Scaling cultivated meat production requires precise control over bioreactor conditions. Minor fluctuations in pH, oxygen levels, or temperature can significantly impact yields when moving from lab-scale to commercial operations. Traditional manual monitoring methods often fail to detect these issues early, risking contamination, inefficiencies, and higher costs.
This case study examines how a production facility implemented real-time monitoring systems, achieving:
- Improved efficiency: Automated sensors continuously tracked critical parameters like pH, oxygen, and cell density, reducing cell damage and ensuring consistent yields.
- Better compliance: Automated data logging created GMP-compliant batch records, simplifying regulatory inspections.
- Lower costs: Automation reduced labour needs and enabled the use of more affordable nutrients.
The facility integrated advanced sensors, flow controllers, and microfluidic devices into bioreactors, ensuring sterility and continuous oversight. Deployment took 18–24 months, with measurable improvements in production efficiency and cost management.
Real-time monitoring has become a key solution for scaling cultivated meat production, offering precise control, reduced risks, and streamlined compliance.
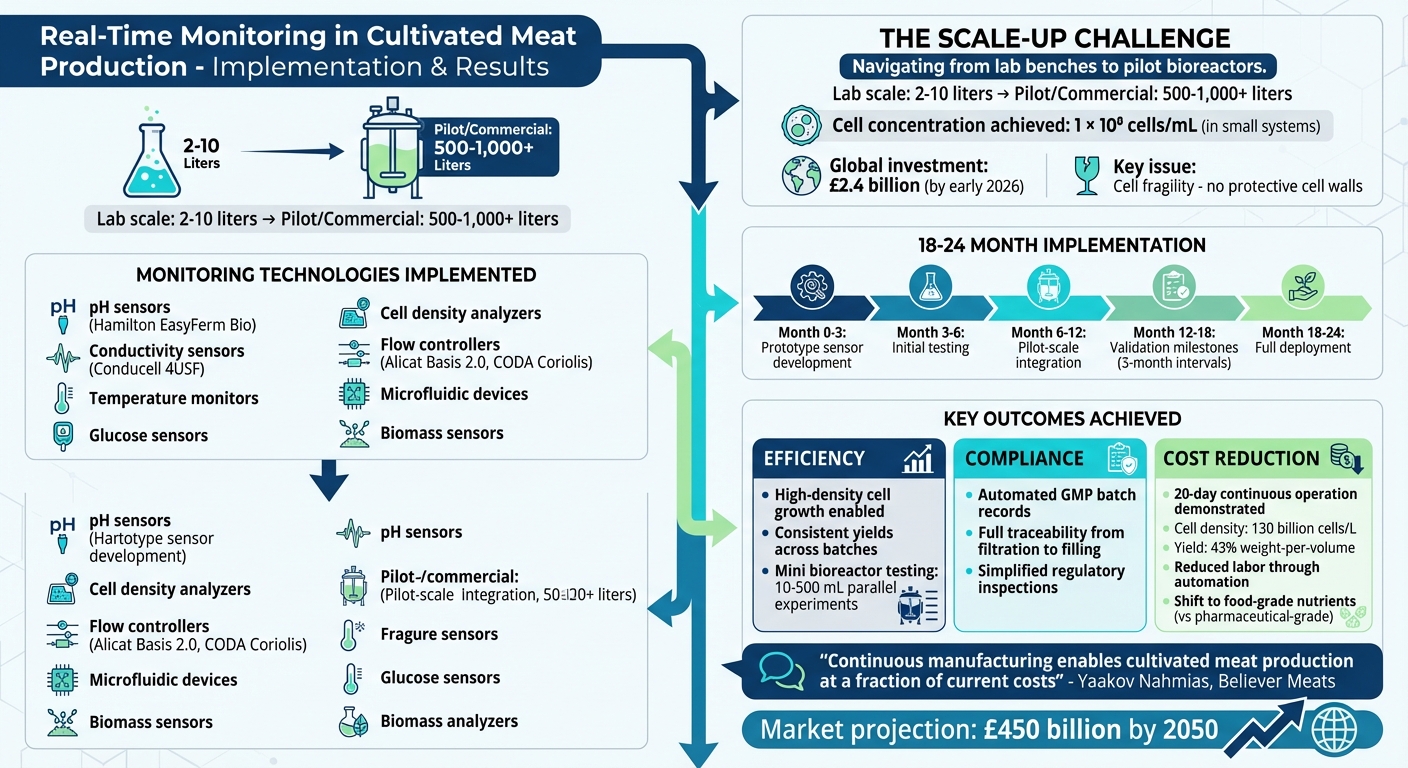
Real-Time Monitoring Implementation Timeline and Key Results in Cultivated Meat Production
The Challenge: Limited Visibility During Scale-Up
Moving from Lab-Scale to Pilot and Commercial Production
Scaling cultivated meat production from small 2–10 litre lab bioreactors to systems exceeding 1,000 litres brings a host of challenges that manual oversight simply cannot handle. For instance, while small perfusion bioreactors have achieved high cell concentrations of over 1 × 10⁸ cells per millilitre, replicating these results in larger stirred tank systems with simpler media has been inconsistent[7]. This case study highlights a facility facing this exact issue - what worked seamlessly in a research lab fell apart when scaled to a 500-litre pilot system.
The root of the problem lies in cell fragility. Unlike the robust microbial cells used in traditional fermentation, cultivated meat cells lack protective cell walls, making them highly susceptible to damage from fluid forces in larger bioreactors[1]. Even minor turbulence at these scales can cause significant cell destruction. Traditional scaling rules, such as the 4× seed train factor, proved inadequate given the tight economic constraints of cultivated meat production[7].
These challenges made it clear that a more dependable and continuous monitoring system was essential.
Problems with Traditional Monitoring Methods
At larger scales, traditional monitoring methods fell short. Manual sampling, for example, posed a contamination risk and created inefficiencies. Each time a sample was taken from the bioreactor, the aseptic environment was jeopardised - a critical issue for the large-scale operations required for commercial production[7]. Maintaining sterility during data collection became a top priority for the production team.
"The system would require aseptic operation (including viral exclusion) at a very large scale beyond the current practice to avoid contamination and potential batch loss."
- ACS Food Science & Technology[7]
Manual processes also drove up labour costs and made it harder to maintain accurate GMP batch records, which are vital for regulatory compliance. Without real-time data on crucial parameters like pH, oxygen levels, and shear stress thresholds, problems often went unnoticed until production yields had already suffered.
The financial stakes were enormous. By early 2026, over £2.4 billion had been invested globally in cultivated meat technology[7], creating immense pressure to achieve consistent, scalable production. To bridge the gap between laboratory success and commercial viability, the facility needed a monitoring solution capable of delivering real-time insights without compromising sterility or efficiency.
sbb-itb-ffee270
How Real-Time Monitoring Systems Were Implemented
Monitoring Technologies Used
The facility introduced sensors to monitor critical bioreactor parameters continuously while maintaining sterility. The system's backbone included temperature monitors, glucose sensors, and cell density analysers. For pH levels, Hamilton Company's EasyFerm Bio sensors were employed in both preparation tanks and bioreactors. Meanwhile, Conducell 4USF sensors kept an eye on conductivity, ensuring media consistency during on-site production[4].
To manage precise gas and fluid delivery, the team utilised Alicat Basis 2.0 and CODA Coriolis Mass Flow Controllers, which were essential for maintaining stable culture environments. Alicat L-Series Liquid Flow Meters tracked media and nutrient delivery rates. Additionally, biomass sensors and lab-on-a-chip microfluidic devices were integrated for real-time monitoring. This shift from manual checks to automated data collection allowed the team to monitor everything from nutrient levels to metabolite build-up in a structured and efficient way.
These advanced sensor capabilities created a foundation for smooth integration with the facility's bioprocessing systems.
Connecting to Existing Bioprocessing Systems
Integration efforts prioritised compatibility with the facility's existing bioreactor and incubation setups. Sensors were carefully positioned in turbulent bioreactors to shield the delicate cultivated meat cells[6]. The monitoring systems were directly connected to automated bioprocess control systems, ensuring compliance monitoring and issuing alerts whenever parameters fell outside the desired range[2][3].
Glucose sensors provided continuous updates on nutrient levels, triggering alerts when adjustments were necessary. Cell density analysers, using live-cell imaging, tracked population growth, enabling precise interventions during scale-up phases[2][5]. A scale-down testing approach was used to identify potential challenges early, while droplet-based integration methods minimised cell stress during sensor installation[2][5]. This integration enhanced process control and ensured traceable, regulatory-compliant data. The result was a seamless data flow from bioreactor sensors to control systems, eliminating the need for frequent manual sampling.
With integration complete, the focus shifted to rolling out the system on a structured timeline.
Deployment Timeline and Milestones
The deployment process spanned 18–24 months, starting with the development and testing of prototype sensors for in-situ measurements. By the third month, the initial prototyping phase was completed. Integration into pilot-scale bioreactors followed, with validation milestones set at three-month intervals[2].
Trends in cultivated meat scale-up and bioprocessing
Results: Measured Improvements in Production
After implementing the system, the facility saw clear advancements in efficiency, traceability, and cost management. By addressing the challenges of scaling cultivated meat, the new monitoring system significantly improved production outcomes.
Improved Process Efficiency and Yield
The introduction of real-time monitoring brought noticeable gains in production by continuously tracking essential culture parameters. This ensured optimal conditions were maintained throughout each batch cycle. By monitoring shear stress thresholds during scale-up, the facility safeguarded the delicate cultivated meat cells from damage caused by fluid forces, resulting in more consistent cell densities and better yields.
Mini bioreactors, ranging from 10 to 500 mL, played a crucial role during the media optimisation phase. Their ability to run parallel experiments accelerated the identification of ideal growth conditions before scaling up. This high-throughput approach reduced the likelihood of errors at the commercial production stage.
Enhanced Data Access and Traceability
Comprehensive data logging created robust GMP batch records and ensured regulatory compliance. This system tracked every step, from filtration to aseptic filling, maintaining consistency across batches and enabling quick troubleshooting when deviations occurred. With industrial-scale bioreactors exceeding 1,000 L, having strong process controls and easily accessible data became even more essential[7]. Beyond compliance, these systems also helped reduce operational costs.
Reduced Labour and Operating Costs
Automation played a key role in cutting down the need for constant manual oversight. AI-driven perfusion systems actively controlled pH, oxygen levels, and shear stress, enabling high-density cell growth while allowing staff to focus on more critical tasks[8]. In August 2024, researchers from the Hebrew University of Jerusalem and Believer Meats demonstrated the effectiveness of continuous manufacturing with tangential flow filtration. Their process operated for 20 days with daily harvests, achieving a cell density of 130 billion cells per litre and a 43% weight-per-volume yield[9].
"Our findings show that continuous manufacturing enables cultivated meat production at a fraction of current costs, without resorting to genetic modification or mega-factories." – Yaakov Nahmias, Founder, Believer Meats[9]
Real-time monitoring also supported the shift from pharmaceutical-grade to more affordable food-grade nutrients. By selecting cell lines capable of thriving in leaner growth media, facilities reduced their reliance on expensive recombinant proteins. This transition, combined with automated assembly-line models inspired by the automotive industry, simplified operations and reduced the labour required to monitor individual batches[9][10].
Lessons Learned and Future Considerations
Solving Technical and Organisational Problems
The deployment of real-time monitoring systems brought to light some unexpected hurdles. One major issue was determining the best sensor placement in turbulent bioreactors during the scale-up phase[6]. Sensors positioned incorrectly often produced unreliable data, pushing teams to create standardised protocols for sensor placement before moving to full-scale operations.
Automating sensor integration proved to be a game-changer, significantly reducing the contamination risks associated with manual sampling[1]. As previously discussed, automated monitoring not only preserves aseptic conditions but also minimises the need for manual intervention. However, the transition to automated data platforms required close collaboration between bioprocess engineers, data scientists, and production managers. Clear protocols became essential for responding to deviations in process parameters[11].
Another critical step was comprehensive staff training. Ensuring that team members understood data interpretation and system calibration was vital for a smooth shift to continuous monitoring. Standard operating procedures (SOPs) for sensor maintenance and data validation replaced outdated manual workflows, creating a more integrated system that allowed for quicker and more informed decision-making.
These lessons highlight the importance of planning scalable monitoring architectures for future operations.
Scaling Monitoring Systems Across Multiple Facilities
Expanding monitoring systems across multiple facilities demands modular designs that can handle increasing production volumes[2]. Facilities that carefully document sensor deployment timelines, integration steps, and troubleshooting methods can build a knowledge base to simplify future installations. A scale-down approach - testing monitoring systems on smaller bioreactors (10 to 500 mL) before full-scale implementation - has proven effective for identifying potential technical issues before they affect commercial production[2].
Consistency is another key factor. Standardising data formats and sensor specifications across facilities ensures the generation of reproducible datasets. This consistency also enables AI-driven analytics to identify performance trends and inform best practices[5]. Establishing baseline metrics is essential for accurately measuring efficiency improvements during scale-up.
How Cellbase Supports Monitoring Technology Adoption
As facilities expand their monitoring systems, having a dependable procurement partner becomes increasingly important. Sourcing specialised sensors, microfluidic devices, and AI-driven analytics tailored to cultivated meat production can be a complex task. Cellbase steps in to connect facilities with trusted suppliers offering equipment designed specifically for bioprocessing needs. Their curated listings simplify the process of identifying tools that integrate smoothly with existing bioreactor systems, reducing deployment challenges and speeding up adoption.
Additionally, technical support plays a crucial role in successful implementation. Cellbase provides access to suppliers who understand the unique requirements of cultivated meat production, such as maintaining sterility and monitoring fluid shear stress. This targeted support helps facilities avoid compatibility issues that could delay deployment. By streamlining the adoption of scalable, real-time monitoring solutions, Cellbase ensures a smoother transition to fully automated systems, enabling facilities to meet the demands of a growing industry.
Conclusion
Real-time monitoring systems play a key role in scaling cultivated meat production from lab experiments to full-scale commercial operations. By continuously collecting data on critical culture parameters, these systems allow for precise control over processes, ensuring consistent product quality and compliance with regulatory standards[1].
The integration of automated monitoring adds another layer of efficiency by reducing the need for manual interventions. This is particularly important for tracking shear stress, which helps safeguard the delicate cultivated meat cells from damage caused by fluid forces[1]. With this level of visibility, production teams can quickly address inefficiencies and maintain detailed batch records required for GMP standards and biosafety inspections[1].
From a regulatory perspective, these systems simplify compliance by automatically generating comprehensive production records for routine inspections, ensuring the safety and integrity of materials[13]. For continuous bioprocessing operations - sometimes spanning 60 days or more - real-time tracking of cell density, metabolites, and potential contaminants is essential to keeping cultures stable and productive[12].
The scalability of these monitoring systems is perhaps their most valuable feature. Modular designs that integrate seamlessly with standard bioreactors and incubation equipment allow facilities to expand monitoring capabilities as production grows, without the need for major infrastructure changes[1]. With the cultivated meat market projected to reach £450 billion by 2050[12], the demand for scalable monitoring solutions will only grow. Meeting these challenges head-on with robust systems is no longer optional - it’s essential.
For teams looking to upgrade their monitoring infrastructure, sourcing reliable sensors, flow controllers, and data logging tools is a critical first step. Cellbase offers a streamlined way to connect with trusted suppliers who understand the technical demands of cultivated meat production, helping to simplify procurement and speed up implementation.
FAQs
Which bioreactor parameters matter most during cultivated meat scale-up?
Key factors to monitor in bioreactors for scaling up cultivated meat include metabolites like lactate and ammonia, biomass density, carbon dioxide (CO₂) levels, glucose, pH levels, and dissolved oxygen. These parameters are essential because they have a direct impact on cell health, growth rates, and the overall feasibility of scaling production processes.
How does real-time monitoring reduce contamination risk without manual sampling?
Real-time monitoring helps keep contamination risks in check by spotting airborne contaminants as soon as they appear. This allows for quick action to address the issue, maintaining sterile conditions without relying on manual sampling. As a result, it not only speeds up the process but also reduces the chance of human error.
What’s the quickest way to source compatible sensors and flow controllers for cultivated meat bioreactors?
The quickest way to source compatible sensors and flow controllers for cultivated meat bioreactors is by using Cellbase. This specialised marketplace is designed specifically for the cultivated meat sector, offering a range of procurement options for real-time monitoring tools, sensors, and bioreactor accessories. It ensures both compatibility and dependability to meet your production requirements.











