When producing cultivated meat, the thermal stability of scaffolds is critical. Scaffolds must maintain their structure at 37°C during cell culture and withstand sterilisation and cooking processes. Here’s a quick breakdown of the key materials and their performance:
- Collagen: Excellent for cell growth but varies in stability. Mammalian collagen is more reliable than fish or marine sources, which degrade at lower temperatures.
- Alginate and Polysaccharides: Highly heat-resistant but lack natural cell-binding sites, requiring surface modifications for effective cell attachment.
- Synthetic Polymers: Durable and thermally stable, but often non-edible, adding complexity to production.
- Decellularised ECM: Plant-based options like asparagus offer heat resilience, edibility, and strong cell attachment but may have variability in structure.
For scalable solutions, platforms like Cellbase provide pre-verified biomaterials tailored to cultivated meat, ensuring thermal stability and production efficiency. Choosing the right scaffold depends on balancing thermal performance, biological compatibility, and production requirements.
Lecture 22: Scaffold Fabrication Techniques in Tissue Engineering | ISSS PMRF Lecture Series
1. Collagen-Based Scaffolds
Collagen, the most plentiful protein in the extracellular matrix, is highly compatible with cell attachment and growth. However, its sensitivity to heat poses a real challenge for use in cultivated meat production. The key lies in preserving its unique triple helical structure, which breaks down when exposed to temperatures above its denaturation point. This denaturation temperature (T₍d₎) is critical because, once exceeded, collagen transforms into gelatin, losing its ability to form fibrils and support cell growth. If T₍d₎ is below 37°C - the standard culture temperature - this structural collapse becomes inevitable, making thermal stability a major consideration when selecting collagen sources.
Thermal stability in collagen varies significantly depending on its source. Bovine skin collagen, for instance, has a T₍d₎ of 40.4°C, which makes it stable under typical culture conditions. In contrast, porcine collagen, with a T₍d₎ of 37.0°C, sits right at the edge of usability. Marine collagen sources are even less stable: silver carp collagen denatures at 28.4°C, and Deep-Sea Redfish collagen loses its structure at a mere 15.7°C. These differences are largely due to hydroxyproline content - a key factor in thermal stability. For example, bovine collagen has about 94 hydroxyproline residues per 1,000, while Deep-Sea Redfish collagen contains only 54 [4]. These variations not only affect how collagen performs but also influence decisions around sterilisation and extraction methods.
Sterilisation processes present another hurdle for collagen stability. High-temperature steam sterilisation can’t be used because it disrupts the hydrogen bonds that stabilise the triple helix [6]. While dry heat sterilisation preserves the structure better, it can still cause some chemical crosslinking [5]. Chemical crosslinking, using agents like glutaraldehyde, offers a solution by raising the glass transition temperature from 60°C to 145°C. However, this approach adds complexity to the processing [7].
Extraction methods also play a role in determining collagen stability. For example, alkali-soluble collagen extracted from pig skin has a T₍d₎ of just 34.5°C, which is below the desired threshold for cell cultures. On the other hand, acid-soluble collagen exhibits higher stability, typically 4–5°C above that of alkali-soluble collagen [4]. Without chemical crosslinking modifications, these thermal limitations make unmodified collagen scaffolds less suitable for cultivated meat production.
2. Alginate and Polysaccharide Scaffolds
Alginate stands out as a resilient option for cultivated meat scaffolds, especially when compared to heat-sensitive materials like collagen. Unlike protein-based scaffolds, alginate and other polysaccharides can withstand temperatures of 37°C without breaking down. Derived from seaweed, alginate is prized for its stability and non-toxic nature, making it a practical choice for these applications [9]. In fact, thermal gravimetric analysis shows that alginate maintains its structure across a wide temperature range, from 25°C to 600°C [8].
That said, alginate isn't perfect. It degrades quickly in culture and lacks the cell-binding domains needed for proper cell attachment. To overcome these shortcomings, researchers often blend alginate with synthetic polymers like polyvinyl alcohol (PVA) and add mineral fillers such as hydroxyapatite (HAp). These composite scaffolds not only enhance mechanical properties, achieving compressive strengths of 8–12 MPa, but also support the growth of mesenchymal stem cells over 14–21 days at 37°C [8].
Another advantage of polysaccharide scaffolds is their ability to withstand sterilisation processes. Thanks to their thermal resilience, researchers can avoid heat-based sterilisation methods that might damage the scaffold's delicate structure. Instead, a 30-minute soak in 70% ethanol is commonly used. Porosity also plays a role in scaffold performance: PVA/CMC-based scaffolds have a porosity of 72%, while PVA/Alg-based scaffolds offer slightly higher porosity at 79% [8], which supports effective nutrient exchange. However, while these scaffolds retain their shape during culture, their lack of inherent cell-binding domains requires additional surface modifications to improve cell adhesion.
The main hurdle for polysaccharide scaffolds isn't heat tolerance - it's cell attachment. Materials like alginate, cellulose, and gellan gum naturally lack cell-binding motifs such as RGD sequences, which are crucial for adhesion. To address this, researchers modify scaffold surfaces to improve cell attachment and promote processes like migration, proliferation, and differentiation. Without these adjustments, cells struggle to adhere effectively, highlighting the need for further engineering to optimise these scaffolds for cultivated meat production. Improving cell adhesion remains a key focus as alternative scaffold materials are explored.
3. Synthetic Polymer Scaffolds
Synthetic polymers stand out for their impressive thermal stability. Take polycaprolactone (PCL), for instance - it maintains its structural integrity at 37°C and boasts a melting point far above typical production temperatures. This makes it ideal for extended culture periods and facilitates heat-based sterilisation during downstream processing.
However, sterilisation remains a tricky issue. Crystalline PLA, with a heat deflection temperature (HDT) of up to 135°C, can handle autoclave sterilisation. Polyhydroxybutyrate-co-valerate (PHBV) performs even better, offering a Vicat softening temperature of 143°C and an HDT of 105°C [11]. In contrast, amorphous PLA struggles under heat, with an HDT that can drop as low as 40°C [11], making it prone to deformation during sterilisation.
Advanced elastomers like PDT offer customisable thermal properties. By tweaking the ratio of flexible trimethylene carbonate segments, researchers can adjust the glass transition temperature between 10.14°C and 41.54°C [2]. This enables shape-memory functions that activate near body temperature, achieving recovery rates of over 95% after repeated deformation [2]. Additionally, trimethylene carbonate helps mitigate local acid degradation, a common issue with rigid polymers like PDLLA during long-term culture [2].
Despite their thermal strengths, synthetic polymers face challenges in biological integration. Unlike natural scaffolds derived from plants or algae, synthetic options such as polyvinyl pyrrolidone (PVP) and polyurethane are not edible [10]. This necessitates an expensive cell dissociation step after cell proliferation, complicating the production process. They also lack the cell-binding domains present in natural extracellular matrix proteins, requiring surface modifications to enhance cell adhesion [10].
Ultimately, the choice between synthetic and natural scaffolds hinges on the trade-off between thermal performance and biological compatibility. Synthetic polymers offer reliable mechanical support and excellent heat resistance but require additional engineering to mimic the cell-friendly environment that natural materials inherently provide. These factors underscore the balance between durability and biological functionality.
sbb-itb-ffee270
4. Decellularised Extracellular Matrix Scaffolds
Decellularised extracellular matrix (ECM) scaffolds provide a solid foundation for cell attachment, maintain thermal stability at 37°C, and can endure cooking temperatures. Among plant-derived scaffolds, asparagus stands out for its ability to support cell attachment and proliferation for up to 22 days in culture [12].
These scaffolds are highly porous and mechanically supportive. Decellularised asparagus scaffolds, for instance, retain around 93.5% porosity, with interconnected pores ranging from 8 to 80 μm in diameter [12]. This porous structure allows for continuous nutrient and gas exchange while also providing mechanical strength. With a Young's modulus of 4.9 ± 1.12 kPa, these scaffolds meet the optimal conditions for both myoblast growth and adipogenic differentiation [12]. The decellularisation process significantly reduces DNA content from 978 ± 62 ng/mg to 254 ± 60 ng/mg, preserving the cellulose-based matrix [12]. These characteristics make them well-suited to handle the thermal and mechanical demands of cultivated meat production.
One of the key advantages is their resilience to heat sterilisation, which often poses challenges for animal-derived scaffolds. For example, fish muscle collagen tends to lose its structure and develop a scaly texture when exposed to cooking temperatures. In contrast, plant-based ECMs hold their shape under heat. Research from January 2024 highlights that porcine adipose-derived mesenchymal stem cells grown on decellularised asparagus scaffolds show a 3.64-fold increase in viability over seven days, even when subjected to pan-frying conditions [12][9].
As noted in npj Science of Food:
Thermogravimetric analysis (TGA) revealed the thermal stability of decellularised plant scaffolds, crucial for potential applications in food products, including cultivated meat subjected to high-temperature cooking conditions. [12]
Unlike synthetic polymers, which need to be removed before consumption, decellularised plant scaffolds are naturally edible. They also enhance the Maillard reaction during cooking, contributing to browning and flavour development. This thermal stability not only meets the demands of cultivated meat production but also eliminates the need for costly cell dissociation steps, simplifying the overall process.
5. Cellbase
Finding scaffold materials with reliable thermal specifications is a persistent challenge for cultivated meat companies. The performance of these materials during bioprocessing and cooking hinges on accurate thermal data. However, traditional lab suppliers rarely provide the level of detail needed to determine if a material can retain its structural integrity throughout these processes. This is where Cellbase steps in. Designed as a specialised B2B marketplace for the cultivated meat sector, it connects researchers and production teams with suppliers who understand the thermal demands of this unique industry.
The platform addresses a critical technical gap by rigorously verifying thermal data. Biomaterials are categorised based on their physical properties - such as hydrogels, microcarriers, and porous scaffolds - making it simpler to find materials that can withstand specific thermal environments [13]. Some of the options available include plant-based materials like broccoli florets, glutenin wheat powder, and chickpea protein, as well as cellulose-based polymers such as cellulose acetate and bio-inks derived from basil or callus [13]. Each material listing includes thermal specifications verified through methods like thermogravimetric analysis (TGA), which tests stability under high-temperature cooking conditions [12].
Unlike general suppliers, Cellbase ensures that listed vendors assess cooking durability, examining how scaffolds perform in real-world scenarios such as pan-frying [12]. This guarantees that materials not only support cell growth at 37°C but also maintain their structure when exposed to heat. By providing verified listings, the platform helps companies avoid materials that lack sufficient thermal stability, reducing risks during production.
Additionally, Cellbase simplifies procurement by offering transparent material specifications and enabling direct communication with suppliers. This streamlined approach helps R&D teams and production managers make quicker sourcing decisions. For instance, polymers like PCL, known for their mechanical strength, need precise thermal monitoring to ensure compatibility with both bioreactor conditions and post-production processes [1]. By focusing solely on cultivated meat applications, Cellbase delivers industry-specific insights that generalist marketplaces simply cannot provide.
Pros and Cons
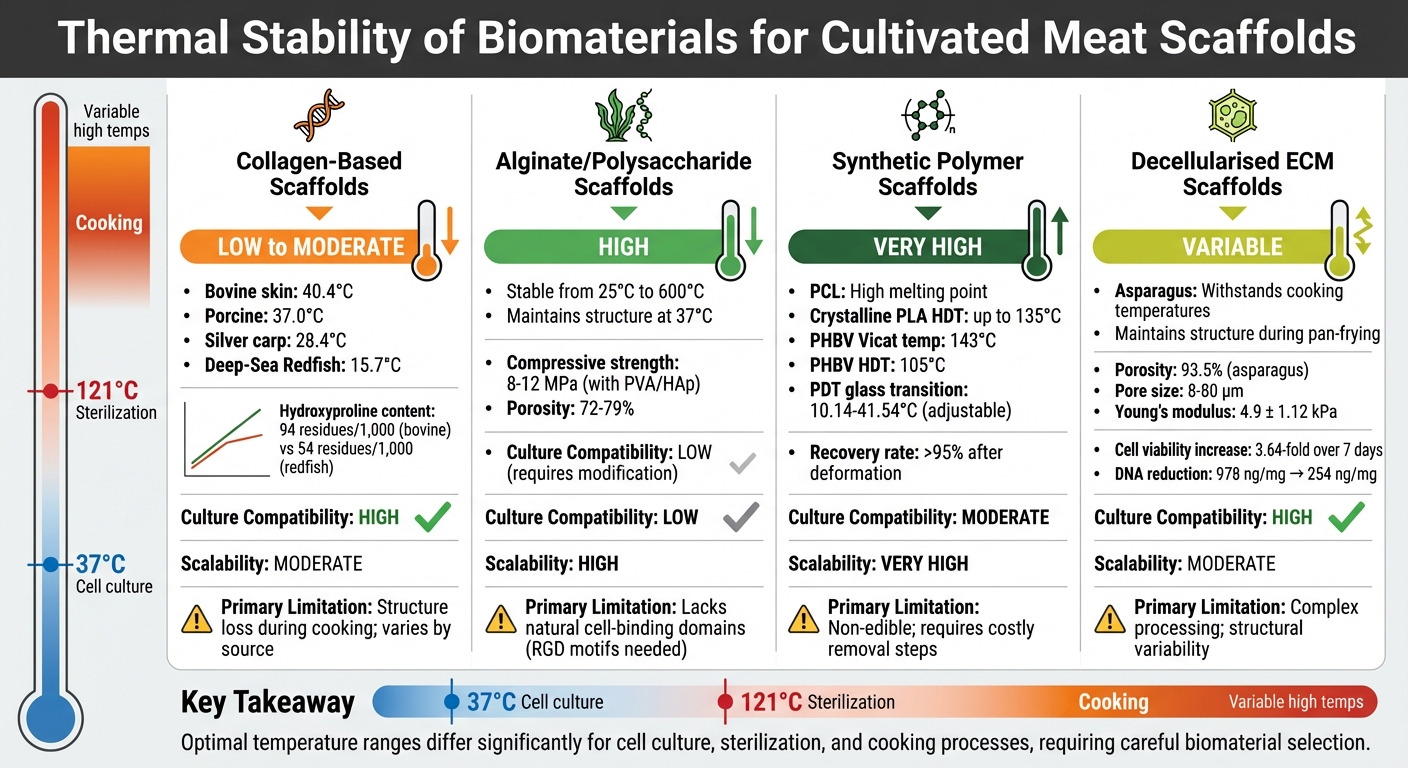
Thermal Stability Comparison of Biomaterials for Cultivated Meat Scaffolds
Here's a breakdown of the thermal performance and limitations for various biomaterial categories:
| Biomaterial Type | Thermal Stability | Compatibility with Culture | Scalability | Primary Limitation |
|---|---|---|---|---|
| Collagen-Based | Low (fish) to moderate (mammalian) | High; provides natural cell-binding sites | Moderate; limited by animal sourcing or fermentation costs | Potential structure loss during cooking; nutritional gaps [1] |
| Alginate/Polysaccharides | High biostability; resistant to degradation | Low; requires RGD motifs or surface modification for adhesion | High; cost-effective and widely available | Unfavourable nutritional profile; lacks natural cell-binding domains [1] |
| Synthetic Polymers | High; precise melting points (e.g. PCL) | Moderate; versatile chemistry but often requires cell dissociation | Very high; uniform production and long shelf-life | Often non-edible; requires costly removal steps; high medical-grade costs [1][10] |
| Decellularised ECM | Variable; depends on the source (plant/tissue) | High; maintains a natural 3D microenvironment | Moderate; relies on consistent plant/tissue sourcing | Complex processing; potential variability in structure [1][3] |
Plant proteins, such as wheat glutenin, show impressive thermal stability, withstanding autoclaving at 121°C for 15 minutes. However, they require surface modifications to support cell adhesion.
Synthetic polymers stand out for their uniformity and extended shelf-life [1][10]. Yet, their non-edible nature necessitates expensive post-culture removal processes.
Fish collagen is excellent for cell attachment but struggles with structural integrity during cooking, often resulting in a scaly texture [1].
Selecting the right biomaterial for cultivated meat is a careful balancing act. Factors like thermal stability, scalability, cell compatibility, and edibility all play a part in ensuring the scaffold remains intact from the culture phase all the way through to cooking. Thermal consistency, in particular, is key to maintaining scaffold integrity throughout the process.
Conclusion
Choosing the right scaffold for cultivated meat involves finding a balance between thermal stability and production efficiency. Each material comes with its own set of strengths, making certain options better suited for specific production and application needs. For instance, alginate and other polysaccharide scaffolds are highly stable and work well for large-scale production, though they often need surface modifications to improve cell adhesion [1]. On the other hand, synthetic polymers like PLA and PLGA provide consistency and long shelf life, but their non-edible nature means they must be removed after production [1][10].
When it comes to thermal stability, fish collagen struggles during cooking, while mammalian collagen holds up better at higher temperatures [1]. For applications involving cartilage or connective tissue, polycaprolactone (PCL) stands out due to its mechanical strength, although its lower melting point can be a limitation [1]. Meanwhile, plant-based proteins like wheat glutenin offer good thermal resilience but may require the addition of RGD motifs to enhance cell adhesion [1].
Beyond the material properties, how scaffolds are sourced plays a major role in their overall performance. Effective sourcing is key to avoiding complications. Platforms like Cellbase specialise in providing food-grade scaffolds tailored for cultivated meat production. As highlighted in npj Science of Food:
Adapting [medical scaffolds] for CM production requires complex modifications... which could compromise the final product quality [10].
By sourcing directly from Cellbase, producers can access pre-verified, food-grade materials designed specifically for cultivated meat, avoiding the delays and risks associated with repurposing medical-grade scaffolds [10].
Ultimately, the thermal properties of the biomaterial determine whether the scaffold can maintain its integrity from the bioreactor all the way to the cooked product. Aligning material characteristics with production needs - and sourcing from dedicated platforms like Cellbase - ensures both technical success and commercial feasibility.
FAQs
What thermal specs should a scaffold meet for culture, sterilisation and cooking?
A scaffold used in cultivated meat production needs to handle a variety of thermal challenges. It must endure sterilisation temperatures of approximately 121°C, remain stable under cell culture conditions, and maintain its integrity during cooking. While exact temperature requirements can differ based on the specific use case, these factors are crucial for ensuring the scaffold performs effectively throughout the process.
How can alginate scaffolds be modified to improve cell adhesion?
Alginate scaffolds can improve cell adhesion when their crosslinking process is fine-tuned. Using specific ionic crosslinking methods, researchers have achieved up to 82% cell attachment, thanks to enhanced surface coverage and better compatibility for cell growth.
When should you choose plant-based decellularised ECM over collagen or synthetic polymers?
Plant-based decellularised extracellular matrix (ECM) offers a natural and edible solution for creating scaffolds with vascular-like networks, essential for cultivated meat production. Typically sourced from plant leaves, these scaffolds are biodegradable and replicate the intricate structure of traditional meat. They enable cell attachment, growth, and development, making them ideal for forming realistic, edible tissue structures. By avoiding synthetic or animal-derived materials, they prioritise biocompatibility, safety, and environmental responsibility.











