Hydrogel scaffolds are critical for cultivated meat production, providing a 3D framework for cell growth and tissue formation. However, ensuring their safety and effectiveness requires thorough biocompatibility testing. Key challenges include:
- Chemical Residues: Toxic by-products from polymerisation and crosslinking agents can harm cells.
- Surface Chemistry Issues: Synthetic hydrogels often lack the bioactivity needed for cell adhesion.
- Immune Responses and Degradation: Some scaffolds provoke inflammation or degrade in ways that harm surrounding tissues.
Solutions to these challenges include purification methods, surface modifications (e.g., RGD peptides), and hybrid scaffold designs that combine synthetic and natural materials. Testing methods like cytotoxicity assays, mechanical property evaluations, and degradation studies ensure scaffolds meet both safety and functional requirements. Platforms like Cellbase simplify sourcing food-grade, GMP-compliant materials tailored for cultivated meat.
3D Hydrogel Scaffolds For Articular Chondrocyte Culture & Cartilage Generation l Protocol Preview
Common Challenges in Biocompatibility Testing
Biocompatibility testing for hydrogel scaffolds comes with its fair share of hurdles, particularly when it comes to ensuring cell viability and effective tissue formation. The main culprits? Chemical residues, surface properties, and degradation behaviour. These factors can significantly impact cell adhesion, growth, and survival. Let’s take a closer look at these challenges.
Residual Toxicity from Chemical Components
Safety is a top priority in cultivated meat production, and controlling residual toxic chemicals is a critical part of the process. Unreacted monomers from free-radical polymerisation, such as HEMA and acrylates, can seriously jeopardise cell survival. Acrylates are especially problematic, being more toxic than methacrylates, which themselves are more harmful than acrylamides [2].
Crosslinkers like ethylene dimethacrylate can leave behind toxic residues that don’t degrade easily [2]. Additionally, polymerisation triggers - such as initiators and radical-inducing agents - pose risks if they’re not fully reacted or properly removed [2].
To tackle this, purification through dialysis is often employed to eliminate these residual monomers and crosslinking agents before scaffolds are seeded with cells [2]. Achieving high conversion rates during polymerisation is also key, particularly for in situ gelation methods where leaching risks are heightened [2]. A systematic assessment approach, in line with ISO 10993 standards, can help pinpoint the source of cytotoxicity - whether it’s sterilisation residues, pH changes, or medium absorption - rather than relying on assumptions from existing literature [4].
Surface Chemistry Problems Affecting Cell Adhesion
Synthetic hydrogels like PEG, PHEMA, and PVA are naturally hydrophilic and bioinert. While this reduces the risk of triggering a foreign body response, it also makes it harder for serum proteins to attach [2]. Christopher D. Spicer from the University of York highlights the issue:
"The high hydrophilicity of PHEMA renders it bioinert, resisting cell and protein adhesion" [2].
Unlike the native extracellular matrix, which provides the necessary chemical signals for cell binding, these synthetic materials lack such cues. As a result, cells tend to adopt a rounded shape, indicating poor interaction with the scaffold material [2]. Moreover, the absence of sufficient surface charge means these scaffolds fail to leverage electrostatic interactions essential for initial cell adhesion [2].
Interestingly, researchers have found that adding micrometre-scale topographical patterns to PHEMA surfaces can help human mesenchymal stem cells spread and elongate, overcoming some of the material’s limitations [2]. Spicer notes:
"In contrast to the rounded morphology adopted on flat surfaces, indicative of poor interactions with the underlying material, cells were able to spread and elongate in response to the topographical cues provided" [2].
Immune Response and Degradation By-products
Scaffolds can provoke immune responses, leading to fibrous encapsulation that isolates the material [2]. This issue is particularly pronounced with chemical crosslinking agents like glutaraldehyde, which are known to trigger strong inflammatory reactions. For instance, in rat subcutaneous implantation studies, glutaraldehyde-crosslinked sponges developed thick tissue layers (0.85 ± 0.34 mm), while sponges crosslinked with microbial transglutaminase showed much thinner layers (0.19 ± 0.16 mm) [5].
The timing and by-products of scaffold degradation add another layer of complexity. Polyester-based scaffolds, such as PLA or PGA, release acidic monomers as they break down, which can lead to local pH increases and tissue damage. As Spicer explains:
"The build-up of glycolic and lactic acid monomers following the degradation of poly(ester)-based scaffolds has been shown to lead to a local increase in pH and resultant tissue damage" [2].
Scaffolds that degrade too quickly lose their structural integrity, which is crucial for cell adhesion and tissue development [5]. For example, after one month of implantation, EDC-crosslinked gelatin sponges retained only 2.7% ± 1.7% of their volume, whereas glutaraldehyde-crosslinked sponges maintained 69.1% ± 4.3% [5]. Even materials considered bioinert, like PEG, can sometimes provoke immune reactions, such as the development of anti-PEG antibodies in certain patients, complicating their use in vivo [2].
Standard Testing Methods for Biocompatibility
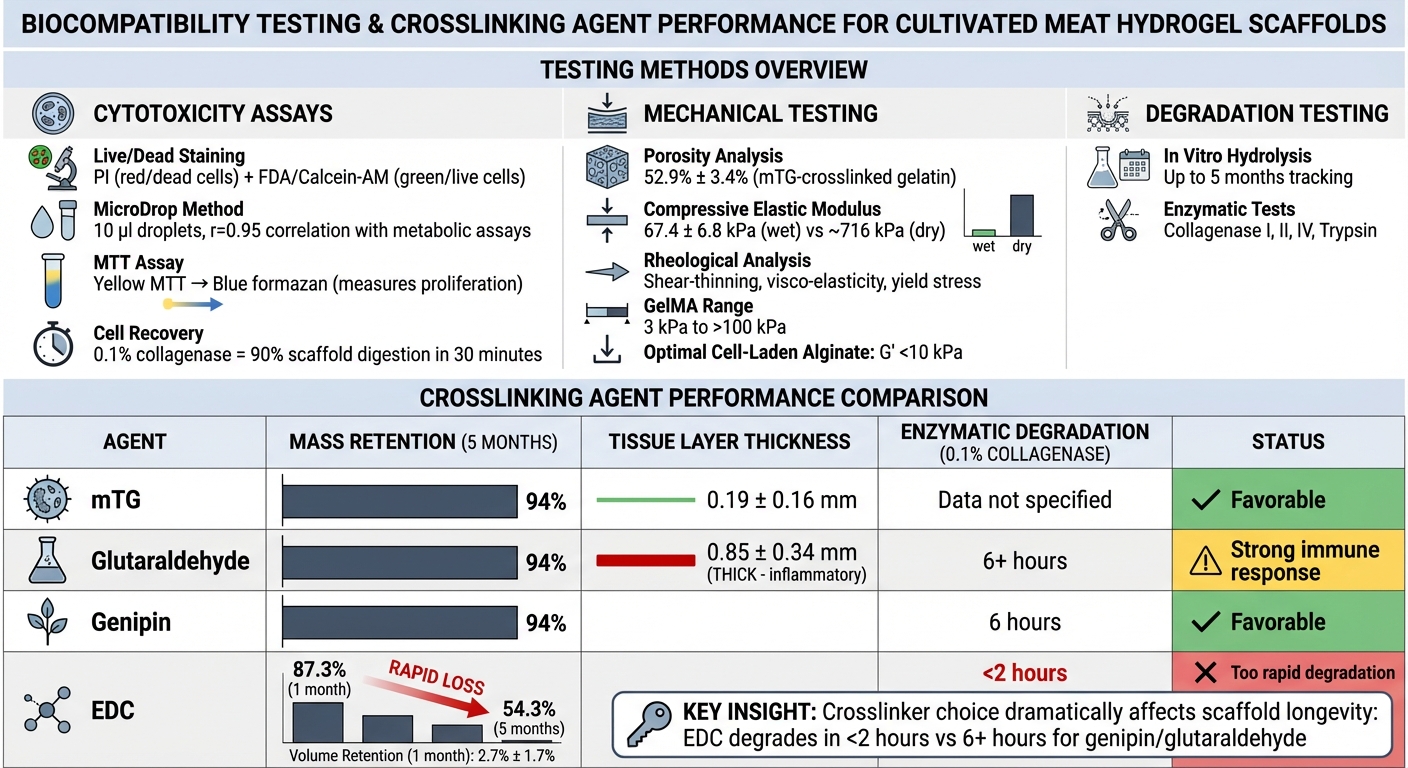
Biocompatibility Testing Methods and Crosslinking Performance Comparison for Hydrogel Scaffolds
Evaluating biocompatibility involves a combination of cytotoxicity tests, mechanical property assessments, and degradation studies. These rigorous methods ensure that hydrogel scaffolds not only support cell growth but also meet the safety and texture standards needed for cultivated meat.
Cytotoxicity and Cell Viability Assays
Live/Dead staining is a trusted method for evaluating cell viability within three-dimensional hydrogel scaffolds. This process uses propidium iodide (PI) to stain dead cell nuclei red, while fluorescein diacetate (FDA) or Calcein-AM highlights live cells in green. This dual-staining approach provides a clear visualisation of cell distribution throughout the scaffold matrix [6][7]. The MicroDrop method, which uses 10 µl droplets, has shown a strong correlation (r=0.95) with metabolic assays, making it a reliable alternative [6].
The MTT assay is another valuable tool, measuring cell proliferation and metabolic activity. It works by converting light yellow MTT into dark blue formazan, offering an effective way to compare long-term cell growth across various scaffold types [7]. However, in viscous hydrogels, the CCK8 assay may produce false-positive results due to non-specific interactions [6]. To recover cells from 3D scaffolds, a 0.1% collagenase solution is highly effective, digesting up to 90% of the scaffold within 30 minutes while minimising cellular damage [7].
Once cell viability is confirmed, the next step is to evaluate the scaffold's structural and mechanical properties.
Mechanical and Structural Property Testing
Mechanical testing ensures that scaffolds can physically support cell growth while allowing for proper nutrient diffusion. Porosity analysis is critical for maintaining cell viability, as it ensures adequate movement of nutrients, oxygen, and waste in 3D cultures [1]. The compressive elastic modulus in a hydrated state is used to measure how closely the scaffold mimics the texture of conventional meat. For example, gelatin sponges crosslinked with microbial transglutaminase (mTG) demonstrated a porosity of 52.9% ± 3.4% and a compressive elastic modulus of 67.4 ± 6.8 kPa when wet [7].
For bioprinted scaffolds, rheological analysis plays a key role in assessing properties like shear-thinning behaviour, visco-elasticity, and yield stress. These parameters ensure smooth extrusion during printing and structural integrity after deposition [3]. GelMA hydrogels, for instance, can be tailored to achieve stiffness ranging from approximately 3 kPa to over 100 kPa, depending on the tissue requirements. However, for cell-laden alginate, optimal printability and cell viability are typically linked to storage modulus (G') values below 10 kPa [3]. As Rency Geevarghese and colleagues have noted:
"Printability, stability, and biocompatibility are not independent and must be adjusted carefully to counterbalance each other" [3].
Beyond immediate mechanical properties, long-term scaffold stability is equally important.
Long-term Biodegradation and Stability Testing
To ensure scaffolds remain functional during cell development, degradation testing evaluates their longevity. In vitro hydrolysis tests track mass loss over extended periods - up to five months in aqueous environments - to assess stability [7]. Enzymatic degradation tests, using proteases like Collagenase I, II, IV, and Trypsin, provide additional insights into how scaffolds behave under biological conditions [7].
The type of crosslinker significantly impacts degradation rates. For instance, in hydrolysis tests, gelatin sponges crosslinked with mTG, glutaraldehyde, or genipin retained 94% of their original mass after five months. In contrast, EDC-crosslinked sponges showed a sharp decline in stability, with mass dropping to 87.3% after one month and only 54.3% remaining after five months [7]. During enzymatic degradation with 0.1% collagenase, EDC sponges dissolved almost completely within two hours, whereas genipin-crosslinked sponges took six hours to degrade fully [7].
Mechanical stability also diminishes significantly after water absorption. For example, the compressive elastic modulus of dry mTG sponges, which is approximately 716 kPa, drops to around 67 kPa when wet [7]. Testing mechanical properties in a hydrated state is therefore essential for accurate evaluation.
sbb-itb-ffee270
Solutions to Improve Hydrogel Biocompatibility
When hydrogel biocompatibility falls short, there are proven methods to refine scaffold performance. These approaches address challenges like chemical toxicity, weak cell adhesion, and rapid degradation, ensuring scaffolds perform better in cultivated meat production. The focus is on improving cell attachment, adjusting mechanical properties, and managing degradation rates.
Surface Modifications for Better Cell Attachment
Synthetic hydrogels, such as PEG, PVA, and PHEMA, are naturally bioinert, making cell attachment difficult without additional cues. A common solution is incorporating RGD peptides, which provide the binding sites cells need. Gelatin and its derivative, GelMA, naturally contain these peptides, making them widely used in cultivated meat scaffolds. Researchers at Silesian University of Technology highlighted this:
"Gelatin has been identified as a promising bioink component endorsing cell growth due to the presence of cell attachment peptide motifs like RGD (arginine–glycine–aspartic acid)" [3].
Other techniques include micrometre-scale topographical patterning, which introduces physical cues to encourage cell spreading on otherwise flat surfaces [2]. Adjusting surface charge can also enhance electrostatic interactions with cells [2]. Additionally, synthetic polymers can be modified with bioactive motifs, such as RGDS or IKVAV, to support cell binding more effectively [2].
Material Composition and Hybrid Scaffold Designs
Hybrid scaffolds combine the strength of synthetic polymers with the bioactivity of natural materials, addressing the limitations of single-component designs. Synthetic polymers like PEG and PCL offer predictable chemistry and strong mechanical properties, while natural polymers such as collagen, chitosan, and alginate provide environments that mimic the extracellular matrix (ECM), promoting cell adhesion and growth [9][2].
For example, a 2023 study published in Scientific Reports demonstrated a hybrid scaffold made by combining a PEG-gelatin hydrogel with a PCL mesh. This design supported the formation of a tight epithelial cell layer using MDCK cells over nine days, with the PCL mesh providing mechanical support for the 100 µm-thick hydrogel membrane [8]. Similarly, a 2012 study showed that immobilising gelatin onto hydrophobic PCL film surfaces enhanced Human Umbilical Vein Endothelial Cell (HUVEC) attachment and growth, with better results linked to higher amounts of immobilised gelatin [10].
Adding carboxymethyl cellulose (CMC) to alginate-based inks can improve both mechanical properties and swelling capacity through electrostatic interactions [3]. Mechanically robust hydrogels typically contain 0.1–10% polymer by weight, but gels with pores smaller than 10 µm may hinder cell movement and infiltration [2].
These strategies not only improve cell compatibility but also allow for precise control over scaffold longevity, which is closely tied to degradation rates.
Controlled Degradation through Crosslinking Adjustments
Crosslinking density plays a key role in both degradation rates and mechanical stiffness. Dual crosslinking methods, such as combining ionic crosslinking (e.g., using CaCl₂ for alginate) with photo-crosslinking (e.g., UV curing for GelMA), offer better control over scaffold stability. The ionic bonds provide temporary support, while covalent bonds ensure long-term structure [3].
GelMA hydrogels can achieve a wide range of storage moduli (G') - from around 3 kPa to over 100 kPa - depending on polymer concentration and UV exposure [3]. For cell-laden alginate, G' values below 10 kPa are often optimal for maintaining printability and cell viability [3]. Including degradable linkages, such as disulfide bonds or polyester sequences, allows scaffolds to break down into resorbable macromers that cells can replace with native ECM [2]. However, polyester-based crosslinks like PLA or PGA require careful pH monitoring, as the release of glycolic or lactic acid can lead to tissue damage from acidity [2].
Using lithium phenyl-2,4,6-trimethylbenzoylphosphinate (LAP) as a photoinitiator for UV curing is another way to improve cytocompatibility compared to older methods [3][8]. Maintaining strict temperature control at 37°C and adhering to precise mixing protocols ensures uniform crosslinking and predictable degradation [3].
Using Cellbase for Scaffold Procurement
Finding the right biocompatible hydrogel scaffolds for cultivated meat production can be tricky, especially when relying on general lab suppliers who may lack expertise in food-grade materials and regulatory compliance. Cellbase steps in to solve this problem. As the first specialised B2B marketplace tailored to the cultivated meat industry, it connects researchers and production teams with trusted suppliers for scaffolds, bioreactors, growth media, and other essential materials. Reliable procurement is crucial for accessing scaffolds that meet the strict biocompatibility standards required in this field. Here's how Cellbase addresses these challenges through supplier verification and its streamlined catalogue system.
Verified Suppliers for Cultivated Meat
Cellbase focuses on suppliers that meet Good Manufacturing Practice (GMP) standards and cater specifically to the cultivated meat industry. For example, the platform offers edible scaffolds like alginate, which not only mimic the texture of meat but are also already approved as food ingredients - saving time and costs by eliminating separation steps. Patrick Inomoto, Technical Director at Innocent Meat, highlights this benefit:
"Alginate is ideal because it mimics the texture of meat very well and is already approved as a food ingredient" [11].
Suppliers listed on Cellbase are rigorously vetted to ensure their products meet cultivated meat requirements. This includes verifying advanced production techniques like cryogelation, which forms interconnected macroporous networks - crucial for large-scale cell growth.
Streamlined Procurement Processes
Beyond verified standards, Cellbase simplifies the procurement process with its searchable catalogues. Each listing includes detailed technical attributes, such as GMP compliance, food-grade certification, and specific porosity ranges, making it easier for buyers to find the right materials quickly. The platform also facilitates direct communication with suppliers, allowing teams to request custom properties like tailored crosslinking for controlled degradation or bioactive coatings such as RGD peptides. This targeted approach removes the hurdles often encountered with non-specialist suppliers, reducing technical risks and speeding up sourcing decisions.
Conclusion
Biocompatibility testing for hydrogel scaffolds in cultivated meat production is a balancing act involving several interconnected factors. The "biocompatibility-printability-stability" trilemma highlights how improving one property can sometimes compromise another. For instance, using high polymer concentrations can enhance structural stability but may also increase shear stress during extrusion, which could harm cells [3]. Similarly, degradation byproducts from materials like PLA can negatively affect surrounding cells [2][1].
Testing methods need to address these complex interactions to ensure scaffolds meet the rigorous standards of cultivated meat production. Techniques such as cytotoxicity assays, mechanical property assessments, and long-term degradation studies collectively help ensure that scaffolds maintain cell viability throughout their lifecycle. As Małgorzata Katarzyna Włodarczyk-Biegun explains:
"Printability, stability, and biocompatibility are not independent and must be adjusted carefully to counterbalance each other" [3].
Innovative approaches like dual crosslinking - which combines ionic and covalent methods - can achieve a storage modulus ranging from ~3 kPa to over 100 kPa while still supporting cell viability [3]. Other advancements, such as surface modifications with bioactive peptides like RGD and hybrid scaffolds that mix natural and synthetic polymers, enhance biocompatibility. Controlled degradation through precise crosslinking further refines scaffold performance. However, challenges remain, such as the batch-to-batch variability of natural polymers, which can affect consistency in large-scale production [1]. These technical adjustments are essential for sourcing materials that meet the specific demands of cultivated meat production. Ultimately, achieving the right balance of chemical, mechanical, and biological properties is key to the success of hydrogel scaffolds.
Cellbase offers a valuable solution by connecting cultivated meat teams with verified, GMP-compliant suppliers. Its platform provides detailed technical specifications, making it easier to identify suitable materials and reduce technical hurdles. In an industry where material consistency directly impacts production outcomes, this dedicated marketplace simplifies the transition from laboratory testing to large-scale manufacturing.
FAQs
How can I identify toxic residues in a hydrogel scaffold?
To spot toxic residues in a hydrogel scaffold, biocompatibility testing is key. This process focuses on detecting cytotoxic responses, which indicate harmful effects on cells. A widely used approach is cytotoxicity assays, such as direct cell sampling, which evaluates cell viability and behaviour.
Signs to look out for include cell membrane damage, apoptosis (programmed cell death), or outright cell death. By combining these methods, you can thoroughly detect and assess any harmful residues that could hinder cell growth.
What tests best predict cell adhesion in 3D hydrogels?
Cell adhesion assays are a reliable way to evaluate how well cells adhere to 3D hydrogels. These tests measure key aspects such as cell attachment and growth on hydrogel scaffolds, offering important information about the material's compatibility with biological systems.
How can I tune scaffold degradation without harming cells?
To fine-tune scaffold degradation without compromising cell health, you can adjust the hydrogel's chemical composition. For example, tweaking the crosslinking density or incorporating biodegradable linkages can help achieve a balance between stability and breakdown. Using particular polymers, like collagen-based hydrogels, offers another approach, enabling controlled degradation to promote cell growth and differentiation. Thoughtful adjustments ensure the scaffold degrades at a pace that supports cellular processes while keeping the cells viable.











