Shear stress can make or break cultivated meat production. Why? Because the cells used lack protective walls, making them prone to damage from fluid forces in bioreactors. This article dives into how shear stress affects these cells, the thresholds they can handle, and ways to design systems that protect them.
Key takeaways:
- Shear stress arises from fluid movement and can harm fragile animal cells, causing membrane damage, detachment, or death.
- Most mammalian cells tolerate 0.3–1.7 Pascals, but even lower levels can activate stress responses.
- Design choices like impeller type, aeration methods, and bioreactor geometry directly impact shear forces.
- Strategies to minimise damage include using gentler bioreactor designs (e.g., airlift or rocking systems), optimising agitation speeds, and adding protective agents like Pluronic F68.
For cultivated meat, managing this balance is critical to ensure cells grow and differentiate without harm, especially as production scales up. Let's explore the science behind these thresholds and practical solutions for bioreactor design.
110: Spinning Like Earth: Designing Low-Shear Bioreactors for Better Cell Culture with Olivier De...
What Affects Shear Stress in Bioreactors
Understanding the factors that influence shear stress in bioreactors is crucial for optimising conditions, especially when delicate cells are involved. Let’s dive into the main elements that shape its intensity and distribution.
Bioreactor Design and Operating Conditions
The design of a bioreactor plays a major role in determining where and how shear stress occurs. One key factor is the type of impeller used. For instance, Rushton turbines can create energy dissipation rates up to 280 times higher than the vessel's average, while high-efficiency axial flow impellers like the HE3 produce rates closer to 180 times the mean dissipation [4]. Other design elements, such as impeller diameter, speed, and positioning, also influence energy distribution.
Interestingly, aeration introduces much harsher forces than agitation. When small bubbles (1–2 mm) rupture, they release energy levels between 10⁷–10⁹ W/m³, which can kill over 1,000 cells in a single event [4]. This makes bubble behaviour a critical consideration, especially in cultivated meat production.
Baffles are another key design element. They prevent the formation of a vortex in the culture, which would otherwise pull bubbles into the liquid and increase rupture events at the surface [4]. Additionally, the impeller-to-vessel diameter ratio and the impeller's height from the bottom influence how energy spreads throughout the bioreactor.
Uneven Distribution of Shear Stress
Shear stress isn’t evenly distributed across the bioreactor. Research shows that energy dissipation tends to concentrate around specific zones, such as the impeller discharge area, trailing vortices, and the liquid surface where bubbles break. These hotspots can pose challenges during scale-up.
Weiwei Hu from Biogen Idec highlights this scaling issue:
The perception of 'shear sensitivity' has historically put an arbitrary upper limit on agitation and aeration in bioreactor operation; however, as cell densities and productivities continue to increase, mass transfer requirements can exceed those imposed by these arbitrary low limits [4].
For example, a 2021 study by Junxuan Zhang and Xueliang Li from Jiangnan University compared a 250 mL spinner flask with a 20 m³ stirred tank reactor using computational fluid dynamics. They observed that even at the lowest stirring speeds, shear forces in the larger reactor were strong enough to detach cells from microcarriers, with sparging introducing even more stress than agitation [3].
Culture Format and Shear Sensitivity
The culture format also determines how cells experience shear stress. Cells grown on microcarriers are particularly vulnerable. If intense mixing or collisions between carriers cause cells to detach, those cells are effectively lost [4]. On the other hand, suspension cultures of hybridoma cells have demonstrated resilience, maintaining viability at agitation speeds as high as 1,500 RPM in baffled bioreactors without an air-liquid interface [4].
Different culture systems handle shear in various ways. Fixed-bed bioreactors minimise shear by keeping cells immobilised on stationary surfaces, while fluidised beds introduce moderate to high shear through the movement of microcarriers and upward fluid flow [2]. Some microcarriers, especially porous ones, offer internal surfaces that can shield cells from extreme forces, providing better protection compared to solid microcarriers [2]. These differences highlight the need to carefully balance nutrient delivery with the risk of cell damage when designing bioreactors.
Shear Stress Thresholds for Different Cell Types
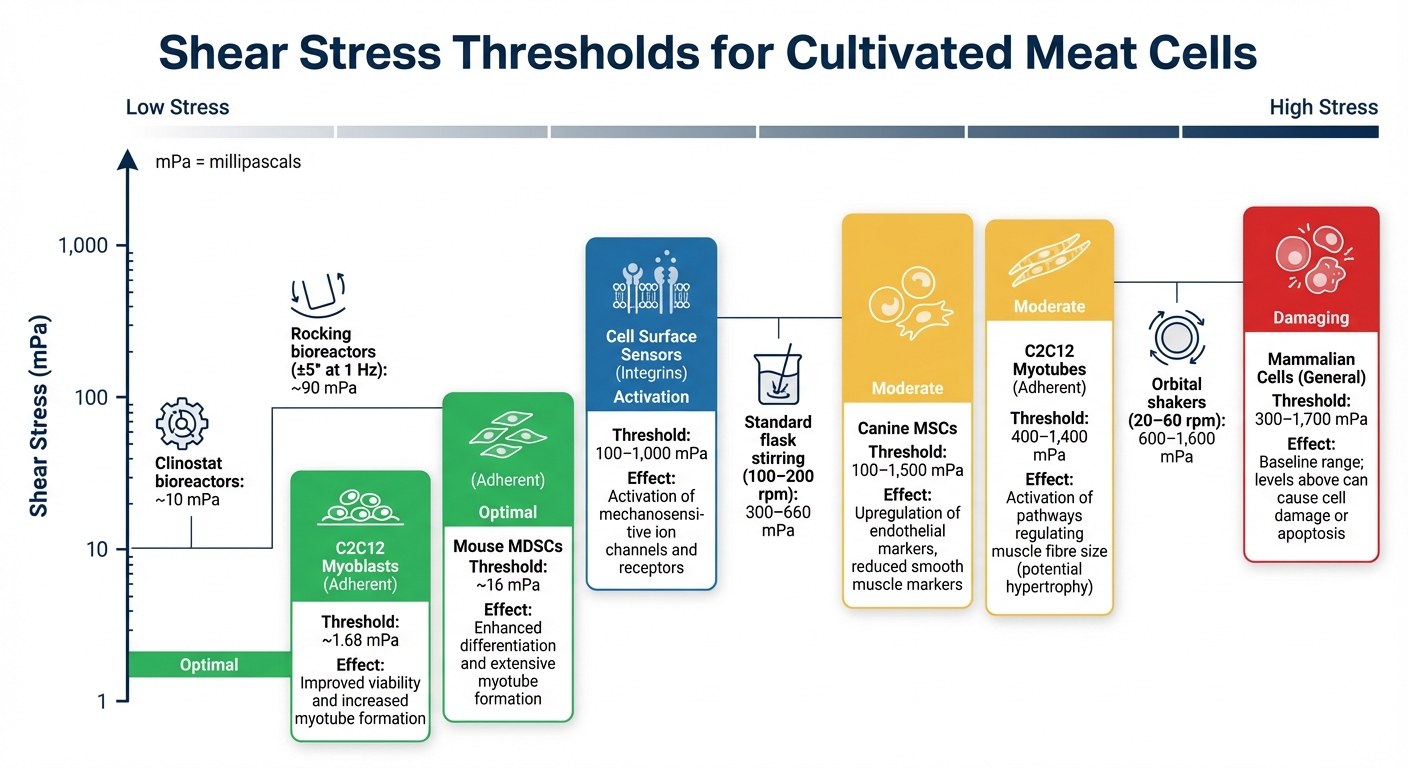
Shear Stress Tolerance Thresholds for Cultivated Meat Cell Types
Managing shear stress is critical for cultivated meat production, as uneven stress can harm cells that lack strong cell walls. Understanding the specific stress levels each cell type can tolerate helps maintain cell health, trigger mechanosensitive responses, or encourage differentiation.
Threshold Values for Common Cell Types
Shear stress tolerance varies significantly among cell types, and knowing these thresholds is key to fine-tuning bioreactor settings.
For instance, cultivated meat myoblasts like the C2C12 line thrive under low shear stress. Cyclic stress of about 1.68 mPa improves myotube formation and fusion [8]. Mouse muscle-derived stem cells (MDSCs) show better myogenic differentiation and more extensive myotube formation when exposed to 16 mPa [8]. As myoblasts mature into myotubes, they can handle higher stress levels; pulsed stress between 400 mPa and 1,400 mPa activates pathways that regulate muscle fibre size, potentially leading to hypertrophy [8].
Mesenchymal stem cells (MSCs) also respond uniquely. For example, canine MSCs exposed to shear stress between 100 mPa and 1,500 mPa upregulate endothelial markers like PECAM-1 and VE-cadherin while downregulating smooth muscle markers [10].
Shear Stress Threshold Comparison Table
Here’s a quick comparison of shear stress thresholds across different cultivated meat cell types:
| Cell Type | Shear Stress Threshold (mPa) | Observed Effects | Source |
|---|---|---|---|
| Mammalian Cells (General) | 300–1,700 | Baseline range; levels above this can lead to cell damage or apoptosis | [1] |
| C2C12 Myoblasts (Adherent) | ~1.68 | Improved viability and increased myotube formation | [8] |
| Mouse MDSCs (Adherent) | ~16 | Enhanced differentiation and extensive myotube formation | [8] |
| C2C12 Myotubes (Adherent) | 400–1,400 | Activation of pathways regulating muscle fibre size (potential hypertrophy) | [8] |
| Canine MSCs | 100–1,500 | Upregulation of endothelial markers, reduced smooth muscle markers | [10] |
| Cell Surface Sensors (Integrins) | 100–1,000 | Activation of mechanosensitive ion channels and receptors | [1] |
For context, stirring a culture at 100–200 rpm in a standard flask generates shear stress levels of 300–660 mPa, while orbital shakers running at 20–60 rpm produce higher forces ranging from 600 mPa to 1,600 mPa [1]. Gentler systems like rocking bioreactors (±5° at 1 Hz) create stress of about 90 mPa [9], and clinostat bioreactors operate at around 10 mPa, staying well below the activation threshold for mechanosensitive cell surface sensors [1].
These thresholds serve as a guide for adjusting bioreactor conditions, helping to maintain optimal environments during scale-up and cell growth phases.
sbb-itb-ffee270
How to Reduce Shear Stress Damage
Minimising shear stress damage in cultivated meat production is all about achieving a delicate balance. The aim is to ensure efficient mixing and oxygen delivery while protecting sensitive cells from mechanical harm. This involves a combination of smart bioreactor design and thoughtful operational strategies.
Bioreactor Design Modifications
Using CFD (Computational Fluid Dynamics) modelling is a key step in optimising bioreactor performance. Modern CFD techniques now include multiphase flow simulations, which account for the interactions between cells and microcarriers. This results in more accurate assessments of shear stress and its potential damage [5].
The type of bioreactor plays a major role in determining shear stress levels. While stirred-tank reactors are still widely used, alternative designs can offer gentler conditions:
- Airlift bioreactors: These eliminate mechanical agitators, instead using gas-induced circulation to reduce mechanical shear [5].
- Wave or rocking bioreactors: By relying on surface motion instead of impellers, these are ideal for low-to-medium density cultures that require gentle mixing [5].
- Vertical-wheel bioreactors: Particularly effective for aggregate-based cultures, these have shown success in maintaining cell viability during the expansion of human iPSC aggregates [11].
Another important factor is the non-Newtonian behaviour of cell suspensions. For example, suspensions containing serum exhibit shear-thinning properties, which traditional models often fail to capture. Using advanced models, like the Sisko model, provides more accurate predictions of shear stress, helping to fine-tune mechanical forces and avoid thresholds that could alter genetic expression [6].
Cell Seeding and Agitation Methods
Operational strategies also play a big part in reducing shear stress damage. For instance, intermittent agitation during the early stages of cell attachment can limit shear exposure while still ensuring nutrients are distributed effectively. Adjusting agitation requires careful consideration of factors like serum content, cell density, and the age of the culture [6].
When determining agitation speeds, CFD modelling can help identify the ideal balance - enough oxygen transfer without causing mechanical harm. Compartmentalised simulations can further refine shear stress distribution, making the process more efficient [5].
Impact on Bioreactor Design and Scale-Up
When scaling up bioreactors for cultivated meat production, understanding and applying shear stress thresholds is critical. These thresholds influence decisions on impeller speed, sparger design, and other parameters to ensure cell viability as production volumes grow.
Setting Bioreactor Operating Parameters
Shear stress thresholds play a key role in defining operational limits. For example, haematopoietic stem cells (HSCs) have a threshold of approximately 0.092 Pa[12]. Staying below this level - such as operating at 50 rpm, which generates about 0.068 Pa - supports healthy cell expansion, achieving a 27.4‐fold increase. However, raising the agitation to 100 rpm increases shear stress to around 0.192 Pa, resulting in a 72% apoptosis rate and limiting expansion to 24.5‐fold[12].
"The threshold shear stress for HSCs proliferation and function has been reported 0.092 Pa." – Hosseinizand et al. [12]
Shear damage occurs when turbulent eddies become smaller than about two-thirds of a cell or aggregate’s diameter[12][13]. At 50 rpm, eddies measure around 280 µm, which is safe for cells. But at 100 rpm, eddies shrink to 166 µm, increasing the risk of mechanical damage.
Sparging introduces additional hydrodynamic stress. Small bubbles (1 mm diameter) generate local fluid velocities of about 6.4 m/s during rupture, while larger 6 mm bubbles produce gentler peaks of 0.94 m/s[13]. To counteract this, additives like Pluronic F68 are used to prevent cells from sticking to bubble surfaces. However, their effectiveness relies on maintaining the right concentration relative to the gas surface area[13].
These parameters are essential when transitioning to larger bioreactor systems.
Maintaining Conditions During Scale-Up
Scaling up from a 250 mL spinner flask to a 20 m³ stirred tank reactor introduces unique challenges. Hydrodynamic conditions in small-scale systems don’t directly translate to industrial volumes. Even operating large reactors at minimal stirring speeds can result in shear forces strong enough to detach cells from microcarriers[3].
"Even when operated at a stirring speed close to the Njs, the shear exerted by the impellers alone could cause cell detachment from microcarriers, while even more hydrodynamic stress is introduced via sparging." – Zhang et al. [3]
To maintain consistent shear conditions during scale-up, one approach is to keep the impeller tip velocity constant. However, this can lead to longer mixing times and the formation of nutrient and oxygen gradients, which may negatively affect cell growth and performance[3]. Computational Fluid Dynamics (CFD) modelling becomes essential for identifying stress zones and optimising reactor design during scale-up[5].
For cell lines that are highly sensitive to shear, alternative reactor designs are often more suitable. Airlift reactors, which eliminate mechanical agitators, have been successfully modelled for volumes up to 300,000 L, achieving theoretical cell densities of 2 × 10⁸ cells/mL[7]. Similarly, rocking bioreactors use gentle wave motions to minimise shear, making them effective for seed trains up to 500 L[14][15]. Platforms like Cellbase provide access to suppliers specialising in low-shear designs tailored for cultivated meat production.
Summary and Recommendations
Managing shear stress effectively is crucial for maintaining cell viability and productivity in cultivated meat production. Research shows that bubble rupture during aeration creates more damaging forces than mechanical stirring. For example, small bubbles (1 mm) generate fluid velocities of 6.4 m/s upon rupture, while larger bubbles (6 mm) produce gentler peaks of 0.94 m/s [13]. To minimise these forces, procurement teams should focus on bioreactors equipped with sintered microspargers (15-μm pore size), which allow for pulsed aeration and reduce the gas-liquid interface. These considerations are vital for scaling up bioreactor systems.
Another important factor is the ratio of eddy scale to cell diameter (η/d_c), which can help reduce damage caused by stirring. A study conducted in August 2017 by the Institute of Bioprocess Engineering and Pharmaceutical Technology highlights this. Using a 3-L Applikon glass bioreactor with Sf21 insect cells, they showed that a six-blade Rushton impeller at 205 rpm, combined with 199 μm bubbles, produced a GFP protein yield of 12.75 μg/mL. In contrast, a pitched blade impeller at 171 rpm, which generated a higher specific gas surface area of 18.0 m²/m³, yielded only 4.0 μg/mL [13]. This demonstrates that total gas surface area is more influential than stirring speed.
Protective agents such as Pluronic F68 (0.5–3 g/L) can form a 16–40 μm protective layer around bubbles, preventing cells from attaching [13]. However, as Tobias Weidner and colleagues observed:
If the [total gas] surface area exceeds a certain threshold, the concentration of Pluronic is no longer sufficient for cell protection [13].
This means engineers must carefully monitor the gas surface area in relation to Pluronic F68 concentration during scale-up to ensure cells remain protected.
For sensitive cell lines, alternative reactor designs can provide solutions. Airlift reactors, for instance, eliminate mechanical agitators, creating a gentler mixing environment [7]. Fixed-bed bioreactors are another option, capable of maintaining ultra-low wall shear stresses ranging from 10⁻³ to 10⁻² Pa [17]. For teams exploring specialised low-shear systems, suppliers like Cellbase offer expertise in cultivated meat bioprocessing.
Additionally, maintaining bovine myoblasts below 25 population doublings is essential to preserve their differentiation capacity [16]. Exceeding this threshold can lead to a decrease in the fusion index by approximately 6.81% with each passage [16], reducing the cells' ability to form muscle fibres. To address this, process engineers should utilise Computational Fluid Dynamics (CFD) modelling to identify high-shear zones before scaling up from laboratory to industrial systems. This approach ensures smoother transitions and better outcomes during scale-up.
FAQs
How do I measure shear stress in my bioreactor?
Shear stress in bioreactors is often assessed using computational modelling techniques like Computational Fluid Dynamics (CFD). These methods allow for the analysis of flow patterns and the identification of shear zones within the bioreactor. Additionally, small-scale shear testing tools are valuable for characterising how sensitive specific cell lines are and for evaluating various process conditions. For continuous monitoring, shear stress can be determined by calculating fluid velocity and viscosity. This approach is particularly effective in microfluidic systems or by utilising online shear stress calculators.
Which aeration method minimises bubble-rupture damage?
Minimising bubble-rupture damage relies heavily on using smaller bubbles. These bubbles cause less cell damage when compared on a volume-to-volume basis. Although exact techniques aren't outlined, managing bubble size and behaviour - like regulating their size - plays a crucial role in reducing the harmful effects of rupture.
What should I keep constant when scaling up to reduce shear?
When increasing the size of cultivated meat bioreactors, it's crucial to keep shear stress under about 3 Pa to prevent harming the cells. Pay close attention to factors like agitation, flow patterns, and aeration to ensure shear levels stay consistent throughout the operation.











