For researchers in cultivated meat production, minimising apoptosis is essential to improve cell viability and productivity in bioreactors. Stressors like nutrient depletion, osmotic imbalances, and waste accumulation often trigger cell death, reducing yields. Anti-apoptotic genes can mitigate these challenges by extending the lifespan of cells during culture. Here's a quick overview of the top genes and their roles:
- BCL-2: Prevents mitochondrial pore formation, blocking apoptosis at its initiation. Effective for undifferentiated cells but requires careful balance with pro-apoptotic proteins.
- BCL-xL: Protects cells during differentiation and supports energy metabolism. Ideal for high-stress phases in bioreactors.
- MCL-1: Offers rapid response to nutrient changes and remains stable during differentiation. Works well in combination with other genes.
- BIRC5 (Survivin): Inhibits caspases to block apoptosis downstream. Supports proliferation in rapidly dividing cells.
- XIAP: A potent caspase inhibitor effective under extreme stress conditions, such as high-density cultures. Monitoring these conditions requires selecting sensors for cultivated meat bioreactors to track nutrient levels and waste accumulation in real-time.
Quick Comparison
| Gene | Key Role | Stability During Differentiation | Best Use Case |
|---|---|---|---|
| BCL-2 | Blocks early apoptosis (BAX/BAK) | Stable | Preserving undifferentiated cells |
| BCL-xL | Prevents caspase activation, supports metabolism | Stage-specific | Differentiating cells under stress |
| MCL-1 | Rapid response to nutrient changes | Stable | Multi-stage survival |
| BIRC5 | Inhibits caspases downstream | Decreases with differentiation | Rapidly dividing cells |
| XIAP | Broad caspase inhibition | Stable | High-stress bioreactor conditions |
1. BCL-2
BCL-2 is a well-researched anti-apoptotic gene that plays a key role in the intrinsic (mitochondrial) apoptosis pathway. This pathway is a major mechanism of cell death, often triggered in cultivated meat cells under bioreactor stresses such as nutrient shortages or low oxygen levels.
BCL-2 works by binding to and neutralising pro-apoptotic proteins like BAX and BAK. This action prevents the formation of mitochondrial pores, stopping the release of cytochrome c and halting the downstream cascade of apoptosis. This mechanism is crucial for extending the viable lifespan of cells in cultivated meat production. As Rønning SB et al. explain:
"The ratio between Bcl-2 and Bax determines the susceptibility of the cells to undergo apoptosis."[5]
Beyond its mitochondrial role, BCL-2 also resides in the endoplasmic reticulum (ER). Here, it reduces calcium levels and inhibits IP3 receptor-mediated calcium release, mitigating calcium-induced apoptosis – a frequent issue in high-density bioreactor cultures[4]. Managing these scaling challenges is a primary focus for the industry. This dual localisation enables BCL-2 to protect cells from multiple apoptosis triggers.
The molecular structure of BCL-2, consisting of an eight-alpha-helix bundle and four well-defined BH domains, makes it an excellent candidate for genetic modifications. Techniques such as CRISPR/Cas9-mediated overexpression or stable vector integration can leverage BCL-2's protective capabilities in cultivated meat cell lines[4]. Moreover, because BCL-2 is highly conserved across mammalian species like bovines and pigs, findings from one cell line are often applicable to others used in cultivated meat production[3].
However, there is a critical caveat: the balance between BCL-2 and pro-apoptotic proteins like BAX must be carefully managed. Even high levels of BCL-2 expression can fail to prevent apoptosis if pro-apoptotic signals become too strong[2]. Monitoring this balance is essential for achieving optimal cell viability.
sbb-itb-ffee270
2. BCL-xL
BCL-xL, encoded by the BCL2L1 gene, plays a central role in the BCL-2 family by localising to the outer mitochondrial membrane and preventing apoptosis. It achieves this by counteracting pro-apoptotic proteins like BAX and BAK. Additionally, it inhibits cleaved caspase-3 (CASP3), which is essential for halting cell death. This mechanism is particularly valuable in high-density bioreactor cultures, where metabolic stress can threaten cell viability.
Interestingly, BCL-xL's activity aligns with specific stages of differentiation. During certain phases, its expression increases, while other anti-apoptotic proteins, such as BCL-2 and MCL-1, remain unchanged. This underscores its importance in maintaining cell survival during differentiation. As noted in Cell Death & Disease:
"BCL-xL/BCL2L1 is a critical anti-apoptotic protein that promotes the survival of differentiating... cells." [2]
Beyond its role in apoptosis, BCL-xL supports cellular energy metabolism. It enhances both glycolysis and oxidative phosphorylation, ensuring high metabolic activity. Inhibition of BCL-xL has been shown to reduce the expression of metabolic genes and lower both basal and maximal mitochondrial respiration. This function is particularly important for cultivated meat cells, which rely on sustained metabolic output.
BCL-xL is highly compatible with gene editing strategies commonly used in cultivated meat research. Techniques like lentiviral transduction allow for stable integration of the BCL2L1 gene, while doxycycline-inducible CRISPR/Cas9 systems provide precise temporal control over its expression [2][6]. This level of precision is often managed through advanced bioprocess control software. These attributes make BCL-xL a strong candidate for improving cell line viability in cultivated meat production.
For differentiation stages with high metabolic demands, BCL-xL may be more effective than BCL-2. Researchers can use the inhibitor WEHI-539 to test a cell line's reliance on BCL-xL before proceeding with permanent genetic modifications [2]. Additionally, co-expressing BCL-xL with MCL-1 could further improve cell survival, as these proteins have been observed to work synergistically in some resistant cell types [6].
3. MCL-1
MCL-1 (Myeloid Cell Leukaemia-1) plays a key role in regulating the intrinsic apoptotic pathway. Found on the outer mitochondrial membrane, it prevents apoptosis by binding and sequestering the pro-apoptotic proteins BAX and BAK, stopping their oligomerisation and subsequent membrane permeabilisation. This action blocks the release of cytochrome c, halting the apoptotic cascade before it reaches the execution phase [8]. Additionally, MCL-1 binds BH3-only proteins - such as Bim, PUMA, and NOXA - with high affinity [8]. Like BCL-2 and BCL-xL, MCL-1 is vital for counteracting apoptotic signals, especially during bioreactor stress.
One of MCL-1's unique attributes is its short half-life, making its expression highly responsive to nutrient availability and metabolic signals, particularly through the AMPK/mTOR pathway. Studies indicate that a 25% reduction in caloric intake can decrease MCL-1 translation by approximately 39% ± 10% [7]. This sensitivity is especially relevant for cultivated meat production, where fluctuations in growth media composition or nutrient depletion during large-scale suspension cultures (which require careful production scale planning) can significantly lower MCL-1 levels. Such reductions compromise cell viability, undermining the improvements in IVCC (integral viable cell concentration) achieved through anti-apoptotic strategies. To mitigate this, serum-free media formulations that support robust mTORC1 activity are essential [7].
Another noteworthy feature of MCL-1 is its stability during differentiation. In pancreatic progenitor models, MCL-1 expression remained steady throughout a 17-day differentiation protocol, unlike BCL-xL, which showed stage-dependent variation [2]. This stability makes MCL-1 particularly advantageous for cultivated meat applications, where cells need to survive multiple maturation stages without requiring precisely timed interventions.
Gene editing tools can be used to modify MCL-1, much like other anti-apoptotic genes, making it a versatile target for cell line engineering.
When used in combination with other anti-apoptotic genes, MCL-1 offers additional benefits. For example, pairing MCL-1 with BCL-xL has shown synergistic effects - simultaneous inhibition of both proteins reduced the EC50 of survival drugs from around 10 μM to less than 20 nM [6]. This approach can significantly improve cell survival during the high-stress phases of cultivated meat production.
4. BIRC5 (Survivin)
BIRC5, often referred to as Survivin, is a member of the Inhibitor of Apoptosis (IAP) protein family [2]. Unlike BCL-2 family proteins, which act at the mitochondrial membrane to prevent apoptosis initiation, BIRC5 operates further downstream. It blocks the caspases responsible for executing apoptosis, effectively serving as a final line of defence against programmed cell death [10].
In suspension cultures, stressors like nutrient depletion, metabolic waste build-up, and mechanical shear stress can trigger apoptosis. By inhibiting caspase activity at this later stage, overexpression of BIRC5 helps prolong cell viability and productivity. This results in an improvement in the time integral of viable cell concentration - a key metric for optimising cell culture performance [9]. Eric Baek, a researcher at KAIST, explains:
"Improving the time integral of viable cell concentration by overcoming cell death, namely apoptosis, is one of the most widely used strategies for the efficient production of therapeutic proteins [and cells]." [9]
This downstream intervention has shown to enhance bioreactor yields in cultivated meat cell lines, including porcine satellite cells and bovine myoblasts.
The most effective strategy involves combinatorial engineering, pairing BIRC5 with mitochondrial protectors like BCL-2 or BCL-xL. Professor Michael Betenbaugh from Johns Hopkins University highlights this approach:
"Strategies that block cell death at multiple points along the cascade may limit the amplification of these apoptosis signals." [10]
By combining BIRC5's caspase inhibition with upstream mitochondrial protection, researchers can establish a multi-layered defence against apoptosis.
BIRC5 also integrates seamlessly into gene editing workflows. CRISPR/Cas9 is the leading method for creating stable cell lines with overexpression [9], though zinc-finger nucleases offer a precise alternative. siRNA can be used for pathway validation before committing to genomic integration [9].
5. XIAP
XIAP (X-linked inhibitor of apoptosis) is recognised as the most potent caspase inhibitor within the IAP (inhibitor of apoptosis protein) family. Alongside genes like BCL-2 and MCL-1, XIAP plays a critical role in targeting apoptosis at its execution phase. As highlighted in Genes & Development:
"XIAP is considered the most potent caspase inhibitor in vitro." [12]
XIAP employs two distinct mechanisms to inhibit apoptosis. First, its BIR2 domain and linker region block effector caspases-3 and -7. Second, its BIR3 domain inhibits caspase-9, effectively halting the intrinsic mitochondrial apoptotic pathway. Additionally, its C-terminal RING domain facilitates the ubiquitination and subsequent proteasomal degradation of target caspases [11]. By intervening in both intrinsic and extrinsic apoptotic pathways, XIAP proves highly effective in addressing apoptosis triggers like nutrient shortages, metabolic by-products, and mechanical stress - factors commonly encountered in cultivated meat production systems. Its functionality is further enhanced by its strong conservation across species.
For instance, human XIAP shares 87.7% protein identity with Bos taurus (bovine) and 89.5% with Mus musculus (mouse) [11]. This high similarity allows research from mammalian model systems to be reliably applied to cell lines used in cultivated meat production.
XIAP can be regulated using tools such as shRNA, antisense oligonucleotides, or CRISPR/Cas9 [11]. Under extreme stress, its RING domain may induce self-ubiquitination [12], while endogenous inhibitors like SMAC/DIABLO and HTRA2 can displace XIAP from caspases [11][13]. These findings make XIAP an attractive target for gene editing approaches aimed at optimising cell lines for cultivated meat development.
Comparing Anti-Apoptotic Genes at a Glance
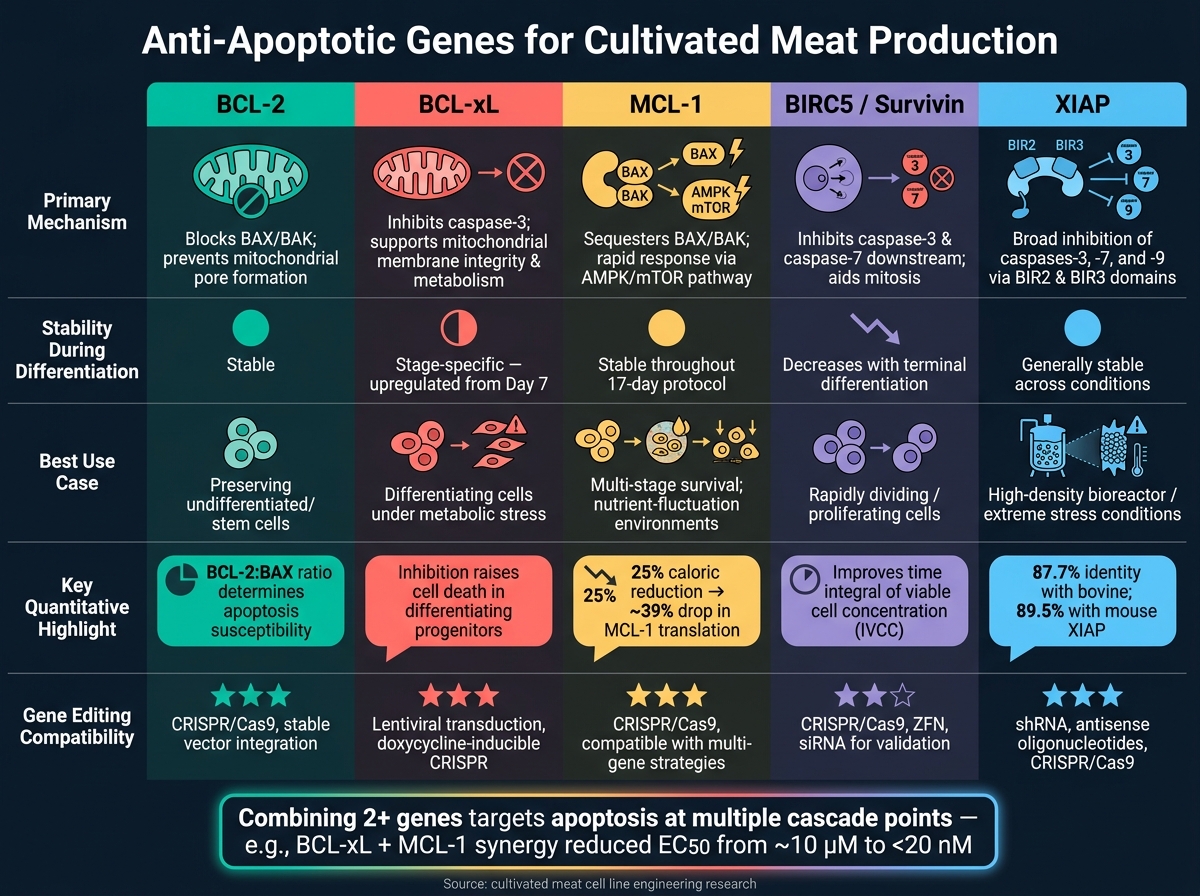
Anti-Apoptotic Genes for Cultivated Meat: Side-by-Side Comparison
When working on cultivated meat production, understanding how different anti-apoptotic genes function can help fine-tune cell line engineering. Each gene has its distinct mechanism, behaviour during differentiation, and potential applications. The table below summarises these differences, making it easier to decide which gene - or combination of genes - might work best for your needs.
| Gene | Primary Mechanism | Expression Stability | Reported Viability Impact | Editing Compatibility |
|---|---|---|---|---|
| BCL-2 | Blocks pro-apoptotic BAX/BAK and ensures survival of undifferentiated cells [2] | Stays relatively stable during differentiation [2] | Essential for preserving the initial stem cell pool [2] | High compatibility with editing tools |
| BCL-xL | Inhibits cleaved caspase-3; maintains mitochondrial membrane integrity and metabolism [2] | Upregulated starting Day 7 of differentiation [2] | Critical for supporting differentiating progenitors; its inhibition increases cell death [2] | High compatibility with editing tools |
| MCL-1 | Modulates pro-apoptotic signals as part of the BCL-2 family [2] | Expression remains steady during lineage specification [2] | Offers broad survival benefits but lacks stage-specific effects like BCL-xL [2] | High compatibility with editing tools |
| BIRC5 (Survivin) | Blocks caspase-3 and caspase-7; aids chromosomal segregation during mitosis | High in proliferating cells; decreases with terminal differentiation | Supports survival and proliferation in rapidly dividing cells | Compatible with both shRNA knockdown and CRISPR editing |
| XIAP | Inhibits multiple caspases, providing broad apoptotic protection | Generally stable across various conditions | Particularly effective under stress, such as high-density bioreactor conditions | High compatibility with editing tools |
BCL-xL stands out for its dual role in promoting cell survival and supporting metabolic activity, particularly during the critical differentiation phase when pro-apoptotic proteins like BAK naturally decline. BCL-2, on the other hand, is ideal for preserving undifferentiated cells, while XIAP delivers broad protection, especially in stressful environments like high-density cultures.
No single gene works best in every scenario. For instance, BIRC5 is particularly useful in situations requiring rapid cell division. In practice, combining two or more genes often offers the most effective protection, addressing a variety of apoptotic triggers simultaneously.
These findings provide a foundation for incorporating these genes into cell line engineering strategies for cultivated meat production. This includes selecting the right cultivated meat inputs to ensure scalability.
Using These Genes in Cultivated Meat Cell Line Engineering
To improve cell viability in cultivated meat production, integrating key genes strategically is crucial. It’s not enough to identify anti-apoptotic genes - their effective incorporation into cell lines is what makes the difference. Two main strategies are commonly employed: overexpressing anti-apoptotic genes like BCL-2, BCL-xL, and MCL-1 to enhance cell survival, or knocking out pro-apoptotic genes such as BAX, BAK, and BOK to eliminate the drivers of cell death. Combining these approaches often results in cell lines that are better suited for large-scale production [1].
Modern gene-editing tools like CRISPR/Cas9 allow for simultaneous edits, such as knocking out Bak1, Bax, and Bok in one step. Alternatives like ZFNs or RNA interference can be used to temporarily reduce the activity of caspases (e.g. caspases-3, -7, -8, and -9). For overexpression strategies, synthetic promoters ensure consistent and high expression levels of genes like BCL-2 during scale-up, which is critical for maintaining cell performance in fed-batch or continuous culture systems. These combined methods strengthen cell line development for cultivated meat applications.
Such genetic modifications directly impact improved integral viable cell concentration (IVCC), a key metric in cultivated meat production. Cell death is most pronounced during the first five days of differentiation, making early interventions with genes like BCL-2 or BCL-xL essential. Research published in Cell Death & Disease highlights that BCL-xL expression increases as cells differentiate, indicating that more mature progenitors rely heavily on its protective role [2]. By monitoring the expression levels of BCL-2 family genes throughout growth phases, interventions can be precisely timed for maximum effect.
"By establishing stable cell lines that overexpress antiapoptotic genes or downregulate proapoptotic genes, the final product yields can be enhanced as cells become more resistant to environmental stresses." - Gyun Min Lee et al. [1]
For bioreactor-based production, cells must also be engineered to withstand hyperosmotic stress and nutrient deprivation. Before scaling up, it’s essential to validate genetic edits using tools like Western blot or FACS. For researchers seeking specialised cell lines or genetic materials tailored to high-density bioreactor environments, platforms like Cellbase provide a marketplace of verified suppliers, simplifying the procurement process for cultivated meat R&D.
Conclusion
Selecting anti-apoptotic genes for cultivated meat cell lines requires a tailored approach. Genes like BCL-2, BCL-xL, and MCL-1 each play unique roles in protecting cells, but their success depends on factors like cell type, developmental stage, and the specific stresses encountered during production. As highlighted in research:
"the balance between the anti-apoptotic and pro-apoptotic members ultimately determines whether a cell lives or dies" [2]
Beyond survival, anti-apoptotic engineering also preserves metabolic functions. For instance, proteins such as BCL-xL are closely tied to maintaining glycolysis and oxidative phosphorylation. However, poorly executed interventions can disrupt these critical processes [2]. Ensuring that engineered cell lines maintain their intended identity and metabolic activity throughout production is a crucial, though sometimes overlooked, step. These insights are shaping the future of cell line engineering.
New multi-gene approaches are emerging, which combine overexpression of protective genes with CRISPR knockouts of pro-apoptotic genes like BAX, BAK1, and BOK to create more robust cell lines for industrial use [1]. Tools for metabolic profiling, such as bioenergetics assays, are becoming essential to confirm that these genetic modifications enhance overall cell performance. For researchers sourcing porcine cell lines, genetic materials, or bioreactor equipment, Cellbase offers a dedicated marketplace connecting cultivated meat researchers with verified suppliers critical for implementing these advanced techniques.
FAQs
Which anti-apoptotic gene should I start with for my cell line?
BCL-2 is often suggested as a starting point when working with cell lines. This well-researched anti-apoptotic gene is recognised for its ability to improve cell survival, making it a popular option in cultivated meat research. Its function in supporting cell viability makes it a practical choice for early-stage experiments.
Is it better to overexpress anti-apoptotic genes or knock out pro-apoptotic ones?
In cultivated meat production, increasing the expression of anti-apoptotic genes, such as members of the BCL-2 family like BCL-xL, tends to yield better results than disabling pro-apoptotic genes. This strategy supports both cell survival and proliferation - key factors for scaling production - while preserving the cell's natural regulatory systems.
By boosting anti-apoptotic gene activity, cells gain greater resistance to apoptosis, especially under stressful conditions. This makes it a more dependable and safer approach for maintaining cell viability during the cultivation process.
How can I confirm an anti-apoptotic edit improves IVCC in my bioreactor?
To determine whether an anti-apoptotic gene edit enhances in vitro cell viability and proliferation (IVCC), you’ll need a systematic approach:
- Assess viability and proliferation rates: Use methods like cell counting or flow cytometry to measure these rates both before and after the gene edit.
- Verify gene expression: Techniques such as qPCR or Western blotting can confirm the successful expression of the targeted gene.
- Monitor apoptosis markers: Check for markers like caspase activity to ensure the edit effectively reduces apoptosis.
For a complete evaluation, it’s critical to test the long-term stability and proliferation of the edited cells in a bioreactor. This ensures the improvements persist across multiple culture cycles.











