Validation protocols ensure cultivated meat production is safe, consistent, and meets regulatory standards. Without heat-treatment steps to kill pathogens, sterility must be maintained at every stage. These protocols focus on:
- Equipment validation: Ensuring bioreactors and sterilisation systems function correctly.
- Materials validation: Verifying growth media and other inputs are sterile and of high quality.
- Process validation: Maintaining aseptic techniques and environmental controls.
By addressing risks like microbial contamination and cell line misidentification, these protocols help producers meet standards from organisations like the FDA and EFSA. Companies such as UPSIDE Foods and Aleph Farms have already achieved regulatory approval by following these measures. Tools like HACCP analysis, sterility testing, and third-party documentation are critical for compliance and consumer trust.
Platforms like Cellbase simplify procurement of validated resources, connecting producers with GMP-compliant suppliers for equipment, media, and testing tools tailored to cultivated meat production.
Risk Mitigation in Cultivated Meat Production
Primary Risk Factors
Transitioning from laboratory research to large-scale production brings several challenges, with three risks standing out. The most immediate concern is microbial contamination caused by adventitious agents. This demands rigorous sterility measures throughout every stage of production to ensure safety.
Another major issue is cell line misidentification. This has been a longstanding problem in scientific research, where mislabelled or cross-contaminated cell lines have led to flawed results. In the context of cultivated meat, such errors can result in products that don't align with the intended species or tissue type, sparking both safety and regulatory issues. Additionally, inconsistent results during the scale-up of cell cultures can disrupt commercial manufacturing processes.
"For commercial viability, cultivated meats require scientifically informed approaches to identify and manage hazards and risks." – Nature Food [4]
How Validation Protocols Manage Risk
To tackle these risks, validation protocols are specifically designed to address each threat. Borrowing from pharmaceutical biotechnology, these protocols establish a robust quality control framework. For example, they incorporate ICH Q5A-based methods to proactively identify and eliminate viral contamination risks. Instead of waiting for contamination to occur, these measures focus on prevention through detailed cell line characterisation, the creation of food-grade cell banks, and continuous monitoring of critical control points.
Microbial contamination is managed with stringent sterility testing and ongoing surveillance. Meanwhile, cell line misidentification is countered with thorough characterisation processes and secure banking protocols.
A key part of this approach is pre-market safety assessments, which document every aspect of production. Companies like UPSIDE Foods and GOOD Meat achieved full regulatory approval in the United States in 2023 by presenting detailed safety dossiers that adhered to these validation protocols. Similarly, Aleph Farms secured the world’s first regulatory approval for cultivated beef in early 2024 by following comparable documentation standards [4]. These protocols not only satisfy regulatory authorities - such as the FDA, Singapore Food Agency, and Food Standards Australia New Zealand - but also ensure batch-to-batch consistency, which is vital for commercial success.
These risk management strategies lay the groundwork for the validation components discussed in the next section.
Validation and Verification Practices for the Food Industry
Core Components of Validation Protocols
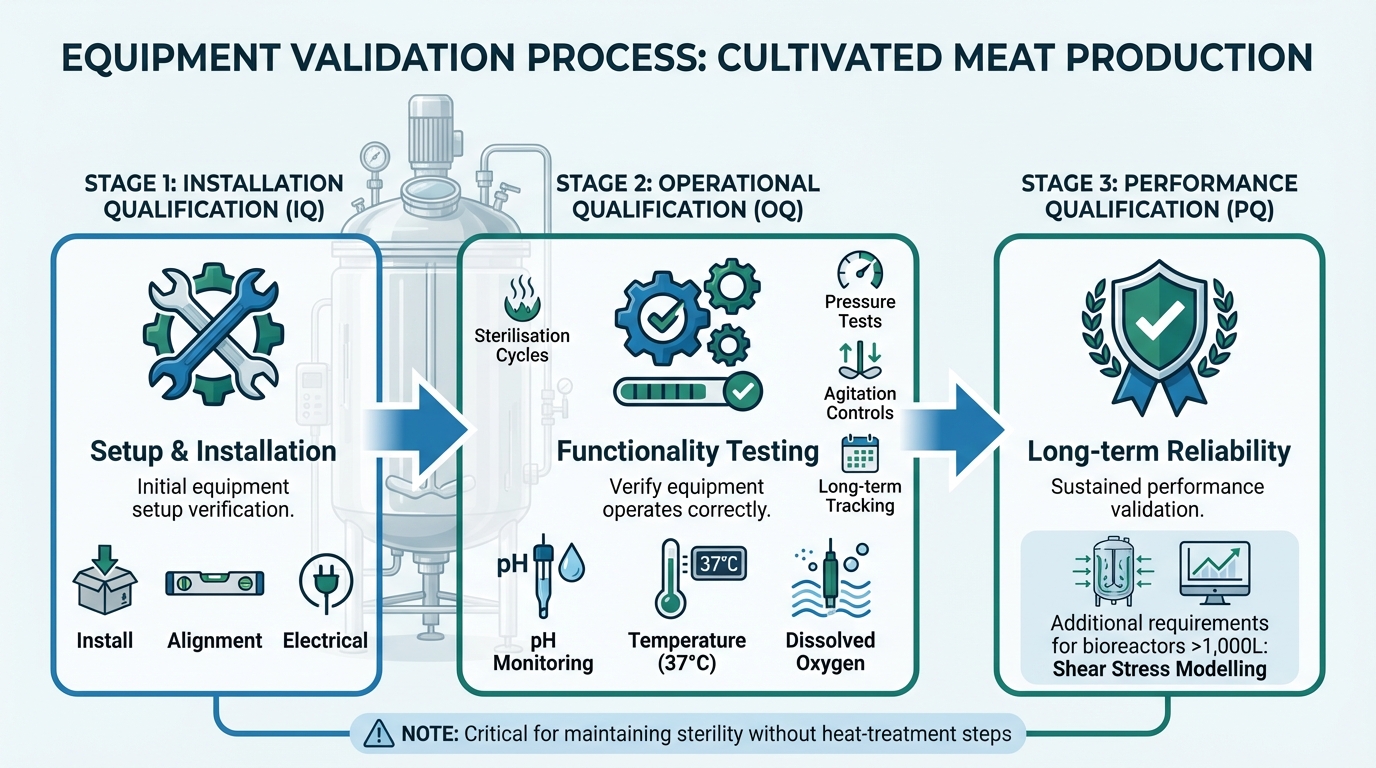
Three-Stage Equipment Validation Process for Cultivated Meat Production
Validation protocols in cultivated meat production are built on three key areas - equipment, materials, and processes - to ensure sterility and consistency in the absence of traditional kill steps [1]. These components form a structured approach to maintaining contamination-free production.
Equipment Validation
Equipment validation happens in three stages: Installation Qualification (IQ) for setup, Operational Qualification (OQ) for functionality, and Performance Qualification (PQ) for long-term reliability. For bioreactors, this involves validating sterilisation cycles, pressure tests, and agitation controls. Sensors need careful calibration to monitor pH, temperature (37°C), and dissolved oxygen in real time [2]. Using purpose-built tools rather than standard pharmaceutical equipment can improve accuracy and reliability [2].
Surfaces are swabbed, and rinse water is tested both before and after use. Additionally, sterilisation cycles, storage conditions, supplier certificates, and expiry dates must be meticulously documented [1]. For larger bioreactors (over 1,000 litres), validation must also address shear stress modelling to ensure that mechanical forces do not damage delicate mammalian cells, a critical step in preventing contamination, which remains a significant challenge [5].
Materials Validation
Once the equipment is validated, materials used in the process must be checked for quality and consistency. Growth media, for instance, is analysed using High-Performance Liquid Chromatography (HPLC) and mass spectrometry to confirm its nutrient profile [3]. Residue screening employs sensitive methods like ELISA or LC-MS/MS to detect antibiotics and hormones, ensuring they don’t contaminate the final product [3]. Scaffolds are tested for sterility and material compatibility to avoid leaching, while genomic sequencing ensures cell lines remain genetically stable. Traceability records are also maintained to track every component in the media supply chain [3].
Antibiotic residue testing is particularly important, as cultivated meat lacks the cooking step that typically eliminates contaminants. Downstream washing processes must remove all antibiotics and growth factors. Multiple samples from each batch are tested against maximum residue limits (MRLs) using validated LC-MS/MS methods, and certificates of analysis confirm residues are below detectable levels [3]. This level of transparency and third-party verification helps meet regulatory standards and builds consumer trust.
Process Validation
Process validation ensures that cell culture and bioprocessing workflows consistently deliver contamination-free results. This involves sterility testing, recovery studies using indicator organisms, microbial testing, and monitoring of key factors like cell culture conditions, media quality, and bioreactor performance [1][2]. Sampling is aligned with growth phases and feeding cycles, focusing on high-risk areas such as media preparation and bioreactor connections [1][2].
Reproducibility tests with multiple operators help identify potential inconsistencies [1]. Recovery studies use indicator organisms to test detection sensitivity, while all processes integrate with HACCP critical control points. These include microbial testing, lipid analysis, and shelf-life assessments at every stage to ensure safe and consistent bioprocessing [1][2]. Comprehensive documentation - covering personnel training, validation photos, deviation logs, and review signatures - creates a complete audit trail for regulatory and quality assurance purposes [1].
sbb-itb-ffee270
Implementation Steps for Validation Protocols
Building on the essential components of validation, here’s how to implement effective protocols step by step.
Conducting HACCP Analysis
Start the HACCP analysis by forming a diverse team of experts across areas like cell sourcing, isolation, proliferation, harvesting, food safety, and facility maintenance. To meet regulatory requirements, ensure at least one team member has Level 4 HACCP training. Begin by clearly defining the product's scope, expected shelf life, packaging, and target audience.
Next, design a process flow diagram that outlines every stage, from cell isolation to the final product. Include all inputs - such as culture media and growth factors - and outputs like waste and by-products. Categorise hazards into four main types:
- Biological: Risks like microbial contamination
- Chemical: Residues from antibiotics, growth factors, or reagents
- Physical: Potential fragments of metal, plastic, or glass
- Allergenic: Cross-contact risks from media components
Use a decision tree to identify Critical Control Points (CCPs) and set measurable limits (e.g., temperature, pH, time) to manage these hazards effectively. Review and update the HACCP plan at least annually or whenever there are changes to the product, processes, or equipment.
Setting Up Monitoring and Prevention Systems
Once hazards and CCPs are identified, the next step is to establish robust monitoring and correction systems.
Define your monitoring protocols by specifying methods, frequency, responsibilities, and the type of data to be recorded. For automated systems, continuous monitoring should be complemented by scheduled equipment checks. In contrast, manual systems require checks aligned with the scale and risk of operations. As the Food Standards Agency explains:
"Monitoring may be as simple or complex as the step requires. It may be just a visual check, or done through the use of specific measuring instruments" [6].
Assign dual roles where one person performs the monitoring and another verifies the checks. If a CCP repeatedly fails, conduct a root cause analysis to identify and resolve underlying issues. Implement a three-stage corrective action plan to restore control, address any affected products, and prevent similar problems in the future. Regular calibration of instruments and periodic audits are vital to ensure the monitoring system remains effective.
Documentation and Compliance Verification
To comply with UK regulations for cultivated meat, maintain meticulous documentation for HACCP, SOPs, and monitoring records. Under Regulation (EC) 853/2004, cultivated meat is classified as a Product of Animal Origin (POAO), requiring adherence to specific hygiene standards. Organise your documentation into three key sections:
- HACCP Plan: Include the team list, scope, process flow diagram, and hazard analysis.
- Policy Documents: Cover Standard Operating Procedures, hygiene policies, and prerequisite programmes.
- Records: Maintain logs for monitoring, corrective actions, test results, and instrument calibration.
Use a version control system for both physical and digital documents to ensure all staff have access to the latest procedures. Validate the HACCP plan before implementation and verify its effectiveness afterward. Conduct internal checks at least once a year, regardless of any issues. If corrective actions are taken, document the root cause analysis thoroughly. As the Food Standards Agency highlights:
"Food business operators shall put in place, implement and maintain a permanent procedure or procedures based on the HACCP principles" [6].
Comprehensive documentation not only ensures regulatory compliance but also prepares your operation for audits. These practices also pave the way for sourcing validated resources through platforms like Cellbase.
Using Cellbase for Validation Resource Procurement
How Cellbase Supports Validation
Once your HACCP plan and monitoring systems are in place, securing validated equipment and materials becomes the next step. This is where Cellbase plays a vital role. It connects professionals in the cultivated meat industry with trusted, GMP-compliant suppliers. You’ll find resources like bioreactors with built-in monitoring systems, sterile sampling tools, and analytical instruments designed for residue testing.
What sets Cellbase apart is its exclusive focus on cultivated meat production. Each listing includes compatibility details tailored to meet your validation requirements. For instance, you can source bioreactors that maintain critical parameters such as temperature (0–4°C) and pH, equipped with sensors for real-time monitoring at the critical control points defined by your HACCP analysis.
Suppliers on Cellbase provide essential documentation, like batch numbers and certificates of analysis, ensuring traceability and adherence to regulatory standards. When sourcing growth media, cell lines, or scaffolds, you’ll also find residue testing documentation - using methods like HPLC/Mass Spec or LC-MS/MS - to confirm that inputs meet maximum residue limits set by agencies like the FDA and EFSA. As David Bell, founder of Cellbase, explains:
"When you're building cell culture workflows for cultivated meat production, the requirements shift. Food-grade certifications. Cost structures optimised for commercial production, not research budgets" [7].
This highly curated approach ensures compliance while streamlining the procurement process.
Procurement Advantages Through Cellbase
Traditional procurement processes often slow down production. Cellbase addresses this with transparent pricing and direct communication with suppliers. Its curated listings make it easy to identify tools specific to validation needs, such as sterility swabbing kits, equipment for testing indicator organisms during recovery studies, and cold chain containers to preserve sample integrity during transport.
Suppliers on Cellbase are well-versed in food-grade sterility requirements, which differ significantly from pharmaceutical standards. This ensures that your validation protocols are supported by sourcing solutions that meet the same rigorous standards.
Conclusion
Validation protocols are the cornerstone of producing cultivated meat safely and at scale, addressing biological, chemical, and physical risks while ensuring compliance with UK regulations. As the Food Safety and Inspection Service explains, "Validation is the process of demonstrating that the HACCP system as designed can adequately control identified hazards to produce a safe product" [8]. Without thorough scientific backing and practical application, production systems cannot meet the stringent standards set by the FSA and FSS.
To implement these protocols, validated equipment, materials, and monitoring systems are essential. Cellbase simplifies this process by connecting you with trusted, GMP-compliant suppliers who meet food-grade requirements. From bioreactors equipped with advanced sensors to analytical tools and detailed traceability documentation, Cellbase provides the resources needed for regulatory compliance. This streamlined approach also facilitates the shift to food-grade materials, a critical step for scaling production.
Transitioning to food-grade materials is a key milestone for commercial success. With Cellbase's carefully curated listings, you can source essentials like serum-free media and scaffold materials in a cost-effective and safe manner.
As the UK's regulatory sandbox programme continues until February 2027, backed by £1.6 million in government funding [9], there’s a unique opportunity to establish robust validation protocols. Cellbase ensures access to the specialised, validated resources necessary to meet these high standards and support the efficient scaling of cultivated meat production.
FAQs
What are the biggest validation risks when scaling cultivated meat?
When scaling up cultivated meat production, two major challenges stand out: contamination control and process consistency.
Contamination becomes a bigger threat as production scales up. Larger operations increase the risk of contamination, which can lead to expensive batch failures. To minimise this, maintaining sterility, qualifying raw materials, and managing environmental conditions are absolutely essential.
Consistency in the production process is another critical factor. Parameters like pH levels and cell density must stay within strict limits to ensure the end product meets quality and regulatory standards. Establishing detailed, rigorous protocols is crucial for managing these risks and achieving a scalable, reliable production process.
How do IQ, OQ and PQ apply to bioreactors in cultivated meat?
When it comes to cultivated meat production, validating bioreactors involves three essential steps: Installation Qualification (IQ), Operational Qualification (OQ), and Performance Qualification (PQ). Each plays a key role in ensuring smooth and compliant operations.
- Installation Qualification (IQ): This step verifies that the bioreactor is installed correctly, adhering to both manufacturer guidelines and regulatory requirements. It’s about making sure the foundation is solid before moving forward.
- Operational Qualification (OQ): Once installed, the bioreactor needs to prove it can function as expected. This involves testing it under specific conditions, such as maintaining precise temperatures and pH levels, to confirm everything works as it should.
- Performance Qualification (PQ): The final step is about consistency. Here, the focus is on ensuring the bioreactor performs reliably during actual production processes, meeting the demands of cultivated meat manufacturing.
By following these steps, producers can reduce risks, maintain compliance, and create a strong foundation for scalable, high-quality production. Tools like Cellbase can simplify the process by helping source equipment designed to meet these stringent standards.
What evidence do regulators expect in a pre-market safety dossier?
Regulators expect companies to provide proof of hazard identification, risk assessment, and the implementation of control measures. This involves maintaining meticulous documentation, covering everything from raw material details to process validation and safety testing. Clear and comprehensive records not only show adherence to safety regulations but also play a crucial role in securing approval for cultivated meat products.











