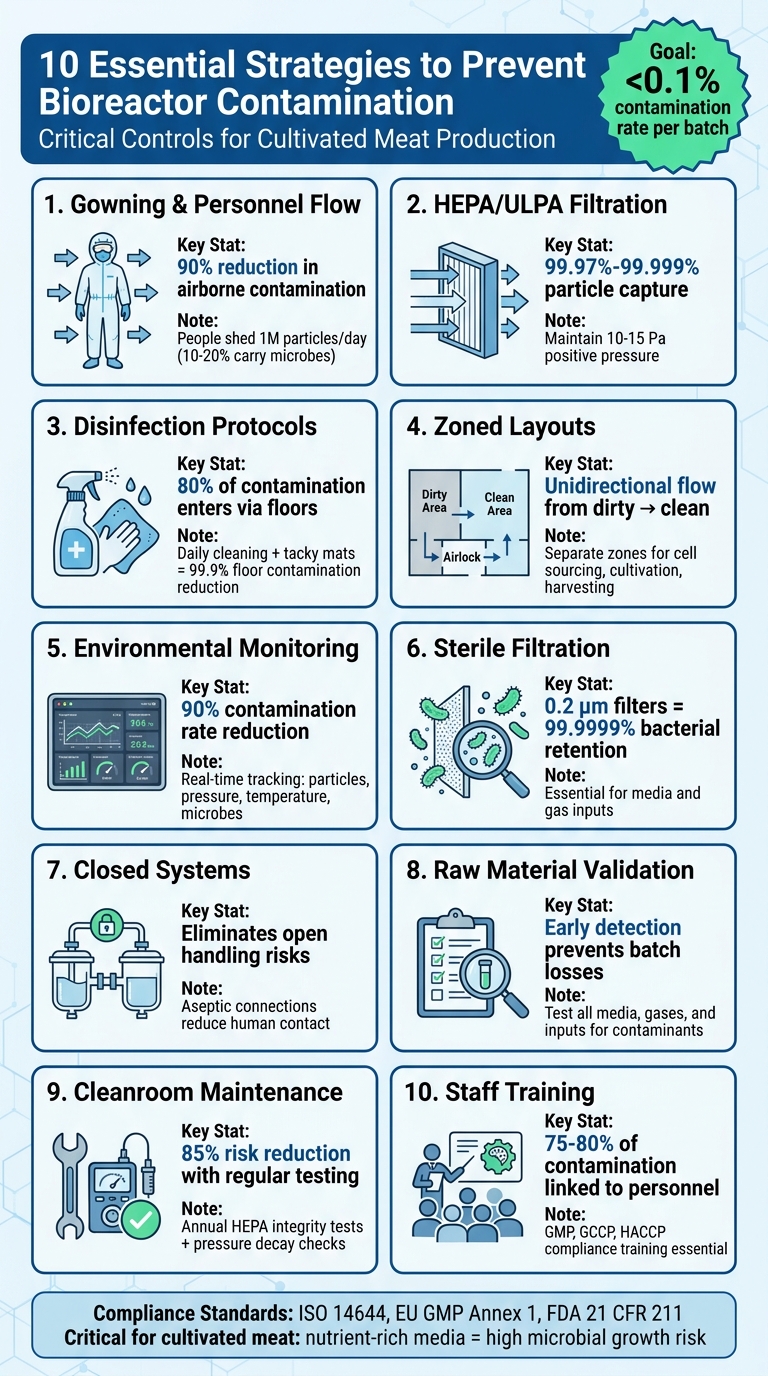
Bioreactor contamination can ruin entire batches and cost thousands. Cultivated meat production relies on maintaining sterile environments to grow sensitive animal cells. The risks are high, but there are proven strategies to prevent contamination. From strict gowning protocols to advanced filtration systems, here’s what you need to know:
- Gowning and Personnel Flow: People are the largest contamination source. Use full-body sterile suits, gloves, and masks alongside strict movement controls.
- HEPA/ULPA Filtration: These filters trap 99.97%-99.999% of particles. Pair them with positive pressure to reduce airborne risks.
- Disinfection Routines: Clean surfaces daily and use tacky mats for floors to block 80% of contamination.
- Zoned Layouts: Separate “dirty” and “clean” areas with unidirectional workflows to minimise cross-contamination.
- Environmental Monitoring: Real-time tracking of particles, pressure, and microbes can cut contamination rates by 90%.
- Sterile Filtration: Use 0.2 μm filters for media and gas inputs to block bacteria and fungi.
- Closed Systems: Aseptic connections reduce human contact and contamination risks.
- Raw Material Validation: Test and verify all inputs, including media and gases, to catch contaminants early.
- Cleanroom Maintenance: Regularly test HEPA filters and airflow to ensure cleanroom integrity.
- Staff Training: Equip personnel with the knowledge to handle materials properly and identify risks.
These steps help keep contamination rates below 0.1%, a critical benchmark for cultivated meat production. Stay vigilant, validate your processes, and invest in reliable tools to safeguard your operations.
10 Essential Strategies to Prevent Bioreactor Contamination in Cleanrooms
Reducing Cell Culture Contamination: Sources of Contamination
sbb-itb-ffee270
1. Use Strict Gowning and Personnel Flow Controls
People are the biggest source of contamination in cleanrooms, shedding around 1 million particles every day. Of these, 10–20% carry microbes that can threaten the sterility of bioreactors [3]. In biopharma facilities, personnel are linked to 75–80% of contamination cases [5], which makes strict gowning and personnel flow controls a critical practice in scaling cultivated meat production.
How Gowning Prevents Contamination
Adhering to strict gowning protocols - such as wearing sterile full-body suits, hoods, boots, gloves, and masks - acts as a barrier, cutting airborne contamination by up to 90% when paired with proper training programmes [2]. For instance, a 2022 study found that rigorous gowning compliance reduced bioreactor contamination rates by 65% during cell culture processes [7]. This level of control is crucial for safeguarding the delicate mammalian cells used in cultivated meat production.
Meeting ISO 14644 and GMP Standards
ISO 14644 requires specific gowning procedures for Class 5 (and cleaner) cleanrooms, which limit particles to fewer than 100 per cubic foot (0.5 μm). These standards rely on laminar flow gowning stations and validated methods [4]. Similarly, EU GMP Annex 1 mandates documented gowning validation in Grade A and B zones, reducing microbial counts from 10³ CFU/m³ to under 1 CFU/m³ [6]. Airlocks, sequential changing rooms, and one-way personnel traffic are additional measures to prevent cross-contamination [1].
Such regulations highlight the importance of robust gowning and flow controls for maintaining sterility in cultivated meat production.
Why This Matters for Cultivated Meat
Cultivated meat bioreactors contain nutrient-rich media that encourage microbial growth. Contaminants like Staphylococcus aureus and Aspergillus species can ruin entire batches, potentially costing tens of thousands of pounds. To avoid this, facilities should enforce a verified seven-step gowning procedure: remove outer clothing, wash hands, apply boot covers, don a hood and mask, wear a jumpsuit, put on gloves, and add goggles. Each step should be validated using contact plate testing to ensure compliance and effectiveness.
2. Maintain HEPA/ULPA Filtration and Positive Pressure Differentials
Effectiveness in Preventing Contamination
HEPA filters are designed to trap 99.97% of particles that are 0.3 μm or larger, while ULPA filters go even further, capturing 99.999% of particles as small as 0.12 μm. These filters are highly effective at removing airborne particles, including Bacillus spores (1–2 μm), which pose a threat to cultivated meat cell cultures[8]. Audits show that combining HEPA/ULPA filtration with positive pressure can significantly lower contamination rates in ISO 7 cleanrooms - from 5–10% down to just 0.1–0.5%[14].
Positive pressure differentials, typically maintained at 10–15 Pa between cleanroom zones, complement these filtration systems by ensuring air flows outward from cleaner areas to less clean ones. Facilities that maintain pressure differentials of 12–20 Pa report up to 80% fewer contamination incidents compared to those with neutral pressure setups[9]. This is particularly important in cultivated meat production, where even a single colony-forming unit (CFU) per litre can spoil a 1,000-litre batch. By combining HEPA/ULPA filtration with positive pressure, contamination risks can be reduced by as much as 95% compared to open processing environments[14].
Compliance with ISO 14644 and GMP Standards
HEPA/ULPA filtration and positive pressure systems are not just effective; they’re also required by international cleanroom standards. ISO 14644-1 specifies that HEPA/ULPA filters must achieve 99.99% efficiency and that positive pressure must be continuously monitored in classified cleanrooms. For instance, ISO 7 cleanrooms (Grade B) are required to maintain particle levels at or below 352,000 particles per cubic metre for particles ≥0.5 μm[10].
EU GMP Annex 1 adds further requirements, such as maintaining a minimum positive pressure of 30 Pa in Class A zones relative to adjacent areas. It also mandates regular filter integrity testing every 6–12 months using DOP/PAO aerosol photometers[10]. Best practices include daily pressure checks (with alarms set for deviations below 10 Pa), quarterly filter testing, and annual recertification. Proper documentation of pressure and particle levels not only supports GMP compliance but can also reduce audit issues by 90%[11].
Relevance to Cultivated Meat Production Processes
For cultivated meat production, where serum-free media is used, bioreactors are especially vulnerable to airborne contaminants like Lactobacillus, which grow rapidly. A UK biopharma cleanroom using ULPA filtration and a 15 Pa pressure differential reduced particle counts from 50 CFU/m³ to under 1 CFU/m³, preventing multiple contamination incidents[14]. This level of control is critical to maintaining cell viability above 90%, especially as operations scale from pilot projects to industrial production systems with capacities of up to 20,000 litres.
To meet these demands, cultivated meat facilities must invest in top-tier cleanroom components, such as advanced filtration units and precise sensors and bioprocess control software for monitoring pressure differentials. Platforms like Cellbase connect industry professionals with verified suppliers, ensuring equipment meets the rigorous standards required for cultivated meat production. When paired with strict gowning protocols, maintaining high-performance HEPA/ULPA systems forms a strong defence against contamination, safeguarding both product quality and production efficiency.
3. Follow Routine Manual and Automated Disinfection Protocols
Effectiveness in Preventing Contamination
Disinfection plays a key role in protecting bioreactors from contamination. Daily manual cleaning of exterior surfaces - such as benches, shelves, tools, and equipment - using disposable cloths and disinfectants can effectively remove microorganisms before they infiltrate bioreactor systems [16].
Floor-level disinfection is especially important, as up to 80% of contamination originates from the floor [15]. Elizabeth Makin from Dycem highlights this risk:
"80% of the contamination in cleanrooms is said to enter critical spaces via the floor level, making it a significant risk to address as part of your contamination control strategy." [15]
Using tacky mats can reduce floor contamination by 99.9% and airborne particulates by 75% [15]. When paired with proper mopping and vacuuming before each shift, these measures significantly lower the chance of contaminants entering bioreactor inputs. Together, these disinfection practices align with the strict requirements of ISO 14644 and GMP standards, discussed below.
Compliance with ISO 14644 and GMP Standards
ISO 14644 sets clear guidelines for cleaning frequencies and materials to maintain cleanroom certification. For ISO Class 5 environments, all cleaning supplies - such as wipes, swabs, and detergents - must be sterile and emit minimal particulates [15]. Environmental testing is required every six months for ISO Class 5 cleanrooms, while ISO Class 6 and higher require testing at least once a year [16].
A systematic cleaning process is essential for compliance. Start with ceilings, then move to walls, windows, and finally the floors. This sequence ensures particles dislodged during cleaning are captured effectively [15][17]. Cleaning personnel should work slowly to minimise air turbulence, which could spread contaminants [17].
Ease of Integration into Cleanroom Workflows
Disinfection protocols can seamlessly fit into cleanroom workflows, complementing gowning and filtration processes. To avoid introducing contaminants, facilities should maintain a separate inventory of cleaning supplies exclusively for the cleanroom. These protocols should align with shift schedules and material intake procedures [17]. Daily tasks should include cleaning and drying windows and pass-throughs to protect material transfer zones. Immediate response to spills is also critical to prevent the spread of contaminants [17].
Relevance to Cultivated Meat Production Processes
In cultivated meat production, strict disinfection protocols work alongside gowning practices and HEPA/ULPA filtration to maintain product integrity. Regular cleaning of bioreactor surfaces, transfer equipment, and sampling ports is crucial to minimise contamination risks. Using ISO-compliant cleaning materials ensures adherence to the rigorous standards required in this field. Platforms like Cellbase help production teams source cleanroom-grade disinfection supplies from verified suppliers, ensuring they meet industry expectations.
4. Design Zoned Layouts with Unidirectional Material and Process Flow
Effectiveness in Preventing Contamination
Zoned layouts with unidirectional material and personnel flow are key to minimising contamination. By guiding movement from "dirty" to "clean" areas, these designs ensure that risks are managed at every stage of production. This is particularly crucial during cell sourcing since raw animal biopsies often carry a higher pathogen load. Dividing the production process into distinct zones - such as cell selection, cultivation, harvesting, and processing - allows targeted contamination controls to be applied at each step.
For example, the seed train phase, where cell cultures are expanded before scaling up production, starts with pharmaceutical-grade cleanroom standards. As the process progresses, controls can shift to food production standards to strike a balance between contamination control and operational efficiency. Closed automation systems further enhance safety by reducing human interaction, speeding up production, and lowering contamination risks [18]. All these measures must comply with ISO 14644 and GMP standards to ensure safety and quality.
Compliance with ISO 14644 and GMP Standards
Cleanroom standards under ISO 14644 are particularly critical for cell banking and expansion phases, where the risk of contamination is highest. During the initial cell sourcing stage, aseptic conditions akin to clinical environments are essential, as animal-derived tissues often come from areas prone to enterobacteria. Maintaining strict hygiene during transport is also non-negotiable [19].
Applying Hazard Analysis and Critical Control Point (HACCP) principles helps pinpoint areas where cross-contamination is most likely to occur within the zoned layout. The Food Standards Agency underscores the value of this approach:
"The principles of Codex and HACCP provide a solid basis to build specific guidelines and quality control plans for this sector, and learnings can be drawn from the clinical / biopharmaceutical industry and adapted to novel food requirements." [19]
Relevance to Cultivated Meat Production Processes
Zoned layouts play a vital role in maintaining the stringent contamination controls required for cultivated meat production. Different areas of the facility demand varying levels of control: ISO-grade cleanrooms are essential for the seed train, medium food-grade standards suffice for large-scale cultivation, and harvesting can adhere to GMP and HACCP protocols. Testing zones are also crucial since bacteria and fungi cause visible turbidity, but viruses and mycoplasma do not, necessitating specialised detection methods [19].
To support these tiered contamination controls, companies like Cellbase provide equipment tailored to each zone. Examples include single-use vs reusable bioreactors, which minimise cleaning needs and reduce downtime. These tools ensure that production remains efficient while meeting safety standards.
5. Install Continuous Environmental Monitoring Systems
Effectiveness in Preventing Contamination
Continuous environmental monitoring systems keep a close eye on critical factors like airborne particles, temperature, humidity, pressure differentials, and microbial contamination in real time. This real-time tracking is a game-changer, capable of cutting contamination rates by up to 90% in biopharmaceutical cleanrooms by enabling immediate corrective actions [22]. For cultivated meat production, this is especially important during cell culture scale-up, where contamination can cause 20–40% of bioreactor batch failures, with airborne microbes responsible for 70% of these incidents [23].
These systems combine tools like particle counters, microbial samplers, and environmental sensors with data logging and alarm functionalities. When parameters - such as particle counts or humidity - go beyond acceptable ISO thresholds, automated alerts prompt swift investigation and action. This proactive approach ensures minor deviations don’t spiral into major contamination issues that could ruin entire production batches, complementing other contamination control measures already in place.
Compliance with ISO 14644 and GMP Standards
Continuous monitoring systems also play a critical role in meeting ISO 14644 and GMP standards. For instance, ISO 14644-1 sets cleanroom classifications based on maximum allowable particle levels - ISO 7, for example, permits ≤352,000 particles/m³ ≥0.5μm. Continuous monitoring ensures facilities maintain less than 10% exceedance over a year, with a 2022 study showing that facilities using automated systems achieved 99.9% compliance with GMP viable particle limits (<1 CFU/m³ in Grade A zones).
Unlike periodic manual sampling, which can miss contamination between checks, these systems generate continuous, auditable records. This is invaluable for cultivated meat producers seeking regulatory approval, as it demonstrates tight process control and ensures product safety certifications. Regular calibration, carried out every quarter following ISO 14644-2 standards, guarantees sensor accuracy and keeps the systems aligned with regulatory requirements.
Ease of Integration into Cleanroom Workflows
Modern monitoring systems are designed to fit seamlessly into existing workflows, offering a unified dashboard for operators. With flexible integration options, these systems minimise installation challenges while maintaining reliability and compliance. For optimal performance, multi-parameter sensors should be positioned at critical locations like bioreactor entry points and material airlocks, collecting data every 1–5 minutes for key parameters [20].
Integration with SCADA systems allows for round-the-clock logging and trend analysis, which is crucial for identifying root causes of contamination. For cultivated meat producers using platforms like Quest Meat to source equipment, choosing monitoring systems compatible with existing bioreactor control software ensures smooth data flow and simplifies validation during setup.
Relevance to Cultivated Meat Production Processes
Cultivated meat production comes with its own set of challenges, requiring precision beyond standard cleanroom parameters. Temperature stability, for example, is critical, as mammalian cell cultures typically need to stay within ±2°C of 37°C. Active air sampling for viable particles during media preparation and inoculation can detect fungal spores early, preventing contamination in bioreactors. Analysing environmental data alongside bioreactor performance can also reveal early warning signs - such as a slight increase in humidity combined with higher particle counts, which might signal a potential HEPA filter issue [21].
6. Use Sterile Filtration for Media and Gas Inputs
Sterile filtration adds another layer of defence against contamination in bioreactor systems, ensuring that microbial contaminants are effectively blocked from entering through media and gas inputs.
Effectiveness in Preventing Contamination
Sterile filtration works by using filters with pore sizes of 0.2 μm or smaller to physically block bacteria, fungi, and other particulates. This technique achieves a 6-log reduction in bacterial presence - over 99.9999% retention - while preserving sensitive media components [8][9]. In cultivated meat production, where nutrient-rich media fosters microbial growth, this level of protection is absolutely critical.
A study in Biotechnology Progress highlighted a 95% reduction in contamination events, dropping from 15% to less than 1% when 0.2 μm filters were used in mammalian cell cultures [14]. In pilot facilities for cultivated meat, validated filtration systems have demonstrated contamination levels as low as less than 1 CFU/100 mL post-filtration, aligning with strict industry standards. This physical barrier is essential to safeguarding entire batches from microbial threats.
Compliance with ISO 14644 and GMP Standards
Sterile filtration systems play a key role in meeting ISO 14644 cleanroom classifications (Classes 5–8) by minimising microbial risks at critical input points. Filters that comply with ASTM F838-05 sterility standards, paired with integrity testing methods such as bubble point or diffusive flow tests, fulfil both EU Annex 1 and FDA 21 CFR 211 GMP requirements [12][13]. This is particularly critical for Grade A and B zones where processes like media preparation and bioreactor inoculation occur.
Documented validation and post-use integrity testing ensure auditable compliance with regulatory guidelines. For facilities using suppliers like Cellbase, filtration systems with built-in integrity testing can streamline regulatory adherence without disrupting operations.
Ease of Integration into Cleanroom Workflows
Modern filtration systems are designed to integrate smoothly into bioreactor workflows. Inline filter housings for media transfer lines and gas manifolds, combined with single-use assemblies like GE ReadyMate, simplify installation, cutting setup time to under 30 minutes and removing the need for cleaning validation. Popular choices include Sartorius Sartopore or Pall Supor capsule filters with 0.2 μm PES membranes for media, and hydrophobic PTFE filters like Millipore Millex for air and CO₂ lines [10][11].
These systems support unidirectional workflows with pre-filtration quality checks, inline filtration during transfers, and post-use integrity testing. Proper sizing of filters - around 50 LMH for media - prevents pressure build-up and extends filter life. Case studies indicate a 20% reduction in filter failures when systems are correctly matched to flow requirements.
Relevance to Cultivated Meat Production Processes
Sterile filtration is non-negotiable in cultivated meat production, where maintaining sterility is vital for uninterrupted cell culture processes. Serum-free media, rich in peptides and growth factors, cannot tolerate heat sterilisation, making filtration the go-to solution. High-density perfusion cultures, operating at 10⁷–10⁸ cells/mL, require continuous contaminant-free gas inputs (O₂/CO₂) to avoid issues like mycoplasma outbreaks, which are particularly problematic in mammalian cell cultures. Hydrophobic PTFE gas filters installed before spargers ensure sterility over long culture periods without the risk of filter wetting.
Using 0.1 μm pre-filters can help prevent clogging when working with high-protein media, while monitoring differential pressure offers early detection of potential problems. This scalable approach supports the transition from research and development to GMP-compliant commercial manufacturing, ensuring reliability at every stage.
7. Apply Closed-System Aseptic Connections and Transfers
Closed-system aseptic connections address one of the most vulnerable areas in bioreactor operations - transfer points. By keeping the culture environment sealed during material transfers, these systems significantly lower the risk of contamination compared to open handling methods.
Effectiveness in Preventing Contamination
Closed systems are designed to maximise automation while reducing the need for human interaction with the culture. As Eileen McNamara, GFI Research Fellow, explains:
"Maximising closed automation throughout cultivated meat production could reduce production time, human contact, and the risk of contamination" [18].
These systems use sterile barriers at every connection point. For example, hydrophobic PTFE membranes in gas filters prevent aqueous aerosols from contaminating the culture, while thermal steam sterilisation is applied directly to reactor inlets and outlets [24]. Single-use bioreactor systems add another layer of safety by eliminating the need for heated sterilisation between batches, which removes the risk of cross-contamination altogether [24].
Compliance with Cleanroom and GMP Standards
Closed-system transfers enhance sterility controls established in earlier stages of production. They reduce the need for constant environmental monitoring during routine operations, allowing facilities to focus stricter controls on early seed train stages. This tiered approach balances safety with cost efficiency [18]. Additionally, removing open handling steps simplifies the validation process required for GMP compliance. Each connection point can be independently verified for sterility, which helps streamline documentation and ensures regulatory requirements are met.
Ease of Integration into Cleanroom Workflows
Modern closed-system technologies are designed to integrate seamlessly into existing bioreactor setups. Automated monitoring systems provide real-time sterility checks and early warnings of contamination [18]. Single-use assemblies further simplify operations by eliminating the need for cleaning validation between batches, saving both time and resources [24]. For facilities using equipment from suppliers like Cellbase, choosing closed-system-compatible components ensures smooth integration from the outset. Pre-sterilised, single-use transfer sets also allow sterile connection and disconnection, making material handling more straightforward.
Relevance to Cultivated Meat Production Processes
In cultivated meat production, closed systems are indispensable. Even brief exposure during transfers can lead to costly contamination issues. Automating labour-intensive steps is crucial to reducing both costs and contamination risks [24]. Since the nutrient-rich media used in cultivation provides an ideal breeding ground for microbes, maintaining sealed transfer points throughout the process is non-negotiable for ensuring successful production.
8. Validate Raw Materials and Supplier Quality Controls
Contamination often sneaks in through raw materials. Whether it’s culture media and supplements, gas supplies, or other inputs, each carries potential risks. That’s why strict validation and supplier quality checks are crucial to keeping contamination out of your bioreactor.
Effectiveness in Preventing Contamination
Validating raw materials acts as a first line of defence, catching contaminants before they ever reach your bioreactor. Targeted testing plays a key role here. For instance, animal-derived ingredients undergo rigorous checks like viral screening, mycoplasma testing (since it doesn’t cause visible turbidity), and endotoxin testing to ensure they’re safe. As a safer alternative, many in the industry now prefer non-animal-derived ingredients to lower the risk of zoonotic diseases. However, when animal tissues are unavoidable for cell sourcing, veterinary certificates confirming the health of the source are essential.
"Early detection, before the medium component is used in culture, is critical" [19].
Compliance with ISO 14644 and GMP Standards
Supplier quality control is an extension of your contamination prevention strategy. Regulatory standards like Good Manufacturing Practice (GMP), Good Cell Culture Practice (GCCP), and HACCP provide clear frameworks for validating supplier quality. These ensure consistent processes and thorough documentation. Organisations such as the United Kingdom Accreditation Services (UKAS) certify that testing services meet ISO 17025 standards, while supplier audits based on Codex and HACCP principles add another layer of microbial safety [19].
Relevance to Cultivated Meat Production Processes
In cultivated meat production, the stakes are even higher. Nutrient-rich media create the perfect conditions for microbial growth, making any contamination especially dangerous. Platforms like Cellbase help ensure suppliers meet GMP standards and provide certified reference materials that align with the sector’s stringent requirements. Additionally, practical measures like using hydrophobic PTFE filters on gas lines prevent aqueous aerosols from entering bioreactors, while sterile filtration of liquid media effectively blocks microbial contaminants before they reach the culture [24].
9. Perform Regular Cleanroom Integrity Testing and Maintenance
Routine integrity testing is essential for all cleanrooms. Over time, HEPA filters wear out, seals can crack, and airflow patterns may shift. Skipping these checks can lead to issues that compromise the sterility of bioreactors.
Effectiveness in Preventing Contamination
Integrity testing helps catch potential problems before they escalate. Just one faulty HEPA filter can release up to 1,000 times more particles, contributing to nearly 40% of contamination incidents in GMP facilities. Regular testing, however, reduces these risks by 85%. Key tests include HEPA filter integrity scans using methods like Dioctyl Phthalate (DOP) or Polyalphaolefin (PAO) aerosol challenges, which confirm 99.99% efficiency for filtering 0.3 μm particles. Pressure decay tests on cleanroom surfaces - such as walls, floors, and ceilings - are also vital for detecting leaks and identifying turbulent airflow that could harbour contaminants. For example, in 2022, a UK biopharma facility discovered a ceiling leak through pressure testing, preventing a £500,000 batch loss.
Compliance with ISO 14644 and GMP Standards
ISO 14644-3 provides clear guidelines for testing schedules. Cleanrooms at risk (ISO 5-8, typical for bioreactor operations) must undergo full integrity tests annually or semi-annually, as well as after any maintenance work. Cultivated meat cleanrooms, often operating at ISO 7, must ensure airborne particle counts stay below 352,000/m³ for 0.5 μm particles. EU GMP Annex 1 also mandates certified testing after installation, repairs, and at specified intervals, with thorough documentation of any issues. Compliance involves using accredited third-party testers with calibrated instruments, documenting results against strict baselines (e.g., <0.01% filter penetration), and resolving any failures within 24 hours. Seamlessly integrating these tests into daily workflows helps ensure product quality remains uncompromised.
Ease of Integration into Cleanroom Workflows
To minimise disruptions, schedule integrity tests during low-activity periods, such as weekends. Testing a 100 m² cleanroom typically takes 4-8 hours. Modern automated systems with remote sensors now allow continuous monitoring, reducing downtime to less than 2% annually. After testing, allow 24 hours for particle levels to stabilise before resuming operations. For instance, a cultivated meat pilot plant in 2024 used smoke pattern tests to address turbulent airflow, cutting viable particle counts by 40% and securing GMP certification.
Relevance to Cultivated Meat Production Processes
In cultivated meat production, where bioreactors handle nutrient-sensitive animal cells at densities exceeding 10⁷ cells/mL, maintaining sterility is non-negotiable. Regular integrity testing prevents the entry of contaminants that could halt cell growth or cause mycoplasma outbreaks. This step is crucial for upholding the high sterility standards required for cultured animal cells, aligning with the contamination prevention strategies discussed throughout this guide. Tools like Cellbase provide verified sensors and testing kits tailored to these processes, ensuring sterility from R&D through to commercial scaling. Post-testing maintenance, such as sealing leaks with silicone, recalibrating pressure gauges to 10-15 Pa, and replacing HVAC filters annually, keeps cleanrooms contamination-free between tests. These efforts complement other controls like strict gowning protocols and advanced filtration systems, ensuring a robust defence against contamination.
10. Train Staff on Contamination Risk Awareness and Response
Effectiveness in Preventing Contamination
In cultivated meat production, operators often present a significant contamination risk [19]. To address this, staff must receive thorough training in areas such as personal hygiene, aseptic techniques, and proper equipment handling. This is especially important during sensitive stages like pre-production and seed train processes [18][19]. Additionally, teaching staff to monitor real-time processing parameters - like pH levels and dissolved oxygen - empowers them to act quickly when contamination is suspected, reducing the risk of losing entire batches.
Compliance with ISO 14644 and GMP Standards
For these practices to be effective, they need to align with recognised regulatory standards.
"Regulatory guidelines and quality assurance schemes such as GMP (Good manufacturing practice), GCCP (Good cell culture practice), HACCP (Hazard analysis and critical control point) are important as they will provide a standard framework to support mitigation measures and the production of safe products." - FSA Research and Evidence [19]
Training programmes should be designed around GMP, GCCP, and HACCP principles to establish strong contamination control measures [19]. Staff must also learn to use cleanroom-specific gear, including clean suits and masks, to guard against contaminants like bacteria, mould, and dust [25]. Environmental monitoring training - covering air, surfaces, and water - further ensures early detection of potential contamination risks through established sterility testing methods [19].
Ease of Integration into Cleanroom Workflows
Adopting widely recognised standards helps simplify quality control processes. By incorporating Codex and HACCP principles alongside aseptic protocols borrowed from the biopharmaceutical sector, cleanroom workflows can become more efficient [19]. Training sessions should ideally take place during quieter production periods to minimise disruptions, with regular refresher courses to keep contamination prevention top of mind [19]. This structured approach supports the sterility measures critical for consistent bioreactor performance.
Relevance to Cultivated Meat Production Processes
Sterile environments are essential for producing cultivated meat. However, the lack of natural microflora means these products can be more vulnerable to rapid spoilage if contamination occurs [19]. Alongside strict gowning protocols and continuous monitoring, comprehensive staff training plays a pivotal role in maintaining the pristine conditions required for production. Training should equip staff to detect subtle signs of contamination, including those caused by undetectable viruses, mycobacteria, and mycoplasma, and to act promptly. Tools like Cellbase provide access to verified testing kits and monitoring equipment tailored for these specific production needs, supporting facilities from research stages to full-scale commercial operations.
Conclusion
Preventing bioreactor contamination in cleanrooms requires a layered approach that combines multiple safeguards. Measures like strict gowning protocols, HEPA/ULPA filtration, continuous environmental monitoring, and thorough staff training work together to maintain aseptic conditions during cultivated meat production. When applied consistently, these strategies allow facilities to scale from research-level production to bioreactors exceeding 1,000 litres, while keeping contamination rates below 0.1% per batch - an essential benchmark for meeting regulatory standards [8].
A strong contamination control plan also hinges on careful sourcing and supplier quality management. The journey to scalable, compliant production starts with validating materials rigorously. Subpar growth media, sensors, or bioreactors can introduce contaminants that jeopardise entire batches, highlighting the need for stringent quality assurance at the procurement stage. This ensures effective raw material validation and supports sterile input processes.
For cultivated meat producers, Cellbase offers a dedicated B2B marketplace connecting production teams with trusted suppliers for bioreactors, growth media, sensors, cell lines, and monitoring equipment. This platform ensures traceability and provides access to products with verified quality certifications.
To minimise contamination risks, focus on these key strategies:
- Strict gowning protocols and controlled personnel movement
- HEPA/ULPA filtration systems and positive pressure differentials
- Regular manual and automated disinfection routines
- Zoned layouts with unidirectional workflows
- Continuous environmental monitoring and data analysis
- Sterile filtration for media and gas inputs
- Closed-system aseptic connections and transfers
- Rigorous validation of raw materials and supplier quality
- Routine cleanroom integrity testing and maintenance
- Comprehensive staff training on contamination risks
Evaluate your cleanroom practices against these strategies. Emphasising staff education and supplier validation can transform contamination prevention from a reactive task into a proactive framework for scaling production. Partnering with platforms like Cellbase ensures you have the tools and resources to build a solid foundation for commercial success.
FAQs
What are the first signs a bioreactor batch is contaminated?
The first signs of bioreactor contamination often stem from several common issues. These include breaches at ports during sampling or maintenance, failures in gas filters, contaminated growth media, risks introduced during sensor installation, and even microplastic contamination. Such problems can result in microbial overgrowth, which may ultimately lead to the failure of an entire batch.
How do I choose the right ISO cleanroom grade for each production step?
In cultivated meat manufacturing, the level of cleanliness required varies depending on the sensitivity of each production stage. Here's how you can match the ISO cleanroom grade to the specific needs of the process:
- ISO Class 5: Reserved for critical steps like cell seeding, where maintaining an ultra-clean environment is essential to prevent contamination.
- ISO Class 6: Ideal for bioreactor operations, offering a controlled setting that balances cleanliness with practicality.
- ISO Class 8: Suitable for less sensitive tasks such as harvesting and transfers, where risks are lower, and closed systems can help maintain product integrity.
By assigning stricter controls to high-risk stages and using lower-grade cleanrooms for less critical steps, manufacturers can manage costs effectively while minimising contamination risks.
What tests should be performed on media and gases before they enter the bioreactor?
Sterility tests play a crucial role in verifying both media and gases before they are introduced into a bioreactor. For media, membrane filtration is the preferred method, while microbiological sampling is commonly used for gases. These procedures are designed to detect any microbial contamination, ensuring that only sterile materials make their way into the production process. This step is vital for minimising contamination risks, which is a key priority in cultivated meat production.











