Protein adsorption is crucial in cultivated meat production. It forms the initial protein layer on scaffolds, enabling cell adhesion, growth, and differentiation. This process mimics the extracellular matrix (ECM), ensuring cells attach and develop properly, particularly with non-animal scaffolds. Here's a quick breakdown:
- Scaffold Surface Properties: Porosity, stiffness, and hydrophilicity affect protein adsorption and cell behaviour.
-
Material Variations:
- Chitosan/Hydroxyapatite (CS/HAp): High porosity, stability, and protein interaction.
- Polyester-Based Scaffolds (e.g., PLA): Depend on growth media proteins for cell adhesion.
- PLLA/HAp Composites: Improved hydrophilicity and protein adsorption compared to pure PLLA.
- Growth Media Proteins: ECM proteins like fibronectin and collagen guide cell activity and tissue formation.
Choosing the right scaffold involves aligning its properties with the protein profile of the growth media. Platforms like Cellbase simplify sourcing materials tailored for cultivated meat production.
Lec 31: Protein Adsorption on Biomaterial Surfaces | Polymeric Biomaterials
sbb-itb-ffee270
How Proteins Adsorb to Scaffold Surfaces
Proteins from growth media naturally rearrange themselves to minimise free energy, forming a film that reduces surface tension and influences how cells interact with the scaffold surface [1]. This process relies on differences in adhesion and interfacial tension, which help organise proteins and affect cell clustering [1]. For scaffolds without inherent cell-binding motifs, such as those made from non-animal sources, surface functionalization like integrating RGD peptides are often necessary to enhance protein adsorption and promote cell attachment [1]. These processes explain the diverse adsorption behaviours seen across various scaffold materials.
Surface Properties That Affect Protein Adsorption
The physical characteristics of scaffolds, such as their surface-to-volume ratio and porosity, play a major role in protein adsorption and subsequent cell responses [1]. For instance, in chitosan/gelatin composites, a balanced 1:1 ratio achieves optimal adhesion energies - 239 kcal mol⁻¹ for collagen I and 149 kcal mol⁻¹ for fibronectin. However, when this ratio is skewed, both adhesion and cell viability are negatively impacted [4]. Additionally, scaffolds that mimic the stiffness of natural muscle tissue (2–12 kPa) are better suited for supporting cell expansion. Conversely, scaffolds with higher stiffness levels may lead to premature cell differentiation [1]. Adjusting scaffold chemistry, such as incorporating RGD peptides, can further fine-tune protein adsorption and improve cell adhesion.
Protein Interactions with Growth Media Components
Protein interactions with components in the growth media also have a significant impact on cell behaviour [1]. Proteins in the media act as a bridge between scaffold surfaces and cells. For example, extracellular matrix proteins like fibronectin and collagen play a critical role in early cultivation stages by encouraging myoblast multiplication and migration. Meanwhile, laminin and type IV collagen provide structural support as myoblasts fuse into multinucleated myotubes [1]. Proteoglycans, such as heparan sulphate and decorin, bind the scaffold’s basement membrane to collagen and help sequester growth factors. This creates localised concentrations of signalling molecules that guide cell activity [1]. Advances in molecular dynamics simulations now allow researchers to predict scaffold biocompatibility by calculating the adhesion energy of these proteins before conducting experimental tests [4].
Protein Adsorption on Different Scaffold Materials
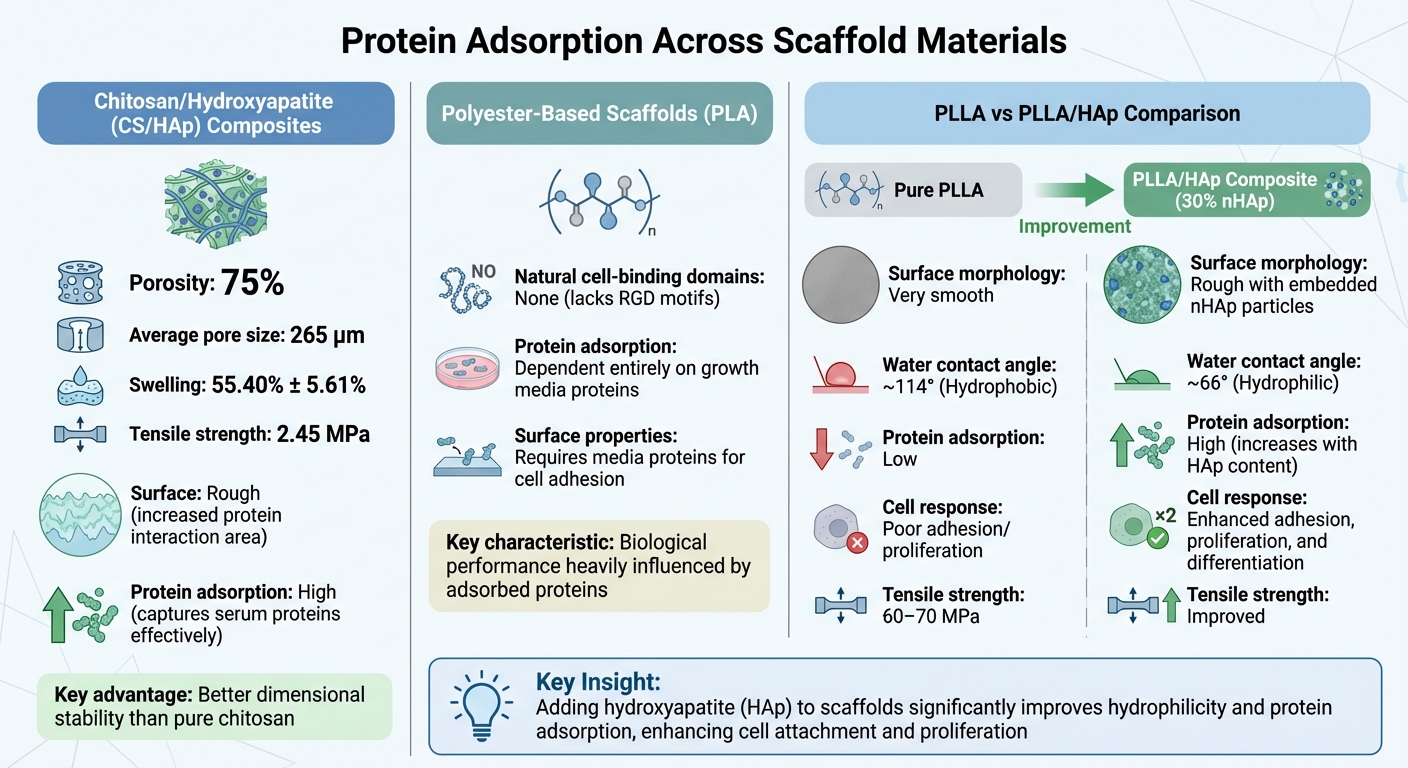
Comparison of Scaffold Materials for Protein Adsorption in Cultivated Meat Production
Scaffold materials exhibit distinct behaviours when it comes to protein adsorption, which plays a key role in determining their suitability for cultivated meat production. By understanding these variations, researchers can choose materials that best match specific cell culture needs and growth media compositions.
Chitosan/Hydroxyapatite (CS/HAp) Composites
Adding hydroxyapatite (HAp) nanoparticles to chitosan changes its surface properties, leading to improved protein adsorption. CS/HAp scaffolds boast a porosity of 75% and an average pore size of 265 μm, which supports effective cell migration while maintaining structural integrity during incubation in growth media [5]. The rough surface created by HAp increases the area available for protein interactions [5].
These composites swell by 55.40% ± 5.61%, compared to 71.03% ± 6.21% in pure chitosan, offering better dimensional stability. This prevents excessive deformation while still allowing nutrients to diffuse from the growth media. Additionally, the tensile strength of CS/HAp scaffolds reaches 2.45 MPa - about twice that of pure chitosan (1.21 MPa) - and falls within the range of cancellous bone [5]. Together, these properties - porosity, controlled swelling, and improved tensile strength - enhance protein adsorption, promoting optimal cell attachment for cultivated meat. Studies using foetal bovine serum (FBS) in minimum essential medium confirm that these scaffolds effectively capture essential serum proteins crucial for cell signalling and attachment [5]. These features set CS/HAp composites apart from synthetic polyester scaffolds.
Polyester-Based Scaffolds
Unlike natural composites, synthetic polyester scaffolds like PLA depend entirely on proteins from the growth media for cell adhesion. These materials lack natural cell-binding domains, such as RGD motifs, making protein adsorption a key factor in regulating cell adhesion, migration, and differentiation [6]. The biological performance of these scaffolds is, therefore, heavily influenced by the specific proteins that adsorb to their surfaces during initial contact with growth media.
PLLA vs PLLA/HAp Scaffolds
Enhancing PLLA with HAp significantly improves its surface hydrophilicity and protein adsorption. Pure PLLA has a hydrophobic surface with a water contact angle of roughly 114° [7]. Adding 30% nano-hydroxyapatite (nHAp) reduces this angle to 66°, creating a more hydrophilic surface and introducing a rough morphology featuring embedded nHAp particles [7].
Research from Wuhan University of Technology demonstrated that embedding 10–30% nHAp into PLA microspheres via emulsion solvent evaporation increased BSA adsorption and enhanced rat mesenchymal stem cell adhesion and osteogenic differentiation [7].
"The composition and conformation of the adsorbed protein layer is considered to be one of the major factors in determining the nature of cell interaction with the materials."
In growth media, the adsorbed protein layer - commonly derived from BSA or FBS - acts as a critical interface, influencing cell spreading and integrin binding [7][9].
| Property | Pure PLLA Scaffold | PLLA/HAp Composite Scaffold |
|---|---|---|
| Surface Morphology | Very smooth [7] | Rough; nHAp particles embedded [7] |
| Water Contact Angle | ~114° (Hydrophobic) [7] | ~66° (Hydrophilic) [7] |
| Protein Adsorption | Low; limited by hydrophobicity [8] | High; increases with HAp content [7] |
| Cell Response | Poor adhesion/proliferation [7] | Enhanced adhesion, proliferation, and osteogenic differentiation [7] |
| Tensile Strength | 60–70 MPa [8] | Improved tensile strength [5] |
How Protein Adsorption Affects Scaffold Selection
When a scaffold comes into contact with growth media, proteins immediately form a thin film on its surface. This initial layer sets the stage for every interaction between cells and the biomaterial [10][11]. To ensure compatibility, scaffold surface properties must align with the protein profile of the growth media. Factors like pH, ionic strength, and additives such as sugars or surfactants also come into play [10]. For scaffolds derived from plants, algae, or fungi, this balance is even more crucial. These materials lack natural cell-binding domains, relying entirely on adsorbing the right proteins from the media to support cell attachment [1]. These considerations are key when selecting scaffolds tailored to specific cell types and growth media.
"If the polymer scaffold does not allow for any protein adsorption, cellular adhesion would not occur and ultimately the device would fail."
- Yaser Dahman, Author, Biomaterials Science and Technology [10]
Selecting Scaffolds with Optimal Protein Adsorption
Effective scaffold selection hinges on matching its protein adsorption traits to the needs of your specific cell type and growth media. Adhesion energy between the scaffold and extracellular matrix proteins - like fibronectin and collagen type I - is a strong indicator of biocompatibility and cell viability [4]. Scaffolds with high surface-to-volume ratios and suitable porosity provide more surface area for protein adsorption, while mechanical stiffness must align with the target tissue. For instance, muscle differentiation requires a Young’s modulus of about 18 kPa, whereas adipogenic differentiation thrives at approximately 3 kPa [2]. To compensate for natural limitations in protein-binding capacity, surface modifications such as RGD motifs or peptide coatings can be added to plant-based scaffolds, ensuring reliable cell adhesion [1].
Optimising hydrophilicity and porosity can significantly enhance protein adsorption. For example, scaffolds with swelling degrees of 2,004% improve serum protein adsorption, promoting cell proliferation [10]. Materials like tricalcium phosphate and silk fibroin can adsorb around 1.5 mg/mL of Bovine Serum Albumin, equivalent to 43% w/w of the protein in the stock solution [10]. This translates into cell-seeding rates exceeding 84%, with cell viability consistently above 95% during culture periods [3].
"The properties of biomaterials are largely determined by the proteins adsorbed on their surfaces, which are critical in regulating cell adhesion, migration, proliferation, and differentiation."
- npj Science of Food [1]
Sourcing Scaffold Materials Through Cellbase
Once you’ve pinpointed the optimal protein adsorption characteristics, finding the right materials becomes the next challenge. General laboratory suppliers often lack the specialised scaffolds needed for cultivated meat production. That’s where Cellbase comes in - a dedicated B2B marketplace tailored to this field. It connects researchers and production teams with verified suppliers offering scaffolds designed specifically for cultivated meat applications.
Each scaffold listing on Cellbase includes detailed use-case specifications, such as compatibility with serum-free media or GMP-compliant designs. This makes it easy to identify materials that match your growth media and cell type requirements. By streamlining the sourcing process, Cellbase reduces procurement challenges and technical risks, ensuring that scaffold surface properties align with your protein adsorption needs. Whether you’re in R&D exploring options or scaling up for commercial production, the platform’s searchable catalogue and direct supplier communication simplify decision-making, saving time and resources.
Conclusion
Protein adsorption plays a central role in determining scaffold performance in cultivated meat production. From the moment a scaffold interacts with growth media, proteins form a layer on its surface, influencing every cellular process - starting with attachment and continuing through to differentiation. This adsorbed protein layer is what drives cell adhesion, proliferation, and eventual maturation into the desired tissue type [1].
For non-animal scaffolds, achieving effective protein adsorption requires more than just basic compatibility. Key factors like biomaterial properties such as surface roughness, charge, hydrophilicity, and mechanical stiffness must all align with the needs of the specific cell type and the protein composition of the growth media.
A compelling study highlights this relationship. In September 2024, researchers at Konkuk University, led by Do Hyun Kim, compared soy and pea protein scaffolds for porcine adipose-derived stem cells. Their results showed that soy protein-agarose scaffolds, with water absorption rates of 2,300–2,500%, significantly outperformed pea protein scaffolds (1,100–1,200%) in promoting cell adhesion and proliferation [12]. This example demonstrates how material properties directly influence cultivation success.
Finding suitable scaffold materials requires sourcing from suppliers who understand these intricate requirements. A clear understanding of protein adsorption not only aids in scaffold design but also simplifies the process of material selection. Platforms like Cellbase support this by connecting researchers and production teams with trusted suppliers offering scaffolds designed for cultivated meat. With detailed specifications - such as serum-free media compatibility and GMP compliance - Cellbase helps streamline procurement, saving time while reducing technical risks.
FAQs
How can I tell if my growth media proteins will adsorb well to a scaffold?
Protein adsorption is influenced by the scaffold's surface characteristics, such as roughness, chemistry, and surface energy, as well as the proteins present in the growth media. Pre-treating scaffolds with serum-containing media can increase protein adsorption, which plays a key role in promoting cell attachment and growth. In the context of cultivated meat, using scaffolds specifically designed to optimise protein binding can significantly aid tissue development.
What scaffold surface tweaks improve cell attachment on non-animal materials?
Improving how cells attach to non-animal scaffold materials often involves tweaking the surface. Techniques like increasing surface roughness or introducing biochemical binding sites can make a big difference. These changes, achieved through treatments or coatings, help strengthen the connection between cells and the scaffold, leading to better compatibility overall.
What quick tests can show if protein adsorption supports good cell adhesion?
To evaluate whether protein adsorption facilitates effective cell adhesion, observe cell attachment following brief incubation periods. Compare results in the presence and absence of serum proteins, and quantify the levels of adsorbed serum proteins. Link these observations to cell proliferation, as higher protein adsorption often leads to improved adhesion.











