The way fluids move in scaffold-based bioreactors is a game-changer for cultivated meat production. Proper flow ensures cells get enough nutrients and oxygen while removing waste, especially for thick tissue structures. Here's why it matters:
- Diffusion limits: Nutrients only penetrate 100–200 μm by diffusion, leaving inner cells starved.
- Perfusion bioreactors: These systems actively push culture medium through scaffolds, improving nutrient delivery and waste removal.
- Shear stress trade-offs: Controlled flow stimulates growth, but excessive shear can harm cells.
Key factors include perfusion rates, scaffold design (pore size, porosity), and computational models to predict flow behaviour. Advanced bioreactors and tools, like those available through Cellbase, play a vital role in scaling up cultivated meat production with consistent quality.
Read on for insights into flow control, scaffold design, and how computational tools are shaping this field.
Perfusion Bioreactor Modeling Using ANSYS Fluent - Part 1
Perfusion Rates and Shear Stress Explained
Optimal Shear Stress Ranges and Flow Parameters for Scaffold-Based Bioreactors
How Perfusion Rates Affect Cell Growth
Perfusion rates are crucial for controlling how nutrients are delivered and waste is removed through medium flow. If the flow is too low, cells are deprived of essential nutrients. On the other hand, excessive flow can physically harm the cells. The key is striking the right balance to maximise nutrient exchange without causing damage.
Studies show that perfusion cultures can lead to more than double the cell proliferation compared to static cultures over two weeks [4]. In some cases, the difference is even more striking. For example, in spherical scaffolds, cell volume increased fourfold compared to cubical scaffolds after three weeks of perfusion [7]. This isn’t just about increasing the flow rate - it’s about creating the right mechanical conditions for growth.
"The mixing and fluid shear stress induced by perfusion will improve development by mechanically stimulating the cells, allowing them to differentiate into the desired cell type." – SN Applied Sciences [4]
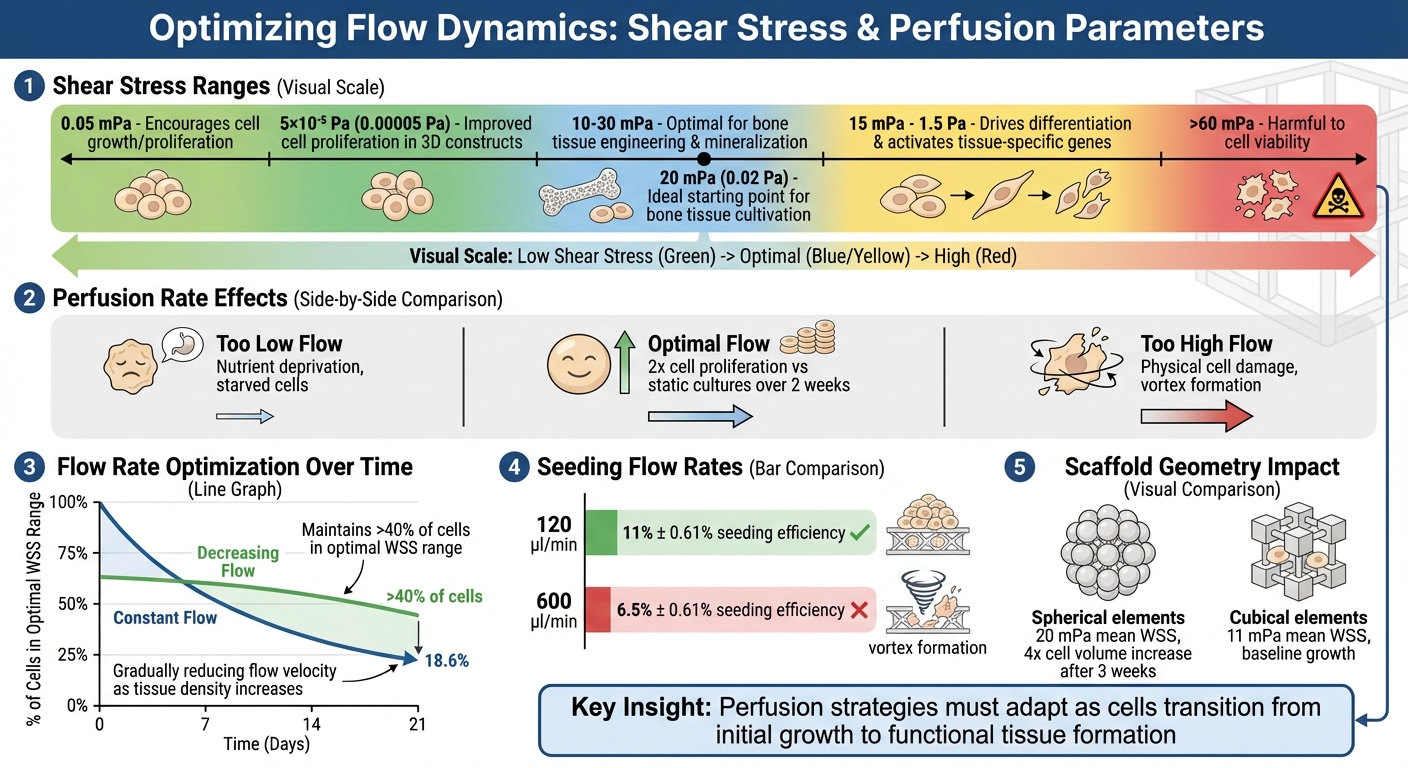
Shear stress also plays a critical role. Low levels (~0.05 mPa) encourage cell growth, while higher levels (15 mPa–1.5 Pa) drive differentiation and activate tissue-specific genes [2][8]. This means perfusion strategies need to adapt as cells move from initial growth to forming functional tissue. The next section dives into how to manage shear stress effectively to protect cell viability.
Controlling Shear Stress to Maintain Cell Viability
Wall shear stress (WSS) is a double-edged sword. For bone tissue engineering, the ideal range is between 10–30 mPa, which supports mineralisation. However, exceeding 60 mPa can harm cell viability [5]. As cell density rises, scaffold porosity decreases, which can restrict flow paths and lead to localised spikes in shear stress if flow rates stay constant.
One way to address this is by gradually reducing flow velocity as tissue density increases. For instance, constant flow conditions reduce the percentage of cells exposed to optimal WSS from 50% to 18.6% over 21 days. In contrast, lowering the flow rate over time keeps optimal conditions for over 40% of cells [5]. During the seeding phase, precise calibration is essential; a flow rate of 120 µl/min is ideal, while higher rates like 600 µl/min can create vortices, preventing proper scaffold attachment [3].
Scaffold geometry also has a major impact. The way flow interacts with the scaffold structure needs to align with its architecture to maintain cell health and support tissue growth. For example, under the same flow conditions, spherical scaffold elements produce a mean WSS of 20 mPa, compared to 11 mPa in cubical elements [7]. This highlights how the right scaffold design, combined with careful flow control, is essential for optimising outcomes.
Bioreactor Design for Flow Control
Scaffold Porosity and Flow Channel Design
The structure of a scaffold plays a critical role in managing fluid flow and cell distribution. Key factors like pore size, porosity percentage, and pore arrangement directly influence how fluid moves and the shear forces acting on cells [1]. Essentially, the pores' size and layout determine the flow velocity and how shear stress is distributed across the scaffold.
"Under the applied perfusion conditions, cell deposition is mainly determined by local wall shear stress, which, in turn, is strongly influenced by the architecture of the pore network of the scaffold." – Biomaterials Journal [1]
Scaffold designs are typically either isotropic or gradient. Isotropic scaffolds have uniform pore sizes - about 412 μm with 62% porosity - resulting in steady shear rates ranging from 15 to 24 s⁻¹. In contrast, gradient scaffolds feature varying pore sizes (250–500 μm) and porosity levels (35%–85%), creating a broader shear range of 12–38 s⁻¹ [1]. This gradient design encourages cells to accumulate in specific zones, while isotropic scaffolds ensure an even distribution throughout the structure.
As cells grow and occupy the scaffold's void spaces, they reduce its porosity, altering fluid dynamics. Denser scaffolds demand higher pressure to maintain flow, which risks generating excessive shear stress. For effective tissue ingrowth, a pore radius of approximately 100 μm is crucial [2][6]. However, the ideal pore size varies depending on the type of tissue being cultivated. These factors are essential for designing bioreactors that effectively manage flow.
Bioreactor Types and Flow Control Methods
Perfusion bioreactors are excellent at evenly delivering nutrients while applying controlled shear stress. By directing medium through the scaffold, they support the development of thicker tissues [2].
Packed-bed reactors, on the other hand, are designed for high-volume operations but face challenges with uneven radial porosity. This can lead to "channelling", where fluid bypasses certain areas, disrupting uniform distribution. For example, in November 2017, researchers tested 3D Biotek's commercial PCL scaffolds (5 mm diameter, 1.5 mm height). They found that a flow rate of 120 μl/min resulted in a seeding efficiency of 11% ± 0.61%. However, at 600 μl/min, efficiency dropped to 6.5% ± 0.61% due to vortex formation, which trapped cells in recirculation zones instead of allowing them to attach to the scaffold fibres [3]. This highlights how critical flow control is for achieving consistent cell seeding.
Different systems employ distinct methods for managing flow. Perfusion bioreactors focus on directing flow through the scaffold, while hollow-fibre systems regulate both the lumen inlet flow and outlet back pressure to simulate nutrient delivery akin to capillaries [9]. Advanced systems incorporate sensors and monitors to maintain stable conditions [8]. Additionally, to avoid air bubbles - which can harm cells or disrupt flow - placing the medium reservoir above the culture chamber utilises hydrostatic pressure to good effect [8].
sbb-itb-ffee270
Using Computational Models to Predict Flow Behaviour
Benefits of CFD in Bioreactor Design
Computational fluid dynamics (CFD) models are powerful tools for predicting how fluids move through scaffold structures. By solving the Navier-Stokes equations, these models provide insights into shear stress and nutrient distribution - without the need for physical prototypes. This not only cuts down on development costs but also eliminates the risk of contamination that can occur during repeated experimental trials [11][3][10].
Scaffold geometries can be designed using CAD for standard shapes or μCT imaging for more intricate structures [2][10]. Back in March 2005, researchers used the Lattice-Boltzmann method with μCT imaging at a 34 μm voxel resolution to simulate how media flows through cylindrical scaffolds. Their model showed that an average surface shear stress of 5×10⁻⁵ Pa was linked to improved cell proliferation [2].
CFD also helps predict how flow patterns evolve as cells grow and fill the void spaces within scaffolds. For example, in November 2021, a study utilised COMSOL Multiphysics to simulate fluid flow through hierarchical 3DP/TIPS scaffolds. By modelling 38 inlet channels in a 10-mm-diameter scaffold, researchers fine-tuned the peristaltic pump speed to achieve a wall shear stress of 20 mPa, ideal for murine preosteoblastic cells [4]. These models can even incorporate complex factors like cell growth kinetics and oxygen consumption rates using Michaelis-Menten equations. This allows designers to anticipate how tissue development will impact fluid dynamics over time [11][12].
"CFD can help in reducing the cost, time, and risk of contamination inherent to the experiments required." – Future Foods Mini-Review [11]
These predictive capabilities also pave the way for integrating sensor feedback to dynamically adjust flow conditions.
Real-Time Monitoring with Sensors
Pairing sensors with computational models takes bioreactor design a step further by enabling real-time adjustments to maintain optimal conditions. For instance, in December 2025, researchers tested the BioAxFlow bioreactor using COMSOL Multiphysics 6.3 to simulate oxygen distribution and fluid velocity. They applied a cell-normalised oxygen consumption rate of 2 nmol min⁻¹ 10⁻⁶ cells for SAOS-2 cells on PLA scaffolds. The results showed that the chamber’s geometry supported even cell distribution without needing mechanical impellers [13].
Advanced systems can now adjust flow rates based on monitored oxygen levels, ensuring that even the centre of the scaffold remains properly oxygenated [13]. However, one challenge persists: measuring local shear stress within scaffolds. As X. Yan from the University of Saskatchewan highlights: "Due to the lack of adequate sensors, it is difficult, even impossible, to measure the local shear stress distribution within a scaffold" [10]. This limitation underscores the value of CFD modelling, which can provide detailed predictions that physical sensors currently cannot achieve.
Applying Flow Dynamics to Cultivated Meat Production
Improving Tissue Quality Through Flow Control
Using controlled flow dynamics can significantly improve the quality of cultivated meat by ensuring uniform cell distribution throughout the scaffold. One of the major issues with static cultures is that cell growth often concentrates around the edges of the scaffold, leaving the centre underdeveloped. Flow dynamics solve this by improving mass transport, allowing oxygen and nutrients to reach the scaffold's core while efficiently removing waste. This balance is essential for producing high-quality, structurally sound cultivated meat products.
Shear stress plays a critical role here. For example, studies show that an average surface shear stress of 5×10⁻⁵ Pa encourages cell proliferation in 3D constructs. In comparison, scaffolds designed for bone tissue often aim for around 20 mPa (0.02 Pa) at the start of cultivation to provide mechanical stimulation [2][4]. However, as cells fill the scaffold's pores, the flow channels narrow, naturally increasing shear stress even if pump speed remains constant [4].
"The observed heterogeneity in matrix synthesis is believed to be a result of inadequate distribution of nutrients and removal of waste products within the constructs." – Robert Guldberg [2]
The effectiveness of initial cell seeding also highlights how flow dynamics influence tissue outcomes. Research using PCL scaffolds found that a flow rate of 120 μl/min was ideal for seeding, while higher rates, like 600 μl/min, reduced efficiency due to vortex formation, which trapped cells in recirculation zones [3]. Achieving even initial cell distribution is crucial for ensuring the final product's quality. These findings emphasise the importance of using equipment capable of meeting precise flow requirements.
Sourcing Equipment Through Cellbase
Achieving precise flow control and optimising tissue quality require access to specialised equipment. This is where Cellbase steps in as a dedicated B2B marketplace, connecting researchers and production teams with suppliers who understand the technical needs of cultivated meat production.
Through Cellbase, teams can source scaffolds with tailored architectures, such as those combining 3D-plotting for macrochannels with thermally-induced phase separation (TIPS) for micropores. These designs enhance nutrient diffusion and cell migration [4]. The marketplace also features a range of equipment, including syringe pumps for low-volume perfusion (12–600 μl/min) and peristaltic pumps for larger-scale operations [3][4].
For those scaling up production, Cellbase offers bioreactor options suited to different flow characteristics. These include stirred-tank bioreactors for high-density cell expansion, wave/rocking bioreactors designed for shear-sensitive stem cells (capable of maintaining shear stress as low as 0.01 Pa), and hollow fibre bioreactors with inner radii between 300 and 400 μm, optimised for dense cell growth [11]. By simplifying procurement and ensuring compatibility, Cellbase helps production teams stay ahead in a market where global meat consumption is projected to grow by 14% by 2030 [11].
Conclusion
Managing flow dynamics in scaffold-based bioreactors is essential for producing high-quality cultivated meat. Success depends on effectively controlling perfusion rates and shear stress throughout the cultivation process. Static cultures fall short of supporting the thick, uniform tissue structures needed for commercial-scale production. Cells located more than 100–200 μm from the surface often fail to receive sufficient nutrients and oxygen, highlighting the importance of advanced flow management in bioreactor design [4].
When flow parameters are optimised, perfusion bioreactors can more than double cell proliferation compared to static cultures [4]. Adjusting perfusion and shear stress is particularly important for achieving consistent tissue growth. For instance, research conducted at the University of Sheffield in April 2020 found that gradually reducing fluid flow over time, instead of maintaining a constant rate, significantly improved outcomes. After 21 days, 40.9% of the cell surface stayed within the optimal shear stress range, compared to just 18.6% under constant flow conditions [5]. This single change can greatly enhance both tissue quality and production efficiency.
"For achieving more mineralised tissue, the conventional manner of loading the perfusion bioreactors (i.e. constant flow rate/velocity) should be changed to a decreasing flow over time." – F. Zhao et al. [5]
Striking the right balance between mass transport and mechanical stimulation is crucial. Insufficient flow leaves inner cells deprived, while excessive flow risks dislodging them [10][3]. Computational Fluid Dynamics (CFD) modelling plays a key role in predicting local flow conditions and optimising bioreactor performance [2][10].
Scaling up production also presents equipment challenges. From scaffolds with hierarchical structures to bioreactors with precise flow control, sourcing the right tools is vital. Cellbase helps cultivated meat companies overcome this hurdle by connecting them with verified suppliers, ensuring that cutting-edge flow dynamics research translates into commercial success.
FAQs
How do I choose a safe perfusion rate for my scaffold?
Balancing the perfusion rate is key to ensuring successful cell attachment and scaffold performance while avoiding potential damage. Starting with moderate flow rates is often a sensible approach. From there, monitor cell viability and scaffold integrity closely as you make gradual adjustments. Using computational models or experimental data tailored to your specific scaffold design can provide valuable insights. This helps fine-tune the perfusion rate to support optimal cell growth and nutrient transport, all while minimising the risk of shear stress damage.
How can I avoid shear-stress damage as tissue thickens?
To reduce the risk of shear-stress damage as tissue thickens, it’s important to gradually lower the perfusion flow rate during cultivation. This adjustment helps keep wall shear stress (WSS) in the ideal range of 10–30 mPa, which protects cells from excessive strain while still promoting mineralisation. Computational studies back this method, showing it can significantly minimise the amount of tissue exposed to high shear stress, helping to safeguard the developing tissue from harm.
What should CFD modelling include for realistic flow predictions?
CFD modelling needs to incorporate the scaffold's microstructure, ensure precise fluid flow simulation, and provide a detailed analysis of shear stress. Additionally, experimental data validation is crucial to ensure the predictions align with real-world conditions. Together, these factors contribute to a deeper understanding of flow dynamics within scaffold-based bioreactors.











