Mini bioreactors are compact systems (10–500 mL) designed for efficient media testing in industries like cultivated meat. They allow researchers to perform multiple experiments simultaneously, saving time, resources, and costs. These systems replicate industrial-scale conditions, ensuring reliable results for scaling up. Key features include automated controls for pH, oxygen, and temperature, and real-time monitoring of cell growth and metabolites. Their small volumes (as low as 10 mL) reduce media use and waste, while automation minimises labour. Popular systems include the ambr™ series and BioLector platforms, each suited to specific research needs.
Key Takeaways:
- High-throughput testing: Run 24–48 experiments in parallel.
- Scalability: Results from small volumes (10–15 mL) translate well to large-scale systems (up to 400 L).
- Resource efficiency: Lower media consumption and reduced labour via automation.
- Specialised designs: Stirred-tank systems for industrial replication, air-lift for low-shear environments, and multi-well plates for early-stage testing.
These tools streamline media optimisation, making them indispensable for cultivated meat production. Platforms like Cellbase simplify sourcing and support researchers with tailored equipment and expert advice.
Benefits of Mini Bioreactors for Media Testing
Faster Growth Media Optimisation
Mini bioreactors make media optimisation quicker by enabling multiple tests to run in parallel. For example, the ambr™ 48 system can handle 48 independent experiments at the same time, allowing researchers to test dozens of media compositions in one batch [1]. This approach avoids the delays caused by testing sequentially, which is a common limitation of traditional bench-top systems.
Using Design of Experiments (DoE) methods adds further precision to the process. In August 2025, researchers at the University of Oklahoma used the Ambr® 250 system and a central composite design to optimise CHO cell cultures. They found that a seeding density of 1.1 × 10⁶ cells/mL and a feeding rate of 2.68% Vc/day produced monoclonal antibody titres of 5 g/L [3]. This systematic approach allowed them to identify the best parameters much faster than relying on trial and error. Plus, the results from these small-scale tests are highly reliable when scaled up to larger systems.
Scalability and Data Reproducibility
Mini bioreactors excel at predicting how processes will perform on a larger scale. In December 2015, UCB Pharma conducted a study comparing the ambr™ 48 system, which uses 15 mL vessels, with larger bioreactors of 2 L, 80 L, and 400 L capacities. The results showed that the mini bioreactors closely matched the larger systems in terms of cell growth, product titres, metabolite profiles, and key product quality attributes like charge variants and molecular weight species [1]. This capability is especially impactful for industries like cultivated meat production.
These systems also feature automated process control, which ensures precise management of pH, dissolved oxygen, temperature, and feeding schedules without manual intervention [8][3]. Automation reduces variability between experiments, giving researchers confidence that results from small-scale trials will hold up in large-scale manufacturing.
Lower Resource Consumption
Mini bioreactors operate with much smaller volumes, typically ranging from 800 µL to 15 mL, which significantly reduces the amount of growth media needed compared to traditional systems. For instance, the BioLector µ-bioreactor works with a final volume of just 800 µL [7]. In January 2021, researchers used this system to screen 22 E. coli expression clones and compared the results with those from a 30 L stirred tank reactor. The study found identical clone rankings and growth characteristics across both scales, proving that the 800 µL system can replace larger, resource-heavy experiments during early development stages [7].
In addition to media savings, these systems cut down on labour costs through automation and require only minimal sample volumes for detailed monitoring. Many mini bioreactors also use single-use, disposable vessels, removing the need for water, energy, and chemicals typically required for cleaning and sterilising stainless steel equipment. This not only saves resources but also simplifies operations.
Recent Studies on Mini Bioreactor Use
Stirred-Tank Mini Bioreactors for Cell Proliferation Media
Stirred-tank mini bioreactors play a crucial role in optimising growth media for cultivated meat. Systems like the ambr™ 15 and ambr250 are designed to mimic the conditions of much larger bioreactors - typically ranging from 3 to 400 litres - while working with volumes as small as 10–15 mL [1]. This allows researchers to test a wide variety of media formulations across up to 48 vessels, all without the hefty resource demands of larger-scale equipment.
Recent studies have verified that these systems successfully replicate the performance of large-scale bioreactors. Unlike traditional shake flasks, stirred-tank systems provide automated control over essential parameters such as pH, dissolved oxygen, and temperature. This level of control is critical for maintaining the consistency needed in cultivated meat cell cultures [1]. These findings open the door for developing more mini bioreactor systems tailored to specific cell culture needs.
Air-Lift Mini Bioreactors for Low Shear Stress Conditions
Air-lift bioreactors address a significant challenge in cultivated meat production: protecting delicate cells from mechanical damage. Instead of mechanical impellers, these systems use gas-driven circulation to create environments with low shear stress. This makes them ideal for adherent cells, such as bovine satellite cells, especially when using microcarriers that provide a high surface-to-volume ratio for cell attachment [6][10].
"Adherent cell cultures are necessary for eventual tissue formation and integration with edible scaffolds, allowing both proliferation and tissue formation to occur in the same production vessel."
- Saam Shahrokhi, VP of Technology, Mission Barns [6]
The gentle mixing offered by air-lift systems also supports bead-to-bead transfer, enabling cells to migrate naturally between microcarriers without relying on harsh enzymatic treatments. This process is vital for producing the vast number of cells - 10¹² to 10¹³ - needed to generate 10–100 kg of cultivated meat [10]. Alongside these systems, multi-well plate setups add another layer of versatility for high-throughput testing.
Multi-Well Plate Mini Bioreactors for Parallel Testing
Multi-well plate (MTP) systems have revolutionised high-throughput media screening by enabling real-time monitoring of critical parameters, a capability previously limited to larger bioreactors. For instance, the BioLector system uses 48-well Flowerplates with a working volume of 800 µL, providing live data on cell growth, pH, dissolved oxygen, and oxygen transfer [7][11].
In January 2025, a study demonstrated the successful scaling of CHO cultures from 96-well MTPs (400 µL) to 600 mL stirred-tank reactors. Remarkably, the process achieved identical antibody titres and metabolite profiles using the µTOM device [11]. These systems now even support fed-batch operations through enzymatic substrate release, allowing researchers to simulate industrial conditions in a high-throughput plate format [7].
Miniature bioreactors
Comparison of Mini Bioreactor Systems
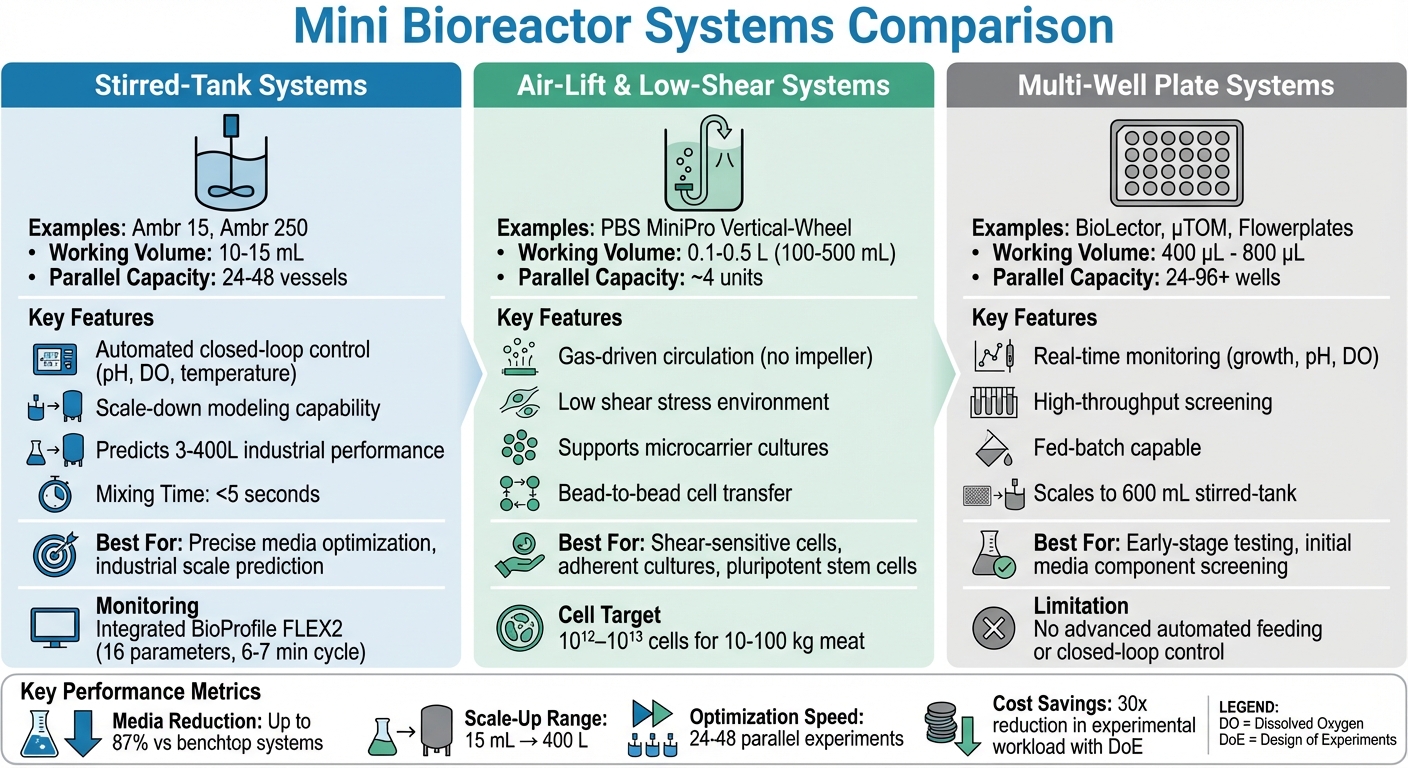
Comparison of Mini Bioreactor Systems for Cultivated Meat Media Optimization
Choosing the right mini bioreactor for cultivated meat media optimisation depends heavily on research objectives and the specific needs of the cell line. Stirred-tank systems like the Ambr 15 and Ambr 250 are a popular choice, offering automated closed-loop control of critical parameters such as pH, dissolved oxygen, and temperature. These systems can handle 24–48 parallel vessels, each with a working volume of 10–15 mL, making them ideal for scale-down modelling and predicting media performance in industrial-scale processes [4][3][1]. Their ability to closely mimic large-scale conditions makes them particularly useful for precise media optimisation in cultivated meat research [12][1].
On the other hand, air-lift and low-shear systems use gas sparging or vertical-wheel mixers to create a gentle circulation environment. A good example is the PBS MiniPro Vertical-Wheel bioreactor, which operates with volumes ranging from 0.1 to 0.5 L while offering precise control over gas exchange, pH, and medium changes [5]. These systems are especially effective for shear-sensitive cells, like pluripotent stem cells, as they help maintain cell quality and aggregate morphology. However, their throughput tends to be lower, typically supporting around four parallel units [5].
Multi-well plate systems focus on high-throughput screening, allowing researchers to test a wide range of variables simultaneously. Available in formats like 24-, 96-, or even higher well numbers, these systems are excellent for initial testing of media components. However, they lack the advanced automated feeding and closed-loop control capabilities seen in stirred-tank systems. With working volumes under 15 mL, they are better suited for early-stage experimental setups rather than comprehensive process optimisation [4]. These differences in design and functionality also influence operational metrics, such as mixing efficiency.
Mixing efficiency is a critical factor in mini bioreactor applications, particularly when addressing scale-up challenges. Laboratory-scale stirred-tank bioreactors, for instance, achieve mixing times under five seconds, which is essential for consistent biomass production [12]. In contrast, larger bioprocesses often face a drop in biomass yield - up to 20% - when scaling from 3 L to 9,000 L due to environmental inconsistencies [12]. To tackle these issues, modern mini-stirred-tank reactors now feature integrated automated analysers like the BioProfile FLEX2. These analysers can monitor up to 16 cell culture parameters within a cycle time of just 6–7 minutes [2].
"Utilising these integrated systems will support scientists to more easily run full QbD studies, without causing a sampling bottleneck or needing additional staff resources."
- Dr. Barney Zoro, ambr Product Manager, Sartorius Stedim Biotech [2]
sbb-itb-ffee270
Challenges in Scaling Up Mini Bioreactor Results
Mini bioreactors are invaluable for high-throughput experimentation, but scaling their results to industrial-scale cultivated meat production is no small feat. The process is riddled with challenges, particularly in maintaining consistent fluid dynamics and biological outcomes across vastly different scales.
One of the main hurdles is ensuring consistent fluidic properties - like energy dissipation, oxygen transfer (kLa), and cell aggregate suspension - when moving from mini bioreactors to larger systems. As Sharon Harvey, Director of Product Management and Strategy at PBS Biotech, explains:
"We had to match energy dissipation, oxygen transfer, and cell aggregate suspension at a fraction of the volume" [14].
This consistency is hard to achieve because mini bioreactors are designed for low-shear environments, which are ideal for fragile cell types. However, as the scale increases, shear forces tend to rise, potentially damaging these sensitive cells. Studies reveal that this mismatch can cause significant performance gaps, with specific product titres in benchtop reactors dropping by as much as 50% compared to titre plate experiments when growth conditions aren’t perfectly aligned [13].
Another limitation is the small operating volume of mini bioreactors, typically around 10–15 mL. This restricts the amount of in-process sampling and introduces variability, with product titres showing up to 20% differences between scales [9][13]. Reducing the operating volume below 10 mL often compromises the results, further limiting the flexibility for process monitoring and optimisation [9].
Biological variability adds yet another layer of complexity. Even when parameters like pH and metabolite levels are consistent, variations in viable cell density and product titres between mini bioreactor runs are common [1]. Each new cell line requires careful adjustments to scale-down models to account for these differences. For example, researchers must measure pH samples immediately after collection to avoid CO₂ degassing, which can artificially alter pH readings [9].
Lastly, the engineering challenge of miniaturisation cannot be overlooked. Designing compact vessels that replicate fluidic properties reliably while integrating advanced measurement tools - like 4 mm dissolved oxygen sensors and mass flow controllers - demands significant precision and innovation [14].
Scaling up from mini bioreactors to industrial systems is a balancing act that requires addressing these fluidic, biological, and mechanical challenges head-on to ensure consistent and reliable results.
Sourcing Mini Bioreactors via Cellbase
As research into mini bioreactors progresses, finding the right equipment becomes essential for refining media in cultivated meat production. The process of sourcing these systems can be time-consuming, especially when navigating fragmented supplier networks or using general-purpose lab equipment. Cellbase steps in to address this issue. It’s the first B2B marketplace designed specifically for the cultivated meat industry, offering a central hub where researchers and production teams can find the tools they need [15]. This streamlined approach not only simplifies the search for equipment but also supports smarter investments in systems tailored to cultivated meat research.
One of Cellbase’s standout features is its transparent pricing for mini bioreactor systems chosen specifically for cultivated meat production. For instance, the ABLE 5 mL Disposable Bioreactor is priced at £934.24, while the ABLE 30 mL Disposable Bioreactor is available for £467.14 [16]. These single-use systems are particularly effective for media optimisation, as they can cut media usage by up to 87% compared to larger benchtop systems. They also reduce downtime between experiments, making them a practical choice for research teams [17].
In addition to equipment, Cellbase connects users with Cell Ag Experts, who provide guidance on system selection, installation, and process optimisation [15][16]. This specialised support ensures that the chosen bioreactor is compatible with specific cell lines and animal-free media formulations - an important factor, given that growth media remains the most significant cost driver in cultivated meat production [15]. The platform’s "Ask us anything" feature further enhances this by enabling researchers to consult with specialists before making a purchase, reducing the likelihood of buying unsuitable equipment [15][16].
To make the process even smoother, Cellbase offers global shipping options, including specialised cold chain logistics [15]. By managing everything from equipment selection to delivery, the platform significantly reduces the administrative workload typically associated with B2B sourcing, allowing research teams to dedicate more time to optimising media [15].
Conclusion
Mini bioreactors have reshaped the way growth media is optimised for cultivated meat production. These systems allow researchers to test 24 to 48 different conditions at the same time, cutting optimisation timelines from months to just weeks [1][7]. Even at small volumes of 15 mL, the data they generate scales reliably to manufacturing volumes of 400 L or more, helping teams identify crucial process parameters early and avoid costly issues during scale-up [1]. This streamlined approach brings significant operational advantages to cultivated meat production.
With operating volumes ranging from 15–500 mL, mini bioreactors also dramatically reduce the use of costly growth factors and basal media. This is a major win since growth media is the biggest expense in cultivated meat production [3]. When paired with tools like Design of Experiments software or Bayesian optimisation, these systems can slash the experimental workload by up to 30 times compared to traditional methods [18].
For example, recent research using the Ambr® 250 system achieved CHO cell culture titres of 5 g/L by fine-tuning feeding rates and seeding densities [3]. Dr. Barney Zoro, ambr® Product Manager at Sartorius Stedim Biotech, highlights the value of these systems:
"Utilizing these integrated systems will support scientists to more easily run full QbD studies, without causing a sampling bottleneck or needing additional staff resources" [2].
As the field advances, choosing the right mini bioreactor becomes essential for unlocking these optimisation benefits. Platforms like Cellbase simplify this process by connecting researchers with verified suppliers, transparent pricing, and expert guidance, allowing teams to focus on refining media rather than navigating procurement challenges.
FAQs
Which mini bioreactor type is best for my cultivated meat cell line?
When choosing the right mini bioreactor, it’s essential to consider factors like the scale of your experiments, the level of control you need, and how well the system matches your cell line. The ambr™ mini-bioreactor system stands out as a flexible solution, offering fine-tuned control over key parameters like pH, oxygen levels, and temperature. For high-throughput testing, disposable options such as 50 mL Bioreactor Tubes can be a cost-effective alternative. Ultimately, your selection should fit the scale of your process and the specific requirements of your cell culture.
What scale-up parameters matter most when moving from 10–15 mL to litres?
When moving from small-scale bioreactors (10–15 mL) to litre-scale systems, oxygen transfer capacity (kLa) becomes a crucial factor. This ensures that cells receive enough oxygen to maintain growth and productivity. Achieving consistent oxygen transfer across different scales is essential for maintaining similar cell performance.
In addition to oxygen transfer, factors like seeding density and feeding rates play a significant role in influencing cell behaviour during scale-up. High-throughput platforms, such as the Ambr®250, can be invaluable for fine-tuning these parameters, enabling more reliable process development for cultivated meat production.
How do I choose the right sensors and sampling plan for tiny volumes?
When working with tiny bioreactor volumes, the choice of sensors hinges on the specific analytes you need to monitor and the reliability of the technology. For example, Raman spectroscopy is highly effective for tracking metabolites like lactate and glucose. On the other hand, 2D-fluorescence is particularly good at detecting ammonium levels.
For sampling in these small-scale systems, it's crucial to adopt automated, aseptic techniques that minimise contamination risks while preserving sample integrity. Pairing these approaches with model-based strategies can further improve the accuracy of your data, ensuring more precise process control.
Finally, it’s a good idea to consult with sensor suppliers to confirm compatibility with your setup. This step can help fine-tune your system for cultivated meat research and ensure optimal performance.











