When designing GMP-compliant facilities for cultivated meat production, the choice between single-use and multi-use equipment affects costs, scalability, operations, and regulatory compliance. Here’s the key takeaway:
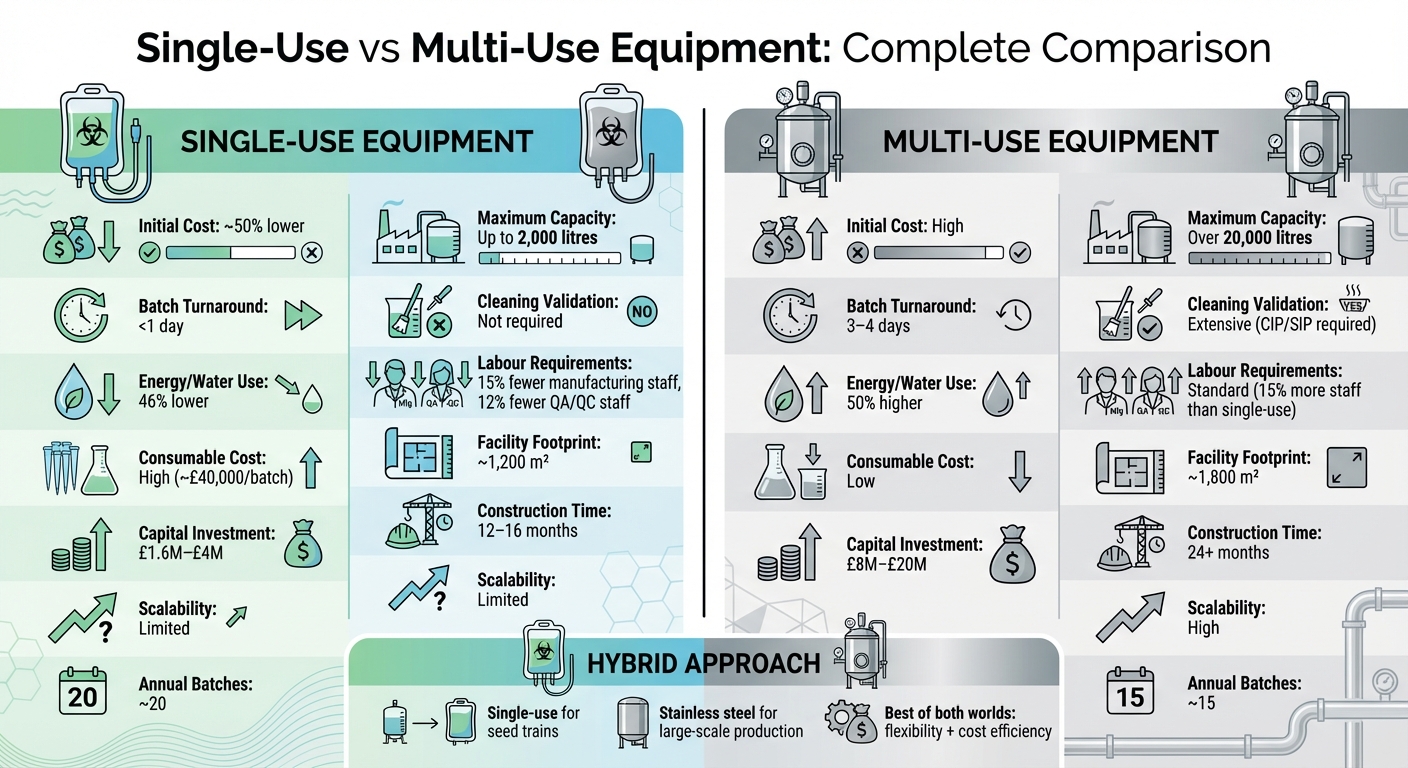
- Single-Use Equipment: Lower upfront costs (up to 50% less), quicker batch turnaround, no cleaning validation, and reduced water/energy use. However, it’s limited to 2,000-litre capacity, has higher recurring costs for consumables (~£40,000/batch), and faces supply chain challenges.
- Multi-Use Equipment: Higher initial investment, but scalable to over 20,000 litres, with lower long-term production costs. It requires complex cleaning protocols (CIP/SIP), more energy, and larger facility footprints.
Quick Comparison
| Feature | Single-Use Equipment | Multi-Use Equipment |
|---|---|---|
| Initial Cost | ~50% lower | High |
| Capacity | Up to 2,000 litres | Over 20,000 litres |
| Turnaround Time | <1 day | 3–4 days |
| Cleaning Validation | Not required | Extensive (CIP/SIP) |
| Energy/Water Use | ~50% lower | High |
| Consumable Cost | High (~£40,000/batch) | Low |
| Scalability | Limited | High |
A hybrid approach - using single-use systems for seed trains and stainless steel for large-scale production - offers a balanced solution for flexibility and cost management. Facility design, cleanroom classifications, and utility needs depend heavily on this choice. Platforms like Cellbase simplify sourcing GMP-compliant equipment for both systems.
Single-Use vs Multi-Use Equipment Comparison for GMP Cultivated Meat Facilities
Single-Use Equipment: Benefits and Drawbacks
Benefits of Single-Use Equipment
Single-use systems offer a major advantage in contamination control. By disposing of product-contact surfaces after each batch, they minimise cross-contamination risks. This approach also reduces the need for high-level cleanroom classifications and complex segregation protocols [1].
Turnaround times are another strong point. While stainless steel systems can take three to four days to prepare, single-use systems are ready in less than a day [5]. For instance, HIPRA S.A., a Spanish animal health company, tested 200-litre stirred single-use bioreactors in 2014. They found that eliminating cleaning cycles saved them two months of setup time and increased productivity by 15–20% [3].
Pre-sterilised single-use equipment also simplifies operations by cutting down on cleaning validation requirements. This efficiency translates to staffing reductions of about 15% in manufacturing and 12% in QA/QC departments [4][5].
Utility consumption is significantly lower with single-use systems. Bruce Rawlings and Hélène Pora from Pall Life Sciences highlight that:
"a single‐use facility is about 50% less energy intensive due to significantly lower consumption of energy otherwise required to heat large volumes of water for cleaning and sterilising stainless steel equipment"
[5]. Overall, single-use facilities can reduce total water and energy use by 46% [1]. When factoring in savings on labour, maintenance, and utilities, operating costs per batch can be about 22% lower [5].
| Feature | Single-Use Equipment | Multi-Use Equipment |
|---|---|---|
| Initial Capital Investment | ~50% lower [1] | High |
| Batch Turnaround Time | <1 day [5] | 3–4 days [5] |
| Cleaning Validation | Not required [5] | Extensive (CIP/SIP) [5] |
| Water/Energy Usage | 46% lower [1] | High |
| Labour Requirements | 15% fewer manufacturing staff [5] | Standard |
However, these benefits are balanced by certain limitations that impact scalability and operational costs.
Drawbacks of Single-Use Equipment
While single-use systems offer operational perks, they come with notable challenges. One major issue is scalability. Single-use bioreactors are often limited in size, requiring a scale-out strategy. In contrast, stainless steel systems can scale up to 20,000 litres or more, making them more suitable for large-scale operations [1][2]. This limitation forces facilities to rely on multiple smaller units rather than a single large vessel.
Another drawback is the cost of consumables, which can increase operating expenses by approximately £40,000 per batch [5]. This makes single-use systems less cost-effective for high-volume production.
Supply chain reliability is also a concern. The industry has faced a "single-use supply crunch", with lead times for some consumables stretching beyond a year. Around 50% of biomanufacturing survey respondents have reported experiencing such delays [1]. Additionally, single-use bags are prone to damage from sharp objects, overpressure, or mishandling during installation [3].
The issue of extractables and leachables further complicates matters. Chemicals from plastic components can leach into the product, potentially affecting cell viability or productivity. Cheryl Scott, Editor in Chief of BioProcess International, cautions:
"Extractables and leachables could compromise cell viability or productivity and even persist throughout purification and drug‐product formulation to present a risk to patients"
[2]. This shifts the focus from cleaning validation to rigorous testing and material characterisation.
Lastly, single-use systems require additional storage space for their consumables. Managing bulky inventory can complicate facility layouts, especially for companies running multiple production lines [1].
Multi-Use Equipment: Benefits and Drawbacks
Benefits of Multi-Use Equipment
Stainless steel bioreactors are a reliable choice for large-scale cultivated meat production, offering well-documented performance that has stood the test of time. Unlike single-use systems, which are limited in capacity, multi-use equipment can scale up to over 20,000 litres, making it ideal for mass production [2][3].
These stainless steel vessels are built to last for decades, providing a permanent infrastructure for long-term GMP (Good Manufacturing Practice) operations [2]. One major advantage is their ability to eliminate the risk of bag integrity failures, a common concern with disposable systems that can lead to biosafety issues and financial losses [3]. Additionally, multi-use systems are highly automated, reducing the need for manual interventions like bag installation and aseptic connections, which are more common in single-use setups [3].
Another cost-saving aspect is the avoidance of recurring consumable expenses. Multi-use systems also allow for faster end-of-production emptying through sterile air pressure, a more efficient process compared to the methods required for single-use bags [3].
| Feature | Multi-Use (Stainless Steel) | Single-Use (Disposable) |
|---|---|---|
| Maximum Scale | >20,000 L | Typically 2,000 L |
| Automation Level | High | Low (More manual work) |
| Risk of Leaks | Minimal | Moderate (Bag failures) |
| Consumable Cost | Low | High (+£40,000/batch) [5] |
| Equipment Lifespan | Decades | Single batch |
Jordi Ruano Bou, Biologics Production Director at HIPRA S.A., explains: "Large-scale MUBs are time-proven technologies around the world. Although they have demonstrated process efficiency, a company must consider many aspects when acquiring one" [3].
Still, while multi-use systems excel in many areas, they come with their own set of challenges.
Drawbacks of Multi-Use Equipment
Despite their strengths, multi-use systems are not without drawbacks, particularly in terms of costs and operational complexity. They require a much higher upfront capital investment, with procurement timelines often exceeding a year. Additionally, the qualification process for these systems can be lengthy, delaying production readiness [3].
Cleaning and sterilisation are another hurdle. Stainless steel bioreactors demand rigorous Clean-in-Place (CIP) and Steam-in-Place (SIP) procedures, which can take 3 to 4 days between batches [5]. This extended downtime limits the number of batches that can be produced annually - around 15 compared to 20 with single-use systems [5].
The physical space required for multi-use equipment is also significantly larger. A traditional stainless steel facility typically needs about 1,800 m², compared to 1,200 m² for single-use systems - a 50% increase driven by extensive piping, utility systems, and cleaning infrastructure [5]. This larger footprint not only impacts facility design but also increases energy consumption. Multi-use systems are estimated to use 50% more energy, largely due to the need to heat large volumes of water for sterilisation [5].
Labour demands are higher as well. Multi-use facilities require approximately 15% more manufacturing staff and 12% more QA/QC personnel to handle cleaning validation and equipment maintenance [5]. Maintaining GMP standards adds further complexity, as cleaning validation involves constant monitoring and rigorous quality assurance efforts [3][5].
How Equipment Choice Affects Facility Design
Facility Layout Requirements
Choosing between single-use and multi-use equipment significantly impacts the design of a GMP facility. Stainless steel systems require an extensive fixed infrastructure, including permanent piping for Clean-in-Place (CIP), Steam-in-Place (SIP), and Water for Injection (WFI) systems [2][8]. This setup creates a rigid facility layout, as equipment placement is dictated by the piping network.
In contrast, single-use technology offers a more flexible approach. Without the need for hard-piped utilities, facilities can adopt adaptable "ballroom" or "dancefloor" layouts. Here, equipment is mobile, and the space is designed around process needs rather than fixed infrastructure [2]. Additionally, single-use systems often allow for lower cleanroom classifications, such as Grade C instead of Grade B, due to their closed processes. This change can lead to reduced HVAC requirements and lower energy consumption [8].
"Single-use facilities can operate with remarkably minimal fixed infrastructure", notes Connected Workplaces [8].
However, the trade-offs aren’t entirely straightforward. While single-use facilities eliminate the need for bulky piping and utility rooms, they require more storage space for consumables like bags, filters, and tubing [10]. For cultivated meat production, a single-use facility typically occupies around 1,200 m², compared to 1,800 m² for stainless steel - a 33% reduction. But this space saving is partially offset by the increased storage demands [5].
The differences extend to capital investment and construction timelines. Single-use systems generally cost between £1.6 million and £4 million to set up, with construction taking 12–16 months. In comparison, multi-use facilities require £8 million to £20 million and over 24 months to complete [8][7][9]. For example, in 2012, Catalent Pharma Solutions transitioned to a GMP-compliant facility in just one year by adopting single-use technology, replacing stainless steel reactors to handle nine products simultaneously [7]. Similarly, AGC Biologics completed a facility start-up in under 16 months at its Yokohama site by installing Cytiva FlexFactory suites for mRNA vaccine production [9]. This adaptability has also opened the door to hybrid designs that combine the strengths of both systems.
Hybrid Equipment Approaches
Many cultivated meat facilities are now opting for hybrid setups, blending single-use and multi-use equipment to balance flexibility and cost. A common strategy involves using single-use systems for seed trains and media preparation while relying on stainless steel for large-scale production bioreactors [2][7]. This approach combines the quick changeover capabilities of disposables for smaller volumes with the cost-efficiency of stainless steel for large-scale operations.
William Hartzel of Catalent Pharma Solutions explains: "Generally speaking, single-use facilities are more flexible than traditional facilities, a major advantage in a multiproduct facility" [7].
Designing hybrid facilities requires careful planning to integrate both types of equipment. Ceiling-mounted utility panels, for instance, enable easy reconfiguration of floor layouts, allowing facilities to switch between single-use skids and fixed equipment as needed [1]. This modular design provides "future-proofing", enabling quick modifications to accommodate evolving products or processes without major reconstruction [1].
Adding single-use operations to existing stainless steel setups can also optimise resource use. Single-use processes can run during downtime for stainless steel equipment, sharing utilities like water supply and reducing infrastructure costs [10]. A notable example is BioInno, a China-based CDMO, which installed 6,000-litre single-use bioreactors alongside its existing infrastructure. This approach provided multi-product flexibility and broke the traditional 2,000-litre size limit for disposable systems [9].
Facility Design Comparison Table
| Design Element | Single-Use | Multi-Use (Stainless Steel) | Hybrid |
|---|---|---|---|
| Fixed Piping | Minimal; flexible tubing | Extensive network required | Mixed infrastructure |
| Cleanroom Grade | Grade C (typically) | Grade B (typically) | Varies by area |
| Facility Footprint | ~1,200 m² [5] | ~1,800 m² [5] | Intermediate |
| Capital Investment | £1.6M–£4M [8] | £8M–£20M [8] | Intermediate |
| Construction Time | 12–16 months [7][9] | 24+ months | 18–24 months |
| Storage Requirements | High (consumables) | Low | Moderate |
| Utility Demand | 50% lower energy [5] | High water/steam needs | Mixed |
| Layout Flexibility | High; mobile equipment | Low; fixed positions | Moderate |
sbb-itb-ffee270
Procurement Strategies for GMP-Grade Equipment
Selecting GMP-Grade Equipment Suppliers
When choosing suppliers for GMP-compliant equipment, it’s crucial to look beyond just the price tag. The primary focus should be on the supplier’s qualification and validation packages. These documents need to confirm that the equipment aligns with regulatory standards - whether it’s FDA, EMA, or local food-grade requirements. They should also cover essential factors like extractables, leachables, and sterility validation to ensure the equipment consistently adheres to GMP standards for cultivated meat production [10].
"Check the robustness of the qualification and validation package supplied, and make sure it meets all relevant regulatory requirements", advises John Joseph, Engineering Project Leader at GE Healthcare [10].
Transparency in the supply chain is another key consideration. Suppliers must provide clear and proactive updates about any changes to raw materials or components. Consistent sourcing of materials helps avoid costly re-validation processes triggered by unexpected component changes. Current supply chain challenges further highlight the importance of planning for longer procurement timelines [1].
To manage costs, cultivated meat producers can evaluate where food-grade standards can replace pharmaceutical-grade specifications without compromising safety or compliance. For example, 304 stainless steel might be an acceptable alternative to the pricier 316 grade in certain applications, and GRAS-certified water could be used in place of pharmaceutical-grade water in some cases [6]. However, these decisions require a thorough risk assessment to ensure they meet all safety and regulatory standards.
Meeting these rigorous criteria has led to the development of specialised platforms designed to simplify GMP equipment sourcing.
Using Cellbase for Equipment Procurement
Cellbase offers a solution to the challenges of sourcing GMP equipment for cultivated meat facilities. This specialised B2B marketplace focuses exclusively on the cultivated meat industry, connecting buyers with verified suppliers who provide bioreactors, single-use consumables, stainless steel systems, and other essential equipment tailored to industry needs.
Each listing on Cellbase includes detailed information on GMP compliance, scale, and material certifications. This enables procurement teams to quickly identify suppliers that meet both regulatory and technical requirements. Whether it’s sourcing 2,000-litre single-use bioreactor bags or evaluating stainless steel vessels for large-scale production, the platform’s transparent pricing and direct messaging features simplify the quotation process and shorten procurement timelines - especially valuable given the current delays affecting the industry [1].
For facilities adopting hybrid approaches, Cellbase’s carefully curated marketplace streamlines the process of sourcing equipment from multiple vendors while maintaining high-quality standards across both single-use and multi-use systems.
Characterisation and scale-up considerations of single-use bioreactors
Conclusion
When designing a cultivated meat facility, the decision between single-use and multi-use equipment plays a pivotal role. Single-use systems bring advantages like 50% lower capital costs, quicker setup times, and reduced energy consumption [1]. However, they come with higher recurring costs due to consumables [8] and are generally limited to volumes of around 2,000 litres [1]. On the other hand, multi-use equipment demands a larger initial investment - ranging from approximately £7.5 million to £19 million - but offers lower per-unit production costs when scaling beyond 10,000 litres [8].
For many producers, a hybrid approach strikes the right balance. Single-use systems are well-suited for seed trains and early-stage processes where contamination risks are higher. Meanwhile, stainless steel vessels are ideal for large-scale production, leveraging economies of scale while ensuring compliance with GMP standards. This combination allows producers to maintain flexibility while optimising long-term costs [1][8].
As industry expert Chardonny Salisbury highlights:
"The most successful manufacturing strategies will be those that thoughtfully balance current operational requirements with future flexibility" [8].
Key facility design elements - such as cleanroom classifications and utilities infrastructure - are also heavily influenced by the choice of equipment. These decisions are critical to achieving operational efficiency and regulatory compliance.
Sourcing GMP-grade equipment requires thorough supplier assessments and transparent supply chains. Platforms like Cellbase simplify this process by providing access to GMP-compliant equipment, ensuring both single-use and multi-use systems meet the necessary standards. Ultimately, aligning equipment selection with facility design is essential for creating a GMP-compliant cultivated meat production facility.
FAQs
When does multi-use become cheaper than single-use?
Reusable equipment often proves more economical than single-use systems when dealing with large-scale production and long-term cost analysis. Although reusable bioreactors demand a higher initial investment, they help cut down on ongoing expenses like consumables, making them a smart choice for high-capacity cultivated meat facilities. On the other hand, single-use systems come with a lower upfront cost and are a better fit for smaller or more adaptable operations. However, as production scales up, these systems can become expensive due to the rising costs of consumables and waste management.
What tests replace cleaning validation for single-use?
Tests designed to substitute cleaning validation for single-use equipment aim to ensure contaminants remain within safe limits. These methods include analytical residue testing, swab sampling, rinse sampling, and visual inspection. They deliver direct proof that residues and contaminants comply with safety standards, offering a practical alternative to traditional validation approaches.
What does a practical hybrid layout look like?
A hybrid approach in cultivated meat production facilities combines stainless-steel systems with single-use technologies to achieve a balance of adaptability, efficiency, and growth potential. For instance, stainless-steel bioreactors are ideal for large-scale, continuous production, while single-use systems offer flexibility for smaller batches or quick adjustments. This blend allows facilities to respond swiftly to shifting demand, lowers upfront costs, and merges reusable and disposable systems for smoother operations in this evolving field.











