolved using automated in-situ checks.
Experts at the Good Food Institute suggest combining inline Raman spectroscopy with at-line mass spectrometry for a more complete monitoring setup. They also recommend using digital twins - virtual bioreactor models updated in real time - to simulate and fine-tune parameters before scaling up. This approach can achieve near-perfect parameter stability, up to 99%.
sbb-itb-ffee270
Managing Transition Phases
To ensure consistent quality in cultivated meat, managing the transition from cell proliferation to differentiation is crucial. This process involves fine-tuning both mechanical and biological factors at just the right moment to guide cells through this critical phase.
Adjusting Mechanical and Biological Cues
Cells become more delicate as they move from proliferation to differentiation, requiring careful handling. Differentiating cells are particularly sensitive to shear forces, so bioreactors should switch to low-shear impeller designs, like pitched-blade or anchor impellers, during this stage [9]. Computational Fluid Dynamics (CFD) can be used to optimise stirring speeds, ensuring cells are protected. For instance, GoodMeat employs 10 units of 250,000 L stirred-tank bioreactors with CFD-optimised low-shear designs and edible microcarriers to support uniform differentiation [9].
Oxygen levels also need precise adjustment. While high oxygenation supports cell expansion, muscle cell differentiation thrives in a hypoxic environment of 2–10% oxygen. This activates hypoxia-inducible factors (HIFs), which are essential for promoting myogenic differentiation [9]. Temperature control is equally critical - maintaining 37°C with fluctuations limited to ±0.1°C prevents metabolic disruptions [9].
Microcarrier confluence must stay within 15,000–25,000 cells/cm² to avoid contact inhibition during the transition. An intermittent stirring regime, such as 30 minutes off followed by 5 minutes on, can facilitate cell transfer between microcarriers while minimising shear stress [2].
Once these mechanical conditions are optimised, the focus shifts to biochemical signals to drive tissue formation.
Optimising Differentiation Conditions
Alongside mechanical adjustments, changes to the medium and growth factor levels are essential to initiate differentiation. For example, reducing FBS from 20% to 2% or switching to serum-free media with growth factor levels reduced to one-tenth can trigger this process [10].
Muscle differentiation is activated by targeting the mTOR signalling pathway. This involves adding insulin or insulin-like growth factor 1 (IGF1) and essential amino acids to stimulate protein synthesis [10]. For fat tissue development, introducing free fatty acids (FFAs) encourages stem cells to differentiate into adipocytes [10].
| Parameter | Proliferation Phase | Differentiation Phase |
|---|---|---|
| Oxygen Level | High (supports density) | 2–10% (hypoxia-induced) [9] |
| Serum/GFs | High (e.g. 20% FBS) | Low (e.g. 2% FBS or reduced GF levels) [10] |
| Key Additives | Proliferation factors | Insulin, IGF1, Free Fatty Acids [10] |
| Mechanical Stress | Moderate mixing | Low-shear (protects myotubes) [9] |
Aleph Farms uses bovine embryonic stem cells in suspension with an animal component-free medium to create thin-cut beef steaks by differentiating cells into collagen-producing cells and muscle fibres [10]. Similarly, Super Meat relies on chicken embryonic stem cells to produce cultivated chicken meat, ensuring batch consistency through rapid propagation [10].
UPSIDE Foods has developed cell lines with genetically encoded glutamine synthetase, which reduce toxic ammonia levels by around 20% while providing additional energy substrates [1].
Overextending seed train doublings can compromise differentiation potential [1]. Monitoring transcription factors like PAX7 (a marker for satellite cells) and MYOG (essential for myoblast fusion into myotubes) helps identify the optimal timing for transitions [10].
Platforms such as Cellbase simplify access to essential tools like edible microcarriers and low-shear impeller systems, which are critical for successfully managing these transitions.
Quality Assurance and Standardisation
Producing consistent batches of cultivated meat requires rigorous quality control, especially since formal ISO standards for the industry are not yet in place. This means companies must establish their own internal benchmarks, focusing on three key areas: cell viability (aiming for over 90% across batches), consistent phenotype expression, and product quality metrics, such as uniform fibre structure.
Internal Standardisation Protocols
In the absence of specific regulatory guidelines, many producers turn to pharmaceutical standards, like those from ISCT, to shape their processes. Key performance indicators (KPIs) are defined for each stage of production. For example, target cell densities range between 10⁷–10⁸ cells/mL, doubling times are set at 24–48 hours, and biomass yields should exceed 10 g/L. These metrics are reviewed and validated quarterly.
Advanced techniques like real-time PCR and flow cytometry are used to ensure consistency in cell phenotypes. For instance, myogenic markers such as MyoD must remain above 80%. Additional tools, including ATP assays and metabolite profiling, help detect any deviations early in the process. Specific metabolic indicators, like maintaining a lactate-to-glucose ratio below 1.5, are critical to avoiding metabolic stress. A 2023 study highlighted the impact of improved quality assurance protocols, showing a drop in batch failure rates from 25% to just 4% in bovine cell cultivation when routine dissolved oxygen validation was introduced.
These internal standards rely heavily on precise sensor calibration and continuous process monitoring, which are detailed below.
Routine Parameter Validation
Daily calibration of key sensors is essential to keep crucial parameters within tight tolerances: pH (±0.1), temperature (±0.5°C), and dissolved oxygen (±5% saturation). Immediate corrective actions are required if these limits are exceeded.
A strict schedule is vital for maintaining consistency. This includes daily checks for pH and dissolved oxygen, bi-weekly calibrations using certified buffers and NIST-traceable thermometers, and monthly mock production cycles. Such practices have proven effective. For example, after implementing weekly sensor recalibration in pilot-scale bioreactors, metabolite accumulation variability dropped to below a 5% coefficient of variation. Similarly, standardising perfusion protocols to keep shear stress under 0.1 Pa improved cell viability consistency by 15–20%. Tools like Cellbase make it easier for producers to access verified sensors and calibration equipment specifically designed for cultivated meat production.
These stringent validation measures are crucial for reducing batch variability and ensuring the reliable production of cultivated meat.
Conclusion
Producing cultivated meat consistently hinges on maintaining tight control over bioreactor parameters like temperature, pH, dissolved oxygen, and nutrient levels. Even minor deviations, such as a 0.2 pH unit shift, can slash yields by half. On the flip side, optimised systems can cut batch failure rates by up to 50% through real-time monitoring and rigorous quality checks[3][11]. Tools like Process Analytical Technology (PAT) allow for automated adjustments, keeping variability between batches under 5%[12][6].
Selecting the right bioreactor design - whether stirred-tank, perfusion, or packed-bed - depends on production goals. Automated feedback systems and regular parameter validation are key to scaling from pilot projects to full-scale production. For instance, daily sensor calibrations and weekly mock runs have achieved 95% consistency during differentiation phases while lowering production costs by 20–40% through increased cell densities[13][7].
Looking ahead, experts anticipate that by 2030, refined parameter control and advanced monitoring systems could deliver tenfold efficiency gains, cut energy consumption by 25%, and sustain cell viability rates above 90%[11][8]. These improvements highlight the importance of equipment tailored specifically for cultivated meat, making precise bioreactor management a cornerstone of commercial success.
To support this, sourcing the right tools and machinery is critical. Cellbase, the first dedicated B2B marketplace for cultivated meat, bridges the gap between R&D teams and verified suppliers. By offering industry-specific equipment with transparent pricing, it eliminates the inefficiencies of generic procurement platforms, streamlining the path to production.
FAQs
Which bioreactor parameter typically causes batch failures first?
pH is one of the most critical bioreactor parameters, often being the first to trigger batch failures. Drops in pH can occur due to metabolic acidification or the accumulation of CO₂, both of which can hinder cell growth. To ensure stable performance in cultivated meat production, it's crucial to closely monitor and regulate pH levels.
How can shear damage be prevented while ensuring proper mixing of oxygen and nutrients?
To protect cells in cultivated meat bioreactors, it's crucial to manage shear forces effectively. This involves fine-tuning agitation and fluid dynamics to create a safe environment for cell growth. Here are some key approaches:
- Use gentle bioreactor systems: Opt for designs like airlift or rocking bioreactors, which naturally minimise shear stress.
- Control impeller speeds: Keep impeller speeds below 1.5 m/s to reduce turbulence that could harm cells.
- Maintain appropriate Kolmogorov eddy lengths: Ensure eddy lengths stay above 20 μm to prevent excessive shear forces.
Additionally, computational modelling can be a valuable tool for identifying potential shear zones within the bioreactor. This allows for targeted adjustments to minimise damage. Protective agents, such as Pluronic F68, can also be introduced to shield cells from shear stress.
By combining these strategies, you can achieve efficient oxygen and nutrient mixing while safeguarding the delicate cells needed for cultivated meat production.
What should change in the bioreactor when cells switch to differentiation?
When cells begin the differentiation process in a bioreactor, it’s crucial to fine-tune parameters like pH, temperature, and shear forces to create the right environment. For instance:
- The pH should be kept within the range of 6.8 to 7.4.
- The temperature needs to be maintained at approximately 37°C.
- Agitation and oxygen levels should be adjusted carefully to encourage proper cell maturation.
These adjustments ensure the cells have the conditions they need to develop effectively.











