Producing cultivated meat is expensive, with growth media being the largest cost driver. Metabolomics, a detailed analysis of cellular metabolism, replaces guesswork with precise data to refine media composition. This approach identifies nutrient deficiencies, tracks how cells use resources, and highlights waste buildup that hinders growth.
Key findings:
- 40.72% increase in cell density achieved in a 2019 study by optimising media for chicken fibroblasts.
- Metabolomics tools pinpointed critical nutrients like glucose, amino acids, and energy-related compounds needed for efficient cell growth.
- Adjustments in nutrient levels (e.g., creatine, inosine-5'-monophosphate) improved cell proliferation while reducing waste.
Spent media analysis to facilitate cultivated meat media optimization - Ted O'Neill - ISCCM9
Initial Growth Media Problems
The research team faced major hurdles with the original media formulation for C2C12 muscle cells. The standard DMEM/F12 medium simply couldn't sustain the cell densities or yields required for large-scale cultivated meat production. The cells consumed nutrients far faster than the media could replenish, leading to early depletion of critical components and poor biomass growth. To address these issues, the team turned to data-driven strategies for optimisation.
Nutrient Deficiencies in the Original Formulation
An analysis of the spent media revealed some glaring nutrient shortages. Glucose and specific amino acids were being used up at an unsustainable pace. To produce just 1 kg of C2C12 muscle cells, the cells required between 1,100–1,500 g of glucose and 250–275 g of amino acids[2]. Among these, glutamine, glycine, and cystine were in particularly high demand, which restricted cell growth and proliferation.
The metabolic profile also exposed inefficiencies in how nutrients were processed. For instance, energy-related metabolites like creatine and inosine-5'-monophosphate were downregulated, while metabolites involved in membrane synthesis - such as phosphoethanolamine and choline - were upregulated[3]. This shift indicated that the cells were prioritising immediate energy consumption over energy storage. Even when nutrients were available, their proportions were far from optimal for efficient biomass production. This imbalance made it clear that a more precise and analytical approach was needed.
Why Metabolomics Was Selected
Traditional trial-and-error methods could have taken months of testing to pinpoint these specific issues. Instead, the team opted for metabolomics, a technique that identifies and measures metabolites in spent media with remarkable precision. This method provided a detailed snapshot of cellular metabolism in a single analysis[2].
"Previous data from metabolic studies performed using serum-containing media may not directly translate to serum-free systems." – ScienceDirect[2]
Metabolomics proved invaluable for detecting subtle biochemical changes, especially as the team worked to develop serum-free formulations. While standard growth assessments - like cell counts or viability tests - could only offer surface-level insights, metabolomics revealed the deeper metabolic needs of the cells. This allowed the team to refine the media composition based on actual data rather than assumptions, paving the way for more targeted and effective improvements.
Results from Metabolomic Analysis
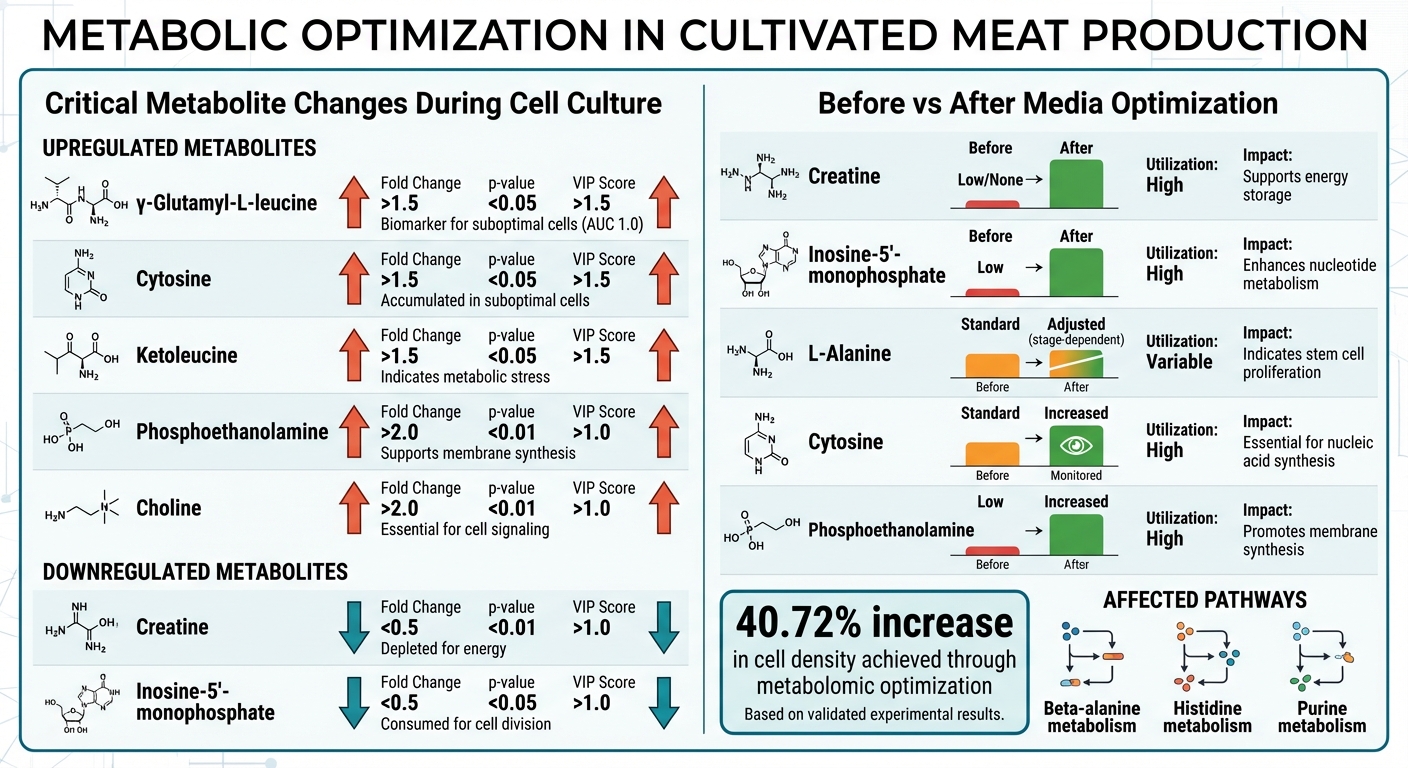
Metabolite Changes and Nutrient Optimization in Cultivated Meat Production
Metabolite Changes During Cell Culture
A detailed metabolomic analysis identified seven critical metabolites that showed notable changes during pig muscle stem cell culture. In April 2024, a team led by Doo Yeon Jung at Seoul National University pinpointed γ‑glutamyl‑L‑leucine, cytosine, and ketoleucine as key biomarkers for recognising suboptimal cell states [5]. These three metabolites achieved an AUC of 1.0, demonstrating perfect accuracy in predicting declines in cell proliferation [5].
The study also uncovered shifts in energy management within the cells. Metabolites like phosphoethanolamine and choline were significantly upregulated, reflecting the heightened demand for membrane synthesis during rapid cell division [6]. On the other hand, creatine and inosine-5′-monophosphate were downregulated, indicating a shift away from energy storage towards immediate energy consumption [6]. These findings provide a strong foundation for further examination of metabolic pathways.
Metabolic Pathway Analysis
The pathway analysis revealed increased activity in three key systems: beta-alanine metabolism, histidine metabolism, and purine metabolism [5][6]. Each of these pathways plays a vital role - protein synthesis, pH buffering, and DNA/RNA production, respectively. Among these, the histidine pathway stood out, showing consistent activity during both the proliferating and differentiating stages. This suggests it may be a limiting factor in the original media formulation [6].
The purine metabolism pathway offered additional insights. A significant depletion of nucleotide-related compounds indicated that cells were using these building blocks faster than they could be replenished by the culture media. This was further supported by the accumulation of waste metabolites like cytosine in later passages, coinciding with reduced cell growth [5].
Metabolite Comparison Table
| Metabolite Name | Fold Change | p-value | VIP Score | Status |
|---|---|---|---|---|
| γ‑Glutamyl‑L‑leucine | > 1.5 | < 0.05 | > 1.5 | Upregulated (accumulated in suboptimal cells) [5] |
| Cytosine | > 1.5 | < 0.05 | > 1.5 | Upregulated (accumulated in suboptimal cells) [5] |
| Ketoleucine | > 1.5 | < 0.05 | > 1.5 | Upregulated (accumulated in suboptimal cells) [5] |
| Phosphoethanolamine | > 2.0 | < 0.01 | > 1.0 | Upregulated (supports membrane synthesis) [6] |
| Choline | > 2.0 | < 0.01 | > 1.0 | Upregulated (essential for cell signalling) [6] |
| Creatine | < 0.5 | < 0.01 | > 1.0 | Downregulated (depleted for energy) [6] |
| Inosine-5′-monophosphate | < 0.5 | < 0.05 | > 1.0 | Downregulated (consumed for cell division) [6] |
Growth Media Adjustments
Changes to Nutrient Concentrations
Researchers at Seoul National University, led by Doo Yeon Jung, used metabolomic analysis to fine-tune the growth media for cultured meat production. By examining spent media, they identified which nutrients were depleted during cultivation and which waste products were building up [5]. This allowed them to adjust nutrient levels to better match cellular needs.
The team focused on three main factors: nutrients that cells consumed quickly, waste products that indicated metabolic stress, and the cost of ingredients (aiming to replace expensive components without sacrificing performance) [7]. For example, L-alanine levels were modified depending on the stage of cell growth, while creatine and inosine‑5′‑monophosphate were increased to support a shift from energy storage to direct energy use.
"Monitoring the levels of these key metabolites in culture media could serve as a quality control measure for cultured meat production by enabling the indirect detection of suboptimal PSCs." - Doo Yeon Jung, Researcher, Seoul National University [5]
Phosphoethanolamine levels were boosted to aid membrane synthesis during cell division, while cytosine concentrations were carefully monitored to avoid excessive build-up [5][6]. These adjustments aimed to create a metabolic balance where nutrients were efficiently converted into biomass, reducing waste and improving the feed conversion ratio [7].
The table below highlights the key changes made to the nutrient concentrations and their impact on cell growth.
Before and After Comparison
| Nutrient | Initial Concentration | Optimised Concentration | Utilisation Rate | Impact on Cell Growth |
|---|---|---|---|---|
| Creatine | Low/None | Increased | High | Supports energy storage; aligns with conventional meat properties [6] |
| Inosine‑5′‑monophosphate | Low | Increased | High | Enhances nucleotide metabolism and energy production [6] |
| L-Alanine | Standard | Adjusted (stage-dependent) | Variable | Indicates stem cell proliferation capacity [5] |
| Cytosine | Standard | Increased/Monitored | High | Essential for nucleic acid synthesis during rapid cell division [5] |
| Phosphoethanolamine | Low | Increased | High | Promotes membrane synthesis and cell structure integrity [6] |
These refinements addressed specific metabolic challenges, particularly in purine, histidine, and sphingolipid metabolism [6]. By tailoring nutrient availability to match cellular consumption, the team reduced waste and achieved more consistent cell proliferation across multiple growth cycles.
sbb-itb-ffee270
Results: Improved Cultivation Performance
Cell Growth and Biomass Improvements
The metabolomic approach brought clear gains in cell performance. A 2025 study from Texas A&M University highlighted this by testing two serum-free formulations: LM7 (chemically defined) and LM8 (chemically undefined, containing mung bean protein isolate). Impressively, the LM8 formulation matched the performance of 20% FBS - a rare achievement in muscle cell culture [8]. This marked a big step forward, as most serum-free media struggle to even match 10% FBS performance.
Further studies using C2C12 cells showed that optimising nutrient ratios not only reduced waste but also improved biomass conversion [2] [7]. Similar benefits were observed in lamb, C2C12, and pig muscle cell studies, demonstrating how widely applicable this metabolomic-driven media optimisation can be.
Scaling these findings was validated in 3D microcarrier systems, where LM8 showed superior performance in shaking flask systems using CellBIND microcarriers [8]. Additionally, research on pig muscle stem cells in April 2024 found that cells at passage 2 (PSC2) had the highest growth rates. In contrast, cells at passage 3 (PSC3) showed a significant loss of myogenic marker genes, making PSC2 a reliable quality control benchmark for scaling production [5]. These advancements not only confirm the effectiveness of the metabolomics approach but also open the door to significant cost savings.
Production Scale and Cost Benefits
These improvements translated into substantial cost reductions. Since media costs often account for over 60% of production expenses, cutting out high-cost animal components and fine-tuning nutrient delivery made a meaningful impact [8].
Beyond cost, these advances strengthen the environmental promise of cultivated meat. With global meat demand expected to rise by around 70% by 2050 [8], cultivated meat offers a way to reduce land and water use by up to 90% compared to conventional livestock farming [8]. By ensuring nutrients are directed efficiently towards biomass production, the metabolomic approach helps maintain this environmental edge while avoiding waste caused by metabolic inefficiencies.
How Cellbase Supports Media Optimisation

Metabolomics-based media optimisation requires specialised tools and materials, which can be challenging to source. Cellbase steps in as a dedicated B2B marketplace tailored for the cultivated meat industry, simplifying the procurement process for metabolomic workflows [4]. These workflows rely on precise tools to carry out the detailed analyses discussed earlier, and Cellbase helps researchers access these resources efficiently.
The platform categorises its offerings to meet specific needs:
- Growth Media & Supplements: Supplies high-quality, serum-free formulations.
- Lab Equipment & Instrumentation: Features metabolomics tools and analytical equipment for spent media analysis.
- Sensors & Monitoring: Provides tools to track nutrient utilisation rates, which is essential given that producing 1 kg of C2C12 cells consumes around 250–275 g of amino acids and 1,100–1,500 g of glucose [2].
What sets Cellbase apart is its exclusive focus on cultivated meat production. This ensures that all tools available are validated for use in serum-free systems, where nutrient usage patterns differ significantly from those in serum-containing media [2]. The platform also simplifies purchasing with transparent pricing and an easy checkout process, reducing administrative burdens [4].
In addition to providing equipment, Cellbase offers expert technical support. Through its "Ask us anything" service, researchers can consult "Cell Ag Experts" for guidance on tackling optimisation challenges [4]. This is particularly useful when transitioning to serum-free media or selecting tools to monitor metabolic changes.
Conclusion
Metabolomics plays a key role in refining growth media for cultivated meat production. By pinpointing metabolic bottlenecks and nutrient gaps, researchers can make targeted adjustments that significantly enhance cell performance. For example, a study from East China University of Science and Technology showed how comparative metabolomic analysis led to notable increases in cell density and virus production [1].
Using insights from metabolomics, spent media analysis moves beyond guesswork. This precision allows scientists to create media formulations that maximise cell proliferation while cutting down on waste and costs.
The advantages span various aspects of production. Metabolomics aids quality control through biomarkers like γ-glutamyl-L-leucine and ketoleucine [5]. It also facilitates the transition from costly, undefined serum-based formulations to affordable, serum-free options - critical for scaling up production. As highlighted by the Good Food Institute:
"Cell culture media is currently the largest cost and environmental impact driver of cultivated meat production" [7].
These advancements underscore the potential of data-driven media optimisation to transform the field.
FAQs
What is metabolomics in growth media optimisation?
Metabolomics plays a key role in optimising growth media by analysing the metabolic profiles of cells used in cultivated meat production. By understanding how these cells utilise nutrients and their metabolic pathways, researchers can design serum-free media that is both more efficient and cost-effective, specifically tailored to the needs of cultivated meat production.
What metabolites are the best early indicators of poor growth?
Key metabolites associated with poor growth in cultivated meat include γ-glutamyl-L-leucine, cytosine, and ketoleucine. These biomarkers serve as indicators of underperforming primary cells and highlight metabolic shifts that may affect cell proliferation.
How is spent media data used to reduce media costs?
Spent media analysis plays a key role in reducing costs in cultivated meat production. By pinpointing nutrients that are either depleted or in excess, it helps refine media formulations for better efficiency. Tools like spectroscopy enable real-time monitoring, cutting down on waste and preventing the overuse of costly components. Additionally, metabolomics provides valuable insights that can support recycling or reusing media, further driving down expenses. This targeted approach ensures resources are used wisely while still supporting robust, high-quality cell growth.











